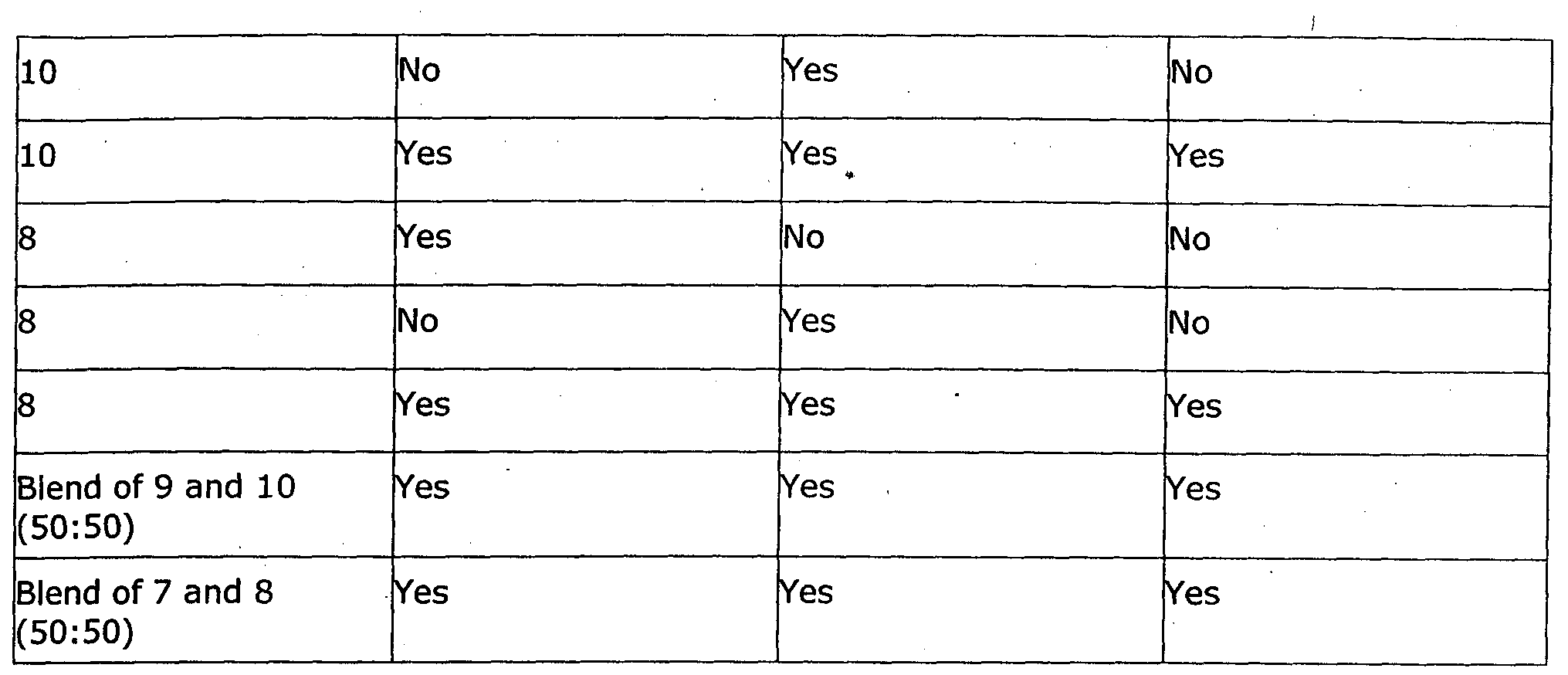
WO2014097309A1 - Stimuli responsive self cleaning coating - Google Patents
Stimuli responsive self cleaning coating Download PDFInfo
- Publication number
- WO2014097309A1 WO2014097309A1 PCT/IN2013/000101 IN2013000101W WO2014097309A1 WO 2014097309 A1 WO2014097309 A1 WO 2014097309A1 IN 2013000101 W IN2013000101 W IN 2013000101W WO 2014097309 A1 WO2014097309 A1 WO 2014097309A1
- Authority
- WO
- WIPO (PCT)
- Prior art keywords
- particles
- water
- coating composition
- aqueous coating
- anyone
- Prior art date
Links
- 238000000576 coating method Methods 0.000 title claims abstract description 247
- 238000004140 cleaning Methods 0.000 title claims abstract description 81
- 239000011248 coating agent Substances 0.000 title claims description 186
- 239000002245 particle Substances 0.000 claims abstract description 350
- 239000000203 mixture Substances 0.000 claims abstract description 218
- 229920000642 polymer Polymers 0.000 claims abstract description 179
- 239000008199 coating composition Substances 0.000 claims abstract description 159
- 230000002209 hydrophobic effect Effects 0.000 claims abstract description 149
- 239000006185 dispersion Substances 0.000 claims abstract description 132
- 239000002253 acid Substances 0.000 claims abstract description 81
- 239000002105 nanoparticle Substances 0.000 claims abstract description 57
- NIXOWILDQLNWCW-UHFFFAOYSA-N acrylic acid group Chemical group C(C=C)(=O)O NIXOWILDQLNWCW-UHFFFAOYSA-N 0.000 claims abstract description 55
- 229920001296 polysiloxane Polymers 0.000 claims abstract description 54
- 239000000758 substrate Substances 0.000 claims abstract description 38
- 230000003075 superhydrophobic effect Effects 0.000 claims abstract description 37
- 230000002441 reversible effect Effects 0.000 claims abstract description 36
- 230000007613 environmental effect Effects 0.000 claims abstract description 26
- 230000001747 exhibiting effect Effects 0.000 claims abstract description 19
- 238000003860 storage Methods 0.000 claims abstract description 13
- XLYOFNOQVPJJNP-UHFFFAOYSA-N water Substances O XLYOFNOQVPJJNP-UHFFFAOYSA-N 0.000 claims description 333
- GWEVSGVZZGPLCZ-UHFFFAOYSA-N Titan oxide Chemical compound O=[Ti]=O GWEVSGVZZGPLCZ-UHFFFAOYSA-N 0.000 claims description 261
- 239000003973 paint Substances 0.000 claims description 207
- 239000000178 monomer Substances 0.000 claims description 175
- VYPSYNLAJGMNEJ-UHFFFAOYSA-N Silicium dioxide Chemical compound O=[Si]=O VYPSYNLAJGMNEJ-UHFFFAOYSA-N 0.000 claims description 144
- -1 trifluoroethyl meth Chemical compound 0.000 claims description 108
- 239000011230 binding agent Substances 0.000 claims description 75
- 239000000049 pigment Substances 0.000 claims description 70
- 230000001699 photocatalysis Effects 0.000 claims description 63
- 239000000839 emulsion Substances 0.000 claims description 60
- 238000000034 method Methods 0.000 claims description 60
- 239000007787 solid Substances 0.000 claims description 60
- PPBRXRYQALVLMV-UHFFFAOYSA-N Styrene Natural products C=CC1=CC=CC=C1 PPBRXRYQALVLMV-UHFFFAOYSA-N 0.000 claims description 50
- 239000002002 slurry Substances 0.000 claims description 46
- SOGAXMICEFXMKE-UHFFFAOYSA-N Butylmethacrylate Chemical compound CCCCOC(=O)C(C)=C SOGAXMICEFXMKE-UHFFFAOYSA-N 0.000 claims description 40
- 239000000463 material Substances 0.000 claims description 40
- 238000011282 treatment Methods 0.000 claims description 39
- 238000002360 preparation method Methods 0.000 claims description 37
- 239000000377 silicon dioxide Substances 0.000 claims description 35
- 150000001412 amines Chemical class 0.000 claims description 33
- 230000008569 process Effects 0.000 claims description 33
- 230000000694 effects Effects 0.000 claims description 32
- SJMYWORNLPSJQO-UHFFFAOYSA-N tert-butyl 2-methylprop-2-enoate Chemical compound CC(=C)C(=O)OC(C)(C)C SJMYWORNLPSJQO-UHFFFAOYSA-N 0.000 claims description 32
- 239000004816 latex Substances 0.000 claims description 31
- 229920000126 latex Polymers 0.000 claims description 31
- 239000004094 surface-active agent Substances 0.000 claims description 31
- CERQOIWHTDAKMF-UHFFFAOYSA-N Methacrylic acid Chemical compound CC(=C)C(O)=O CERQOIWHTDAKMF-UHFFFAOYSA-N 0.000 claims description 29
- 229910000077 silane Inorganic materials 0.000 claims description 29
- 239000002904 solvent Substances 0.000 claims description 29
- 238000012360 testing method Methods 0.000 claims description 29
- BLRPTPMANUNPDV-UHFFFAOYSA-N Silane Chemical compound [SiH4] BLRPTPMANUNPDV-UHFFFAOYSA-N 0.000 claims description 28
- 230000008859 change Effects 0.000 claims description 28
- 239000002270 dispersing agent Substances 0.000 claims description 28
- 230000002829 reductive effect Effects 0.000 claims description 28
- 238000009472 formulation Methods 0.000 claims description 26
- LYCAIKOWRPUZTN-UHFFFAOYSA-N Ethylene glycol Chemical compound OCCO LYCAIKOWRPUZTN-UHFFFAOYSA-N 0.000 claims description 25
- NIXOWILDQLNWCW-UHFFFAOYSA-M Acrylate Chemical compound [O-]C(=O)C=C NIXOWILDQLNWCW-UHFFFAOYSA-M 0.000 claims description 24
- 229920001577 copolymer Polymers 0.000 claims description 22
- 239000004606 Fillers/Extenders Substances 0.000 claims description 21
- 239000006184 cosolvent Substances 0.000 claims description 20
- 150000004756 silanes Chemical class 0.000 claims description 20
- 238000009736 wetting Methods 0.000 claims description 20
- 239000004568 cement Substances 0.000 claims description 19
- 210000003608 fece Anatomy 0.000 claims description 19
- 239000000080 wetting agent Substances 0.000 claims description 19
- 230000008901 benefit Effects 0.000 claims description 18
- 239000003139 biocide Substances 0.000 claims description 18
- 239000003795 chemical substances by application Substances 0.000 claims description 18
- VVQNEPGJFQJSBK-UHFFFAOYSA-N Methyl methacrylate Chemical compound COC(=O)C(C)=C VVQNEPGJFQJSBK-UHFFFAOYSA-N 0.000 claims description 17
- 150000001875 compounds Chemical class 0.000 claims description 17
- 239000004815 dispersion polymer Substances 0.000 claims description 17
- 239000000843 powder Substances 0.000 claims description 17
- 235000012239 silicon dioxide Nutrition 0.000 claims description 17
- SMZOUWXMTYCWNB-UHFFFAOYSA-N 2-(2-methoxy-5-methylphenyl)ethanamine Chemical compound COC1=CC=C(C)C=C1CCN SMZOUWXMTYCWNB-UHFFFAOYSA-N 0.000 claims description 15
- JEIPFZHSYJVQDO-UHFFFAOYSA-N iron(III) oxide Inorganic materials O=[Fe]O[Fe]=O JEIPFZHSYJVQDO-UHFFFAOYSA-N 0.000 claims description 15
- 238000004381 surface treatment Methods 0.000 claims description 15
- 229920002313 fluoropolymer Polymers 0.000 claims description 14
- 239000004811 fluoropolymer Substances 0.000 claims description 14
- 239000010954 inorganic particle Substances 0.000 claims description 14
- 239000011146 organic particle Substances 0.000 claims description 14
- 239000002562 thickening agent Substances 0.000 claims description 14
- 239000004908 Emulsion polymer Substances 0.000 claims description 13
- 150000001252 acrylic acid derivatives Chemical class 0.000 claims description 13
- 239000000654 additive Substances 0.000 claims description 13
- 230000000845 anti-microbial effect Effects 0.000 claims description 13
- WGCNASOHLSPBMP-UHFFFAOYSA-N hydroxyacetaldehyde Natural products OCC=O WGCNASOHLSPBMP-UHFFFAOYSA-N 0.000 claims description 12
- 229910052751 metal Inorganic materials 0.000 claims description 11
- 239000002184 metal Substances 0.000 claims description 11
- 238000002156 mixing Methods 0.000 claims description 11
- 239000003921 oil Substances 0.000 claims description 11
- 239000003755 preservative agent Substances 0.000 claims description 11
- 239000005871 repellent Substances 0.000 claims description 11
- ISXSCDLOGDJUNJ-UHFFFAOYSA-N tert-butyl prop-2-enoate Chemical class CC(C)(C)OC(=O)C=C ISXSCDLOGDJUNJ-UHFFFAOYSA-N 0.000 claims description 11
- YCKRFDGAMUMZLT-UHFFFAOYSA-N Fluorine atom Chemical compound [F] YCKRFDGAMUMZLT-UHFFFAOYSA-N 0.000 claims description 10
- 150000002148 esters Chemical class 0.000 claims description 10
- 239000011737 fluorine Substances 0.000 claims description 10
- 229910052731 fluorine Inorganic materials 0.000 claims description 10
- 239000003960 organic solvent Substances 0.000 claims description 10
- 229920000058 polyacrylate Polymers 0.000 claims description 10
- 150000004760 silicates Chemical class 0.000 claims description 10
- 239000007921 spray Substances 0.000 claims description 10
- BQCIDUSAKPWEOX-UHFFFAOYSA-N 1,1-Difluoroethene Chemical compound FC(F)=C BQCIDUSAKPWEOX-UHFFFAOYSA-N 0.000 claims description 9
- 239000003619 algicide Substances 0.000 claims description 9
- 238000001723 curing Methods 0.000 claims description 9
- 230000002335 preservative effect Effects 0.000 claims description 9
- 230000007480 spreading Effects 0.000 claims description 9
- 238000003892 spreading Methods 0.000 claims description 9
- 239000003381 stabilizer Substances 0.000 claims description 9
- 239000013543 active substance Substances 0.000 claims description 8
- 125000004432 carbon atom Chemical group C* 0.000 claims description 8
- 239000004927 clay Substances 0.000 claims description 8
- 239000004567 concrete Substances 0.000 claims description 8
- 125000000524 functional group Chemical group 0.000 claims description 8
- HCDGVLDPFQMKDK-UHFFFAOYSA-N hexafluoropropylene Chemical group FC(F)=C(F)C(F)(F)F HCDGVLDPFQMKDK-UHFFFAOYSA-N 0.000 claims description 8
- 229910052500 inorganic mineral Inorganic materials 0.000 claims description 8
- 229910044991 metal oxide Inorganic materials 0.000 claims description 8
- 150000004706 metal oxides Chemical class 0.000 claims description 8
- 239000011707 mineral Substances 0.000 claims description 8
- 229920001343 polytetrafluoroethylene Polymers 0.000 claims description 8
- 239000004810 polytetrafluoroethylene Substances 0.000 claims description 8
- PNEYBMLMFCGWSK-UHFFFAOYSA-N Alumina Chemical class [O-2].[O-2].[O-2].[Al+3].[Al+3] PNEYBMLMFCGWSK-UHFFFAOYSA-N 0.000 claims description 7
- 238000010521 absorption reaction Methods 0.000 claims description 7
- 150000001343 alkyl silanes Chemical class 0.000 claims description 7
- 239000000417 fungicide Substances 0.000 claims description 7
- 238000011065 in-situ storage Methods 0.000 claims description 7
- 239000004814 polyurethane Substances 0.000 claims description 7
- 238000010526 radical polymerization reaction Methods 0.000 claims description 7
- KAKZBPTYRLMSJV-UHFFFAOYSA-N Butadiene Chemical compound C=CC=C KAKZBPTYRLMSJV-UHFFFAOYSA-N 0.000 claims description 6
- RTZKZFJDLAIYFH-UHFFFAOYSA-N Diethyl ether Chemical compound CCOCC RTZKZFJDLAIYFH-UHFFFAOYSA-N 0.000 claims description 6
- OAKJQQAXSVQMHS-UHFFFAOYSA-N Hydrazine Chemical group NN OAKJQQAXSVQMHS-UHFFFAOYSA-N 0.000 claims description 6
- RRHGJUQNOFWUDK-UHFFFAOYSA-N Isoprene Chemical compound CC(=C)C=C RRHGJUQNOFWUDK-UHFFFAOYSA-N 0.000 claims description 6
- 239000006096 absorbing agent Substances 0.000 claims description 6
- 125000000217 alkyl group Chemical group 0.000 claims description 6
- 230000003373 anti-fouling effect Effects 0.000 claims description 6
- 239000012612 commercial material Substances 0.000 claims description 6
- 238000004132 cross linking Methods 0.000 claims description 6
- 230000000855 fungicidal effect Effects 0.000 claims description 6
- 239000012770 industrial material Substances 0.000 claims description 6
- 229920000515 polycarbonate Polymers 0.000 claims description 6
- 239000004417 polycarbonate Substances 0.000 claims description 6
- 229920002635 polyurethane Polymers 0.000 claims description 6
- 230000002940 repellent Effects 0.000 claims description 6
- BFKJFAAPBSQJPD-UHFFFAOYSA-N tetrafluoroethene Chemical group FC(F)=C(F)F BFKJFAAPBSQJPD-UHFFFAOYSA-N 0.000 claims description 6
- 229920001169 thermoplastic Polymers 0.000 claims description 6
- RUMACXVDVNRZJZ-UHFFFAOYSA-N 2-methylpropyl 2-methylprop-2-enoate Chemical compound CC(C)COC(=O)C(C)=C RUMACXVDVNRZJZ-UHFFFAOYSA-N 0.000 claims description 5
- 239000004971 Cross linker Substances 0.000 claims description 5
- UQSXHKLRYXJYBZ-UHFFFAOYSA-N Iron oxide Chemical compound [Fe]=O UQSXHKLRYXJYBZ-UHFFFAOYSA-N 0.000 claims description 5
- 239000004599 antimicrobial Substances 0.000 claims description 5
- 239000003086 colorant Substances 0.000 claims description 5
- HGASFNYMVGEKTF-UHFFFAOYSA-N octan-1-ol;hydrate Chemical compound O.CCCCCCCCO HGASFNYMVGEKTF-UHFFFAOYSA-N 0.000 claims description 5
- RVTZCBVAJQQJTK-UHFFFAOYSA-N oxygen(2-);zirconium(4+) Chemical compound [O-2].[O-2].[Zr+4] RVTZCBVAJQQJTK-UHFFFAOYSA-N 0.000 claims description 5
- 229920000728 polyester Polymers 0.000 claims description 5
- 229920000098 polyolefin Polymers 0.000 claims description 5
- 238000005096 rolling process Methods 0.000 claims description 5
- 229910052709 silver Inorganic materials 0.000 claims description 5
- 229920002554 vinyl polymer Polymers 0.000 claims description 5
- PXHVJJICTQNCMI-UHFFFAOYSA-N Nickel Chemical compound [Ni] PXHVJJICTQNCMI-UHFFFAOYSA-N 0.000 claims description 4
- 239000004952 Polyamide Substances 0.000 claims description 4
- BQCADISMDOOEFD-UHFFFAOYSA-N Silver Chemical compound [Ag] BQCADISMDOOEFD-UHFFFAOYSA-N 0.000 claims description 4
- XTXRWKRVRITETP-UHFFFAOYSA-N Vinyl acetate Chemical compound CC(=O)OC=C XTXRWKRVRITETP-UHFFFAOYSA-N 0.000 claims description 4
- 239000002318 adhesion promoter Substances 0.000 claims description 4
- 229910052783 alkali metal Inorganic materials 0.000 claims description 4
- 125000000484 butyl group Chemical group [H]C([*])([H])C([H])([H])C([H])([H])C([H])([H])[H] 0.000 claims description 4
- 239000000084 colloidal system Substances 0.000 claims description 4
- 229910002026 crystalline silica Inorganic materials 0.000 claims description 4
- 125000004122 cyclic group Chemical group 0.000 claims description 4
- 230000001419 dependent effect Effects 0.000 claims description 4
- FJKIXWOMBXYWOQ-UHFFFAOYSA-N ethenoxyethane Chemical compound CCOC=C FJKIXWOMBXYWOQ-UHFFFAOYSA-N 0.000 claims description 4
- 125000001495 ethyl group Chemical group [H]C([H])([H])C([H])([H])* 0.000 claims description 4
- 230000006870 function Effects 0.000 claims description 4
- 239000010440 gypsum Substances 0.000 claims description 4
- 229910052602 gypsum Inorganic materials 0.000 claims description 4
- 239000002071 nanotube Substances 0.000 claims description 4
- 229910052757 nitrogen Inorganic materials 0.000 claims description 4
- 238000005192 partition Methods 0.000 claims description 4
- 229920002647 polyamide Polymers 0.000 claims description 4
- 239000004332 silver Substances 0.000 claims description 4
- 229920001909 styrene-acrylic polymer Polymers 0.000 claims description 4
- 229910052717 sulfur Inorganic materials 0.000 claims description 4
- 239000000725 suspension Substances 0.000 claims description 4
- OGIDPMRJRNCKJF-UHFFFAOYSA-N titanium oxide Inorganic materials [Ti]=O OGIDPMRJRNCKJF-UHFFFAOYSA-N 0.000 claims description 4
- 125000000391 vinyl group Chemical group [H]C([*])=C([H])[H] 0.000 claims description 4
- 229910001928 zirconium oxide Inorganic materials 0.000 claims description 4
- JKNCOURZONDCGV-UHFFFAOYSA-N 2-(dimethylamino)ethyl 2-methylprop-2-enoate Chemical group CN(C)CCOC(=O)C(C)=C JKNCOURZONDCGV-UHFFFAOYSA-N 0.000 claims description 3
- XHZPRMZZQOIPDS-UHFFFAOYSA-N 2-Methyl-2-[(1-oxo-2-propenyl)amino]-1-propanesulfonic acid Chemical compound OS(=O)(=O)CC(C)(C)NC(=O)C=C XHZPRMZZQOIPDS-UHFFFAOYSA-N 0.000 claims description 3
- WDQMWEYDKDCEHT-UHFFFAOYSA-N 2-ethylhexyl 2-methylprop-2-enoate Chemical compound CCCCC(CC)COC(=O)C(C)=C WDQMWEYDKDCEHT-UHFFFAOYSA-N 0.000 claims description 3
- JTHZUSWLNCPZLX-UHFFFAOYSA-N 6-fluoro-3-methyl-2h-indazole Chemical compound FC1=CC=C2C(C)=NNC2=C1 JTHZUSWLNCPZLX-UHFFFAOYSA-N 0.000 claims description 3
- NOWKCMXCCJGMRR-UHFFFAOYSA-N Aziridine Chemical group C1CN1 NOWKCMXCCJGMRR-UHFFFAOYSA-N 0.000 claims description 3
- MYMOFIZGZYHOMD-UHFFFAOYSA-N Dioxygen Chemical compound O=O MYMOFIZGZYHOMD-UHFFFAOYSA-N 0.000 claims description 3
- 239000004593 Epoxy Substances 0.000 claims description 3
- LFQSCWFLJHTTHZ-UHFFFAOYSA-N Ethanol Chemical compound CCO LFQSCWFLJHTTHZ-UHFFFAOYSA-N 0.000 claims description 3
- OAICVXFJPJFONN-UHFFFAOYSA-N Phosphorus Chemical compound [P] OAICVXFJPJFONN-UHFFFAOYSA-N 0.000 claims description 3
- 239000004793 Polystyrene Substances 0.000 claims description 3
- 239000002174 Styrene-butadiene Substances 0.000 claims description 3
- NINIDFKCEFEMDL-UHFFFAOYSA-N Sulfur Chemical compound [S] NINIDFKCEFEMDL-UHFFFAOYSA-N 0.000 claims description 3
- IAXXETNIOYFMLW-COPLHBTASA-N [(1s,3s,4s)-4,7,7-trimethyl-3-bicyclo[2.2.1]heptanyl] 2-methylprop-2-enoate Chemical compound C1C[C@]2(C)[C@@H](OC(=O)C(=C)C)C[C@H]1C2(C)C IAXXETNIOYFMLW-COPLHBTASA-N 0.000 claims description 3
- WDJHALXBUFZDSR-UHFFFAOYSA-M acetoacetate Chemical compound CC(=O)CC([O-])=O WDJHALXBUFZDSR-UHFFFAOYSA-M 0.000 claims description 3
- 150000003926 acrylamides Chemical class 0.000 claims description 3
- 229920006397 acrylic thermoplastic Polymers 0.000 claims description 3
- 150000001338 aliphatic hydrocarbons Chemical class 0.000 claims description 3
- 229920000180 alkyd Polymers 0.000 claims description 3
- 125000000746 allylic group Chemical group 0.000 claims description 3
- 150000001414 amino alcohols Chemical class 0.000 claims description 3
- 150000004945 aromatic hydrocarbons Chemical class 0.000 claims description 3
- 239000000872 buffer Substances 0.000 claims description 3
- MTAZNLWOLGHBHU-UHFFFAOYSA-N butadiene-styrene rubber Chemical compound C=CC=C.C=CC1=CC=CC=C1 MTAZNLWOLGHBHU-UHFFFAOYSA-N 0.000 claims description 3
- 150000001718 carbodiimides Chemical group 0.000 claims description 3
- 239000002131 composite material Substances 0.000 claims description 3
- 230000001877 deodorizing effect Effects 0.000 claims description 3
- 125000004386 diacrylate group Chemical group 0.000 claims description 3
- MTHSVFCYNBDYFN-UHFFFAOYSA-N diethylene glycol Chemical compound OCCOCCO MTHSVFCYNBDYFN-UHFFFAOYSA-N 0.000 claims description 3
- 125000003700 epoxy group Chemical group 0.000 claims description 3
- HQQADJVZYDDRJT-UHFFFAOYSA-N ethene;prop-1-ene Chemical group C=C.CC=C HQQADJVZYDDRJT-UHFFFAOYSA-N 0.000 claims description 3
- SUPCQIBBMFXVTL-UHFFFAOYSA-N ethyl 2-methylprop-2-enoate Chemical compound CCOC(=O)C(C)=C SUPCQIBBMFXVTL-UHFFFAOYSA-N 0.000 claims description 3
- 229940119545 isobornyl methacrylate Drugs 0.000 claims description 3
- 150000002576 ketones Chemical class 0.000 claims description 3
- PBOSTUDLECTMNL-UHFFFAOYSA-N lauryl acrylate Chemical compound CCCCCCCCCCCCOC(=O)C=C PBOSTUDLECTMNL-UHFFFAOYSA-N 0.000 claims description 3
- 150000002734 metacrylic acid derivatives Chemical class 0.000 claims description 3
- 239000004570 mortar (masonry) Substances 0.000 claims description 3
- 229920005615 natural polymer Polymers 0.000 claims description 3
- 150000002825 nitriles Chemical class 0.000 claims description 3
- 125000004433 nitrogen atom Chemical group N* 0.000 claims description 3
- 239000003605 opacifier Substances 0.000 claims description 3
- 229910052760 oxygen Inorganic materials 0.000 claims description 3
- 239000001301 oxygen Substances 0.000 claims description 3
- 125000005010 perfluoroalkyl group Chemical group 0.000 claims description 3
- 239000002957 persistent organic pollutant Substances 0.000 claims description 3
- 229910052698 phosphorus Inorganic materials 0.000 claims description 3
- 239000011574 phosphorus Substances 0.000 claims description 3
- 239000011505 plaster Substances 0.000 claims description 3
- 229920000233 poly(alkylene oxides) Chemical group 0.000 claims description 3
- 229920001084 poly(chloroprene) Polymers 0.000 claims description 3
- 229920003229 poly(methyl methacrylate) Polymers 0.000 claims description 3
- 229920001515 polyalkylene glycol Polymers 0.000 claims description 3
- 229920001610 polycaprolactone Polymers 0.000 claims description 3
- 239000004632 polycaprolactone Substances 0.000 claims description 3
- 229920000647 polyepoxide Polymers 0.000 claims description 3
- 230000000379 polymerizing effect Effects 0.000 claims description 3
- 229920002223 polystyrene Polymers 0.000 claims description 3
- 125000001436 propyl group Chemical group [H]C([*])([H])C([H])([H])C([H])([H])[H] 0.000 claims description 3
- 230000009467 reduction Effects 0.000 claims description 3
- 150000003839 salts Chemical class 0.000 claims description 3
- 238000004528 spin coating Methods 0.000 claims description 3
- 239000011115 styrene butadiene Substances 0.000 claims description 3
- 229920003048 styrene butadiene rubber Polymers 0.000 claims description 3
- BDHFUVZGWQCTTF-UHFFFAOYSA-N sulfonic acid Chemical class OS(=O)=O BDHFUVZGWQCTTF-UHFFFAOYSA-N 0.000 claims description 3
- 239000011593 sulfur Substances 0.000 claims description 3
- 229920001897 terpolymer Polymers 0.000 claims description 3
- 125000000383 tetramethylene group Chemical group [H]C([H])([*:1])C([H])([H])C([H])([H])C([H])([H])[*:2] 0.000 claims description 3
- 229920001187 thermosetting polymer Polymers 0.000 claims description 3
- 239000004416 thermosoftening plastic Substances 0.000 claims description 3
- 239000002023 wood Substances 0.000 claims description 3
- KHXKESCWFMPTFT-UHFFFAOYSA-N 1,1,1,2,2,3,3-heptafluoro-3-(1,2,2-trifluoroethenoxy)propane Chemical compound FC(F)=C(F)OC(F)(F)C(F)(F)C(F)(F)F KHXKESCWFMPTFT-UHFFFAOYSA-N 0.000 claims description 2
- BLTXWCKMNMYXEA-UHFFFAOYSA-N 1,1,2-trifluoro-2-(trifluoromethoxy)ethene Chemical compound FC(F)=C(F)OC(F)(F)F BLTXWCKMNMYXEA-UHFFFAOYSA-N 0.000 claims description 2
- 229910000640 Fe alloy Inorganic materials 0.000 claims description 2
- 229920001774 Perfluoroether Polymers 0.000 claims description 2
- 229920002732 Polyanhydride Polymers 0.000 claims description 2
- 241000276425 Xiphophorus maculatus Species 0.000 claims description 2
- 239000000956 alloy Substances 0.000 claims description 2
- 239000011449 brick Substances 0.000 claims description 2
- 230000000593 degrading effect Effects 0.000 claims description 2
- 239000003085 diluting agent Substances 0.000 claims description 2
- 238000007598 dipping method Methods 0.000 claims description 2
- 239000002657 fibrous material Substances 0.000 claims description 2
- 239000003063 flame retardant Substances 0.000 claims description 2
- 230000009969 flowable effect Effects 0.000 claims description 2
- 239000011440 grout Substances 0.000 claims description 2
- 238000011416 infrared curing Methods 0.000 claims description 2
- 239000012784 inorganic fiber Substances 0.000 claims description 2
- 239000002121 nanofiber Substances 0.000 claims description 2
- 239000002070 nanowire Substances 0.000 claims description 2
- 229910052759 nickel Inorganic materials 0.000 claims description 2
- 229910000480 nickel oxide Inorganic materials 0.000 claims description 2
- 239000012454 non-polar solvent Substances 0.000 claims description 2
- GNRSAWUEBMWBQH-UHFFFAOYSA-N oxonickel Chemical compound [Ni]=O GNRSAWUEBMWBQH-UHFFFAOYSA-N 0.000 claims description 2
- 239000004014 plasticizer Substances 0.000 claims description 2
- 239000002798 polar solvent Substances 0.000 claims description 2
- 239000002952 polymeric resin Substances 0.000 claims description 2
- 125000001453 quaternary ammonium group Chemical group 0.000 claims description 2
- 229920005573 silicon-containing polymer Polymers 0.000 claims description 2
- 239000012798 spherical particle Substances 0.000 claims description 2
- 238000013112 stability test Methods 0.000 claims description 2
- 239000004575 stone Substances 0.000 claims description 2
- 125000003011 styrenyl group Chemical group [H]\C(*)=C(/[H])C1=C([H])C([H])=C([H])C([H])=C1[H] 0.000 claims description 2
- 229920003002 synthetic resin Polymers 0.000 claims description 2
- 238000001029 thermal curing Methods 0.000 claims description 2
- 239000004634 thermosetting polymer Substances 0.000 claims description 2
- 125000004356 hydroxy functional group Chemical group O* 0.000 claims 1
- 125000003277 amino group Chemical group 0.000 abstract description 11
- 239000011859 microparticle Substances 0.000 abstract description 5
- 230000004044 response Effects 0.000 abstract description 4
- 229920005989 resin Polymers 0.000 description 179
- 239000011347 resin Substances 0.000 description 179
- ZMANZCXQSJIPKH-UHFFFAOYSA-N Triethylamine Chemical compound CCN(CC)CC ZMANZCXQSJIPKH-UHFFFAOYSA-N 0.000 description 69
- 239000010408 film Substances 0.000 description 57
- 238000002474 experimental method Methods 0.000 description 38
- KFZMGEQAYNKOFK-UHFFFAOYSA-N Isopropanol Chemical compound CC(C)O KFZMGEQAYNKOFK-UHFFFAOYSA-N 0.000 description 33
- 239000004408 titanium dioxide Substances 0.000 description 33
- QTBSBXVTEAMEQO-UHFFFAOYSA-N Acetic acid Chemical compound CC(O)=O QTBSBXVTEAMEQO-UHFFFAOYSA-N 0.000 description 32
- 238000006243 chemical reaction Methods 0.000 description 32
- 229910002012 Aerosil® Inorganic materials 0.000 description 26
- POAOYUHQDCAZBD-UHFFFAOYSA-N 2-butoxyethanol Chemical compound CCCCOCCO POAOYUHQDCAZBD-UHFFFAOYSA-N 0.000 description 23
- 239000011941 photocatalyst Substances 0.000 description 23
- 229940086542 triethylamine Drugs 0.000 description 23
- 125000001153 fluoro group Chemical group F* 0.000 description 22
- ZNQVEEAIQZEUHB-UHFFFAOYSA-N 2-ethoxyethanol Chemical compound CCOCCO ZNQVEEAIQZEUHB-UHFFFAOYSA-N 0.000 description 20
- 239000000126 substance Substances 0.000 description 20
- XQAXGZLFSSPBMK-UHFFFAOYSA-M [7-(dimethylamino)phenothiazin-3-ylidene]-dimethylazanium;chloride;trihydrate Chemical compound O.O.O.[Cl-].C1=CC(=[N+](C)C)C=C2SC3=CC(N(C)C)=CC=C3N=C21 XQAXGZLFSSPBMK-UHFFFAOYSA-M 0.000 description 19
- NLYAJNPCOHFWQQ-UHFFFAOYSA-N kaolin Chemical compound O.O.O=[Al]O[Si](=O)O[Si](=O)O[Al]=O NLYAJNPCOHFWQQ-UHFFFAOYSA-N 0.000 description 19
- 229960000907 methylthioninium chloride Drugs 0.000 description 19
- 125000000129 anionic group Chemical group 0.000 description 18
- 230000029087 digestion Effects 0.000 description 18
- 230000005661 hydrophobic surface Effects 0.000 description 18
- 239000000243 solution Substances 0.000 description 17
- 229920002125 Sokalan® Polymers 0.000 description 15
- NKSJNEHGWDZZQF-UHFFFAOYSA-N ethenyl(trimethoxy)silane Chemical compound CO[Si](OC)(OC)C=C NKSJNEHGWDZZQF-UHFFFAOYSA-N 0.000 description 15
- 239000007788 liquid Substances 0.000 description 15
- VTYYLEPIZMXCLO-UHFFFAOYSA-L Calcium carbonate Chemical compound [Ca+2].[O-]C([O-])=O VTYYLEPIZMXCLO-UHFFFAOYSA-L 0.000 description 14
- 239000000945 filler Substances 0.000 description 14
- 239000010410 layer Substances 0.000 description 14
- 229920005604 random copolymer Polymers 0.000 description 14
- GJBRNHKUVLOCEB-UHFFFAOYSA-N tert-butyl benzenecarboperoxoate Chemical compound CC(C)(C)OOC(=O)C1=CC=CC=C1 GJBRNHKUVLOCEB-UHFFFAOYSA-N 0.000 description 14
- 230000015556 catabolic process Effects 0.000 description 13
- 238000006731 degradation reaction Methods 0.000 description 13
- 238000010438 heat treatment Methods 0.000 description 13
- 238000005259 measurement Methods 0.000 description 13
- QGZKDVFQNNGYKY-UHFFFAOYSA-N Ammonia Chemical compound N QGZKDVFQNNGYKY-UHFFFAOYSA-N 0.000 description 12
- 241000195493 Cryptophyta Species 0.000 description 12
- XLOMVQKBTHCTTD-UHFFFAOYSA-N Zinc monoxide Chemical compound [Zn]=O XLOMVQKBTHCTTD-UHFFFAOYSA-N 0.000 description 12
- DCAYPVUWAIABOU-UHFFFAOYSA-N hexadecane Chemical compound CCCCCCCCCCCCCCCC DCAYPVUWAIABOU-UHFFFAOYSA-N 0.000 description 12
- 230000001965 increasing effect Effects 0.000 description 12
- 239000001993 wax Substances 0.000 description 12
- XDLMVUHYZWKMMD-UHFFFAOYSA-N 3-trimethoxysilylpropyl 2-methylprop-2-enoate Chemical compound CO[Si](OC)(OC)CCCOC(=O)C(C)=C XDLMVUHYZWKMMD-UHFFFAOYSA-N 0.000 description 11
- 229960000583 acetic acid Drugs 0.000 description 11
- 238000004519 manufacturing process Methods 0.000 description 11
- 229920006313 waterborne resin Polymers 0.000 description 11
- 239000013035 waterborne resin Substances 0.000 description 11
- 230000012010 growth Effects 0.000 description 10
- 239000011164 primary particle Substances 0.000 description 10
- 229920002050 silicone resin Polymers 0.000 description 10
- 230000015572 biosynthetic process Effects 0.000 description 9
- CQEYYJKEWSMYFG-UHFFFAOYSA-N butyl acrylate Chemical compound CCCCOC(=O)C=C CQEYYJKEWSMYFG-UHFFFAOYSA-N 0.000 description 9
- 125000002091 cationic group Chemical group 0.000 description 9
- 239000013530 defoamer Substances 0.000 description 9
- 238000011156 evaluation Methods 0.000 description 9
- 230000005660 hydrophilic surface Effects 0.000 description 9
- GOXQRTZXKQZDDN-UHFFFAOYSA-N 2-Ethylhexyl acrylate Chemical compound CCCCC(CC)COC(=O)C=C GOXQRTZXKQZDDN-UHFFFAOYSA-N 0.000 description 8
- XEEYBQQBJWHFJM-UHFFFAOYSA-N Iron Chemical compound [Fe] XEEYBQQBJWHFJM-UHFFFAOYSA-N 0.000 description 8
- 240000002853 Nelumbo nucifera Species 0.000 description 8
- 239000000499 gel Substances 0.000 description 8
- 239000011521 glass Substances 0.000 description 8
- 230000000996 additive effect Effects 0.000 description 7
- 230000001098 anti-algal effect Effects 0.000 description 7
- 229910000019 calcium carbonate Inorganic materials 0.000 description 7
- 238000001035 drying Methods 0.000 description 7
- 238000007306 functionalization reaction Methods 0.000 description 7
- 230000007062 hydrolysis Effects 0.000 description 7
- 238000006460 hydrolysis reaction Methods 0.000 description 7
- 244000005700 microbiome Species 0.000 description 7
- 235000010755 mineral Nutrition 0.000 description 7
- 239000000523 sample Substances 0.000 description 7
- 238000003980 solgel method Methods 0.000 description 7
- 235000014692 zinc oxide Nutrition 0.000 description 7
- KBPLFHHGFOOTCA-UHFFFAOYSA-N 1-Octanol Chemical compound CCCCCCCCO KBPLFHHGFOOTCA-UHFFFAOYSA-N 0.000 description 6
- OKTJSMMVPCPJKN-UHFFFAOYSA-N Carbon Chemical compound [C] OKTJSMMVPCPJKN-UHFFFAOYSA-N 0.000 description 6
- 235000006508 Nelumbo nucifera Nutrition 0.000 description 6
- 235000006510 Nelumbo pentapetala Nutrition 0.000 description 6
- DAKWPKUUDNSNPN-UHFFFAOYSA-N Trimethylolpropane triacrylate Chemical compound C=CC(=O)OCC(CC)(COC(=O)C=C)COC(=O)C=C DAKWPKUUDNSNPN-UHFFFAOYSA-N 0.000 description 6
- 229910021529 ammonia Inorganic materials 0.000 description 6
- 230000007423 decrease Effects 0.000 description 6
- 229910021485 fumed silica Inorganic materials 0.000 description 6
- 239000004615 ingredient Substances 0.000 description 6
- 150000001282 organosilanes Chemical class 0.000 description 6
- 239000010453 quartz Substances 0.000 description 6
- 239000011787 zinc oxide Substances 0.000 description 6
- XOLBLPGZBRYERU-UHFFFAOYSA-N SnO2 Inorganic materials O=[Sn]=O XOLBLPGZBRYERU-UHFFFAOYSA-N 0.000 description 5
- 150000001298 alcohols Chemical class 0.000 description 5
- 238000004630 atomic force microscopy Methods 0.000 description 5
- 238000010276 construction Methods 0.000 description 5
- 239000003995 emulsifying agent Substances 0.000 description 5
- 239000000835 fiber Substances 0.000 description 5
- 238000007667 floating Methods 0.000 description 5
- 229920001600 hydrophobic polymer Polymers 0.000 description 5
- 239000003999 initiator Substances 0.000 description 5
- 239000003607 modifier Substances 0.000 description 5
- 239000012071 phase Substances 0.000 description 5
- 239000006254 rheological additive Substances 0.000 description 5
- CSCPPACGZOOCGX-UHFFFAOYSA-N Acetone Chemical compound CC(C)=O CSCPPACGZOOCGX-UHFFFAOYSA-N 0.000 description 4
- 241000894006 Bacteria Species 0.000 description 4
- 241000254173 Coleoptera Species 0.000 description 4
- CTQNGGLPUBDAKN-UHFFFAOYSA-N O-Xylene Chemical group CC1=CC=CC=C1C CTQNGGLPUBDAKN-UHFFFAOYSA-N 0.000 description 4
- 239000004698 Polyethylene Substances 0.000 description 4
- 239000004743 Polypropylene Substances 0.000 description 4
- 239000012615 aggregate Substances 0.000 description 4
- 230000000844 anti-bacterial effect Effects 0.000 description 4
- 229940121375 antifungal agent Drugs 0.000 description 4
- 230000003115 biocidal effect Effects 0.000 description 4
- 239000003054 catalyst Substances 0.000 description 4
- 229920002678 cellulose Polymers 0.000 description 4
- 239000001913 cellulose Substances 0.000 description 4
- 238000007796 conventional method Methods 0.000 description 4
- 238000005260 corrosion Methods 0.000 description 4
- 230000007797 corrosion Effects 0.000 description 4
- 239000013078 crystal Substances 0.000 description 4
- 238000004042 decolorization Methods 0.000 description 4
- LSXWFXONGKSEMY-UHFFFAOYSA-N di-tert-butyl peroxide Chemical compound CC(C)(C)OOC(C)(C)C LSXWFXONGKSEMY-UHFFFAOYSA-N 0.000 description 4
- 239000004205 dimethyl polysiloxane Substances 0.000 description 4
- KPUWHANPEXNPJT-UHFFFAOYSA-N disiloxane Chemical class [SiH3]O[SiH3] KPUWHANPEXNPJT-UHFFFAOYSA-N 0.000 description 4
- 238000009826 distribution Methods 0.000 description 4
- 238000000227 grinding Methods 0.000 description 4
- 125000002887 hydroxy group Chemical group [H]O* 0.000 description 4
- 238000010348 incorporation Methods 0.000 description 4
- 229910052742 iron Inorganic materials 0.000 description 4
- XMGQYMWWDOXHJM-UHFFFAOYSA-N limonene Chemical compound CC(=C)C1CCC(C)=CC1 XMGQYMWWDOXHJM-UHFFFAOYSA-N 0.000 description 4
- 229920003023 plastic Polymers 0.000 description 4
- 239000004033 plastic Substances 0.000 description 4
- 229920000435 poly(dimethylsiloxane) Polymers 0.000 description 4
- 229920000573 polyethylene Polymers 0.000 description 4
- 238000006116 polymerization reaction Methods 0.000 description 4
- 229920001155 polypropylene Polymers 0.000 description 4
- 230000003746 surface roughness Effects 0.000 description 4
- 239000010936 titanium Substances 0.000 description 4
- 238000012876 topography Methods 0.000 description 4
- 229940096522 trimethylolpropane triacrylate Drugs 0.000 description 4
- DAFHKNAQFPVRKR-UHFFFAOYSA-N (3-hydroxy-2,2,4-trimethylpentyl) 2-methylpropanoate Chemical compound CC(C)C(O)C(C)(C)COC(=O)C(C)C DAFHKNAQFPVRKR-UHFFFAOYSA-N 0.000 description 3
- JNDVNJWCRZQGFQ-UHFFFAOYSA-N 2-methyl-N,N-bis(methylamino)hex-2-enamide Chemical compound CCCC=C(C)C(=O)N(NC)NC JNDVNJWCRZQGFQ-UHFFFAOYSA-N 0.000 description 3
- 241000195940 Bryophyta Species 0.000 description 3
- WSFSSNUMVMOOMR-UHFFFAOYSA-N Formaldehyde Chemical compound O=C WSFSSNUMVMOOMR-UHFFFAOYSA-N 0.000 description 3
- 241000233866 Fungi Species 0.000 description 3
- 206010061217 Infestation Diseases 0.000 description 3
- DNIAPMSPPWPWGF-UHFFFAOYSA-N Propylene glycol Chemical compound CC(O)CO DNIAPMSPPWPWGF-UHFFFAOYSA-N 0.000 description 3
- 229920002472 Starch Polymers 0.000 description 3
- 238000005299 abrasion Methods 0.000 description 3
- 229920005822 acrylic binder Polymers 0.000 description 3
- 230000002776 aggregation Effects 0.000 description 3
- 230000005791 algae growth Effects 0.000 description 3
- 239000007864 aqueous solution Substances 0.000 description 3
- 238000010560 atom transfer radical polymerization reaction Methods 0.000 description 3
- 239000011324 bead Substances 0.000 description 3
- 229920001400 block copolymer Polymers 0.000 description 3
- 239000001045 blue dye Substances 0.000 description 3
- 125000002843 carboxylic acid group Chemical group 0.000 description 3
- 238000000113 differential scanning calorimetry Methods 0.000 description 3
- 238000010790 dilution Methods 0.000 description 3
- 239000012895 dilution Substances 0.000 description 3
- 238000003618 dip coating Methods 0.000 description 3
- 238000002845 discoloration Methods 0.000 description 3
- GVGUFUZHNYFZLC-UHFFFAOYSA-N dodecyl benzenesulfonate;sodium Chemical compound [Na].CCCCCCCCCCCCOS(=O)(=O)C1=CC=CC=C1 GVGUFUZHNYFZLC-UHFFFAOYSA-N 0.000 description 3
- 238000007720 emulsion polymerization reaction Methods 0.000 description 3
- 238000001704 evaporation Methods 0.000 description 3
- 230000008020 evaporation Effects 0.000 description 3
- 229910052737 gold Inorganic materials 0.000 description 3
- 239000010931 gold Substances 0.000 description 3
- 229920001477 hydrophilic polymer Polymers 0.000 description 3
- 125000001165 hydrophobic group Chemical group 0.000 description 3
- 230000007774 longterm Effects 0.000 description 3
- 150000002739 metals Chemical class 0.000 description 3
- 230000000813 microbial effect Effects 0.000 description 3
- 230000003472 neutralizing effect Effects 0.000 description 3
- 230000001590 oxidative effect Effects 0.000 description 3
- 230000036961 partial effect Effects 0.000 description 3
- BASFCYQUMIYNBI-UHFFFAOYSA-N platinum Substances [Pt] BASFCYQUMIYNBI-UHFFFAOYSA-N 0.000 description 3
- 238000000926 separation method Methods 0.000 description 3
- 150000003377 silicon compounds Chemical class 0.000 description 3
- 229940080264 sodium dodecylbenzenesulfonate Drugs 0.000 description 3
- 238000001228 spectrum Methods 0.000 description 3
- 239000008107 starch Substances 0.000 description 3
- 235000019698 starch Nutrition 0.000 description 3
- 239000002344 surface layer Substances 0.000 description 3
- 238000006557 surface reaction Methods 0.000 description 3
- 239000008399 tap water Substances 0.000 description 3
- 235000020679 tap water Nutrition 0.000 description 3
- 125000000999 tert-butyl group Chemical group [H]C([H])([H])C(*)(C([H])([H])[H])C([H])([H])[H] 0.000 description 3
- 229910052719 titanium Inorganic materials 0.000 description 3
- 229910052725 zinc Inorganic materials 0.000 description 3
- 239000011701 zinc Substances 0.000 description 3
- MYRTYDVEIRVNKP-UHFFFAOYSA-N 1,2-Divinylbenzene Chemical compound C=CC1=CC=CC=C1C=C MYRTYDVEIRVNKP-UHFFFAOYSA-N 0.000 description 2
- CUVLMZNMSPJDON-UHFFFAOYSA-N 1-(1-butoxypropan-2-yloxy)propan-2-ol Chemical compound CCCCOCC(C)OCC(C)O CUVLMZNMSPJDON-UHFFFAOYSA-N 0.000 description 2
- LIKMAJRDDDTEIG-UHFFFAOYSA-N 1-hexene Chemical compound CCCCC=C LIKMAJRDDDTEIG-UHFFFAOYSA-N 0.000 description 2
- DOWKETVLGQEPMI-UHFFFAOYSA-N 5,10-Nonacosandiol Natural products CCCCCCCCCCCCCCCCCCCC(O)CCCCC(O)CCCC DOWKETVLGQEPMI-UHFFFAOYSA-N 0.000 description 2
- 0 CCC1(C)*CCC1 Chemical compound CCC1(C)*CCC1 0.000 description 2
- DGAQECJNVWCQMB-PUAWFVPOSA-M Ilexoside XXIX Chemical compound C[C@@H]1CC[C@@]2(CC[C@@]3(C(=CC[C@H]4[C@]3(CC[C@@H]5[C@@]4(CC[C@@H](C5(C)C)OS(=O)(=O)[O-])C)C)[C@@H]2[C@]1(C)O)C)C(=O)O[C@H]6[C@@H]([C@H]([C@@H]([C@H](O6)CO)O)O)O.[Na+] DGAQECJNVWCQMB-PUAWFVPOSA-M 0.000 description 2
- VQTUBCCKSQIDNK-UHFFFAOYSA-N Isobutene Chemical compound CC(C)=C VQTUBCCKSQIDNK-UHFFFAOYSA-N 0.000 description 2
- CERQOIWHTDAKMF-UHFFFAOYSA-M Methacrylate Chemical compound CC(=C)C([O-])=O CERQOIWHTDAKMF-UHFFFAOYSA-M 0.000 description 2
- YNAVUWVOSKDBBP-UHFFFAOYSA-N Morpholine Chemical compound C1COCCN1 YNAVUWVOSKDBBP-UHFFFAOYSA-N 0.000 description 2
- 229910019142 PO4 Inorganic materials 0.000 description 2
- 239000002202 Polyethylene glycol Substances 0.000 description 2
- 229920002873 Polyethylenimine Polymers 0.000 description 2
- OFOBLEOULBTSOW-UHFFFAOYSA-N Propanedioic acid Natural products OC(=O)CC(O)=O OFOBLEOULBTSOW-UHFFFAOYSA-N 0.000 description 2
- 239000004809 Teflon Substances 0.000 description 2
- 229920006362 Teflon® Polymers 0.000 description 2
- WYURNTSHIVDZCO-UHFFFAOYSA-N Tetrahydrofuran Chemical compound C1CCOC1 WYURNTSHIVDZCO-UHFFFAOYSA-N 0.000 description 2
- ATJFFYVFTNAWJD-UHFFFAOYSA-N Tin Chemical compound [Sn] ATJFFYVFTNAWJD-UHFFFAOYSA-N 0.000 description 2
- RTAQQCXQSZGOHL-UHFFFAOYSA-N Titanium Chemical compound [Ti] RTAQQCXQSZGOHL-UHFFFAOYSA-N 0.000 description 2
- 229920004482 WACKER® Polymers 0.000 description 2
- HCHKCACWOHOZIP-UHFFFAOYSA-N Zinc Chemical compound [Zn] HCHKCACWOHOZIP-UHFFFAOYSA-N 0.000 description 2
- 238000005054 agglomeration Methods 0.000 description 2
- 230000032683 aging Effects 0.000 description 2
- 238000007605 air drying Methods 0.000 description 2
- 235000010443 alginic acid Nutrition 0.000 description 2
- 229920000615 alginic acid Polymers 0.000 description 2
- 239000000783 alginic acid Substances 0.000 description 2
- 229960001126 alginic acid Drugs 0.000 description 2
- 125000003545 alkoxy group Chemical group 0.000 description 2
- CBTVGIZVANVGBH-UHFFFAOYSA-N aminomethyl propanol Chemical compound CC(C)(N)CO CBTVGIZVANVGBH-UHFFFAOYSA-N 0.000 description 2
- 150000003863 ammonium salts Chemical class 0.000 description 2
- 230000000843 anti-fungal effect Effects 0.000 description 2
- 239000003429 antifungal agent Substances 0.000 description 2
- TZCXTZWJZNENPQ-UHFFFAOYSA-L barium sulfate Chemical compound [Ba+2].[O-]S([O-])(=O)=O TZCXTZWJZNENPQ-UHFFFAOYSA-L 0.000 description 2
- 239000002585 base Substances 0.000 description 2
- 238000009835 boiling Methods 0.000 description 2
- 150000001735 carboxylic acids Chemical class 0.000 description 2
- 230000003197 catalytic effect Effects 0.000 description 2
- 238000012512 characterization method Methods 0.000 description 2
- 239000003610 charcoal Substances 0.000 description 2
- IJOOHPMOJXWVHK-UHFFFAOYSA-N chlorotrimethylsilane Chemical compound C[Si](C)(C)Cl IJOOHPMOJXWVHK-UHFFFAOYSA-N 0.000 description 2
- 238000005345 coagulation Methods 0.000 description 2
- 230000015271 coagulation Effects 0.000 description 2
- 239000011247 coating layer Substances 0.000 description 2
- 239000008119 colloidal silica Substances 0.000 description 2
- 239000013068 control sample Substances 0.000 description 2
- 239000011258 core-shell material Substances 0.000 description 2
- 238000005336 cracking Methods 0.000 description 2
- 229910052906 cristobalite Inorganic materials 0.000 description 2
- ZSWFCLXCOIISFI-UHFFFAOYSA-N cyclopentadiene Chemical compound C1C=CC=C1 ZSWFCLXCOIISFI-UHFFFAOYSA-N 0.000 description 2
- LPIQUOYDBNQMRZ-UHFFFAOYSA-N cyclopentene Chemical compound C1CC=CC1 LPIQUOYDBNQMRZ-UHFFFAOYSA-N 0.000 description 2
- 238000000354 decomposition reaction Methods 0.000 description 2
- 238000000151 deposition Methods 0.000 description 2
- 230000008021 deposition Effects 0.000 description 2
- 238000011161 development Methods 0.000 description 2
- 230000018109 developmental process Effects 0.000 description 2
- IEJIGPNLZYLLBP-UHFFFAOYSA-N dimethyl carbonate Chemical compound COC(=O)OC IEJIGPNLZYLLBP-UHFFFAOYSA-N 0.000 description 2
- 238000012674 dispersion polymerization Methods 0.000 description 2
- 239000012153 distilled water Substances 0.000 description 2
- 239000000428 dust Substances 0.000 description 2
- 229940087810 eco-soft Drugs 0.000 description 2
- 230000002708 enhancing effect Effects 0.000 description 2
- STVZJERGLQHEKB-UHFFFAOYSA-N ethylene glycol dimethacrylate Chemical compound CC(=C)C(=O)OCCOC(=O)C(C)=C STVZJERGLQHEKB-UHFFFAOYSA-N 0.000 description 2
- 229920001002 functional polymer Polymers 0.000 description 2
- 150000002334 glycols Chemical class 0.000 description 2
- PCHJSUWPFVWCPO-UHFFFAOYSA-N gold Chemical compound [Au] PCHJSUWPFVWCPO-UHFFFAOYSA-N 0.000 description 2
- FFUAGWLWBBFQJT-UHFFFAOYSA-N hexamethyldisilazane Chemical compound C[Si](C)(C)N[Si](C)(C)C FFUAGWLWBBFQJT-UHFFFAOYSA-N 0.000 description 2
- 125000002768 hydroxyalkyl group Chemical group 0.000 description 2
- 239000002054 inoculum Substances 0.000 description 2
- YOBAEOGBNPPUQV-UHFFFAOYSA-N iron;trihydrate Chemical compound O.O.O.[Fe].[Fe] YOBAEOGBNPPUQV-UHFFFAOYSA-N 0.000 description 2
- 229940094522 laponite Drugs 0.000 description 2
- 238000000707 layer-by-layer assembly Methods 0.000 description 2
- 230000000670 limiting effect Effects 0.000 description 2
- XCOBTUNSZUJCDH-UHFFFAOYSA-B lithium magnesium sodium silicate Chemical compound [Li+].[Li+].[OH-].[OH-].[OH-].[OH-].[OH-].[OH-].[OH-].[OH-].[OH-].[OH-].[OH-].[OH-].[Na+].[Na+].[Mg+2].[Mg+2].[Mg+2].[Mg+2].[Mg+2].[Mg+2].[Mg+2].[Mg+2].[Mg+2].[Mg+2].[Mg+2].[Mg+2].[Mg+2].[Mg+2].[Mg+2].[Mg+2].O1[Si](O2)([O-])O[Si]3([O-])O[Si]1([O-])O[Si]2([O-])O3.O1[Si](O2)([O-])O[Si]3([O-])O[Si]1([O-])O[Si]2([O-])O3.O1[Si](O2)([O-])O[Si]3([O-])O[Si]1([O-])O[Si]2([O-])O3.O1[Si](O2)([O-])O[Si]3([O-])O[Si]1([O-])O[Si]2([O-])O3.O1[Si](O2)([O-])O[Si]3([O-])O[Si]1([O-])O[Si]2([O-])O3.O1[Si](O2)([O-])O[Si]3([O-])O[Si]1([O-])O[Si]2([O-])O3 XCOBTUNSZUJCDH-UHFFFAOYSA-B 0.000 description 2
- 238000011068 loading method Methods 0.000 description 2
- 230000014759 maintenance of location Effects 0.000 description 2
- VZCYOOQTPOCHFL-UPHRSURJSA-N maleic acid Chemical compound OC(=O)\C=C/C(O)=O VZCYOOQTPOCHFL-UPHRSURJSA-N 0.000 description 2
- 239000011976 maleic acid Substances 0.000 description 2
- 230000007246 mechanism Effects 0.000 description 2
- 125000002496 methyl group Chemical group [H]C([H])([H])* 0.000 description 2
- 238000000465 moulding Methods 0.000 description 2
- AFFLGGQVNFXPEV-UHFFFAOYSA-N n-decene Natural products CCCCCCCCC=C AFFLGGQVNFXPEV-UHFFFAOYSA-N 0.000 description 2
- 239000002086 nanomaterial Substances 0.000 description 2
- 238000006386 neutralization reaction Methods 0.000 description 2
- 238000012705 nitroxide-mediated radical polymerization Methods 0.000 description 2
- CPGCVOVWHCWVTP-UHFFFAOYSA-N nonacosan-10-ol Chemical compound CCCCCCCCCCCCCCCCCCCC(O)CCCCCCCCC CPGCVOVWHCWVTP-UHFFFAOYSA-N 0.000 description 2
- 229940078552 o-xylene Drugs 0.000 description 2
- UTOPWMOLSKOLTQ-UHFFFAOYSA-N octacosanoic acid Chemical compound CCCCCCCCCCCCCCCCCCCCCCCCCCCC(O)=O UTOPWMOLSKOLTQ-UHFFFAOYSA-N 0.000 description 2
- 229920000620 organic polymer Polymers 0.000 description 2
- 125000005375 organosiloxane group Chemical group 0.000 description 2
- 238000010979 pH adjustment Methods 0.000 description 2
- 235000021317 phosphate Nutrition 0.000 description 2
- 238000007146 photocatalysis Methods 0.000 description 2
- 229910052697 platinum Inorganic materials 0.000 description 2
- 229920002401 polyacrylamide Polymers 0.000 description 2
- 229920006294 polydialkylsiloxane Polymers 0.000 description 2
- 229920000120 polyethyl acrylate Polymers 0.000 description 2
- 229920001223 polyethylene glycol Polymers 0.000 description 2
- 229920006324 polyoxymethylene Polymers 0.000 description 2
- 229920001451 polypropylene glycol Polymers 0.000 description 2
- 125000002924 primary amino group Chemical group [H]N([H])* 0.000 description 2
- HJWLCRVIBGQPNF-UHFFFAOYSA-N prop-2-enylbenzene Chemical compound C=CCC1=CC=CC=C1 HJWLCRVIBGQPNF-UHFFFAOYSA-N 0.000 description 2
- 230000004224 protection Effects 0.000 description 2
- 230000005855 radiation Effects 0.000 description 2
- 238000011160 research Methods 0.000 description 2
- 238000000518 rheometry Methods 0.000 description 2
- 229910052703 rhodium Inorganic materials 0.000 description 2
- 239000010948 rhodium Substances 0.000 description 2
- 238000004626 scanning electron microscopy Methods 0.000 description 2
- 239000011163 secondary particle Substances 0.000 description 2
- 239000011734 sodium Substances 0.000 description 2
- 229910052708 sodium Inorganic materials 0.000 description 2
- 241000894007 species Species 0.000 description 2
- QAOWNCQODCNURD-UHFFFAOYSA-L sulfate group Chemical group S(=O)(=O)([O-])[O-] QAOWNCQODCNURD-UHFFFAOYSA-L 0.000 description 2
- 125000000542 sulfonic acid group Chemical group 0.000 description 2
- 239000000375 suspending agent Substances 0.000 description 2
- 238000003786 synthesis reaction Methods 0.000 description 2
- 238000010998 test method Methods 0.000 description 2
- LFQCEHFDDXELDD-UHFFFAOYSA-N tetramethyl orthosilicate Chemical compound CO[Si](OC)(OC)OC LFQCEHFDDXELDD-UHFFFAOYSA-N 0.000 description 2
- 229910001887 tin oxide Inorganic materials 0.000 description 2
- VZCYOOQTPOCHFL-UHFFFAOYSA-N trans-butenedioic acid Natural products OC(=O)C=CC(O)=O VZCYOOQTPOCHFL-UHFFFAOYSA-N 0.000 description 2
- 238000012546 transfer Methods 0.000 description 2
- GKASDNZWUGIAMG-UHFFFAOYSA-N triethyl orthoformate Chemical compound CCOC(OCC)OCC GKASDNZWUGIAMG-UHFFFAOYSA-N 0.000 description 2
- ZIBGPFATKBEMQZ-UHFFFAOYSA-N triethylene glycol Chemical compound OCCOCCOCCO ZIBGPFATKBEMQZ-UHFFFAOYSA-N 0.000 description 2
- BPSIOYPQMFLKFR-UHFFFAOYSA-N trimethoxy-[3-(oxiran-2-ylmethoxy)propyl]silane Chemical compound CO[Si](OC)(OC)CCCOCC1CO1 BPSIOYPQMFLKFR-UHFFFAOYSA-N 0.000 description 2
- ZNOKGRXACCSDPY-UHFFFAOYSA-N tungsten trioxide Chemical compound O=[W](=O)=O ZNOKGRXACCSDPY-UHFFFAOYSA-N 0.000 description 2
- 229920001567 vinyl ester resin Polymers 0.000 description 2
- 238000005406 washing Methods 0.000 description 2
- LNAZSHAWQACDHT-XIYTZBAFSA-N (2r,3r,4s,5r,6s)-4,5-dimethoxy-2-(methoxymethyl)-3-[(2s,3r,4s,5r,6r)-3,4,5-trimethoxy-6-(methoxymethyl)oxan-2-yl]oxy-6-[(2r,3r,4s,5r,6r)-4,5,6-trimethoxy-2-(methoxymethyl)oxan-3-yl]oxyoxane Chemical compound CO[C@@H]1[C@@H](OC)[C@H](OC)[C@@H](COC)O[C@H]1O[C@H]1[C@H](OC)[C@@H](OC)[C@H](O[C@H]2[C@@H]([C@@H](OC)[C@H](OC)O[C@@H]2COC)OC)O[C@@H]1COC LNAZSHAWQACDHT-XIYTZBAFSA-N 0.000 description 1
- WYTZZXDRDKSJID-UHFFFAOYSA-N (3-aminopropyl)triethoxysilane Chemical compound CCO[Si](OCC)(OCC)CCCN WYTZZXDRDKSJID-UHFFFAOYSA-N 0.000 description 1
- DXODQEHVNYHGGW-UHFFFAOYSA-N 1,1,2,2,3,3,4,4,5,5,6,6,7,7,8,8,8-heptadecafluorooctyl-tris(trifluoromethoxy)silane Chemical group FC(F)(F)O[Si](OC(F)(F)F)(OC(F)(F)F)C(F)(F)C(F)(F)C(F)(F)C(F)(F)C(F)(F)C(F)(F)C(F)(F)C(F)(F)F DXODQEHVNYHGGW-UHFFFAOYSA-N 0.000 description 1
- LAVARTIQQDZFNT-UHFFFAOYSA-N 1-(1-methoxypropan-2-yloxy)propan-2-yl acetate Chemical compound COCC(C)OCC(C)OC(C)=O LAVARTIQQDZFNT-UHFFFAOYSA-N 0.000 description 1
- UZKWTJUDCOPSNM-UHFFFAOYSA-N 1-ethenoxybutane Chemical compound CCCCOC=C UZKWTJUDCOPSNM-UHFFFAOYSA-N 0.000 description 1
- QJJDJWUCRAPCOL-UHFFFAOYSA-N 1-ethenoxyoctadecane Chemical compound CCCCCCCCCCCCCCCCCCOC=C QJJDJWUCRAPCOL-UHFFFAOYSA-N 0.000 description 1
- IXPNQXFRVYWDDI-UHFFFAOYSA-N 1-methyl-2,4-dioxo-1,3-diazinane-5-carboximidamide Chemical compound CN1CC(C(N)=N)C(=O)NC1=O IXPNQXFRVYWDDI-UHFFFAOYSA-N 0.000 description 1
- KJCVRFUGPWSIIH-UHFFFAOYSA-N 1-naphthol Chemical compound C1=CC=C2C(O)=CC=CC2=C1 KJCVRFUGPWSIIH-UHFFFAOYSA-N 0.000 description 1
- KWKAKUADMBZCLK-UHFFFAOYSA-N 1-octene Chemical compound CCCCCCC=C KWKAKUADMBZCLK-UHFFFAOYSA-N 0.000 description 1
- CPGCVOVWHCWVTP-LJAQVGFWSA-N 10-Nonacosanol Natural products CCCCCCCCCCCCCCCCCCC[C@@H](O)CCCCCCCCC CPGCVOVWHCWVTP-LJAQVGFWSA-N 0.000 description 1
- GRWZFPFQSHTXHM-UHFFFAOYSA-N 11-methyldodec-1-ene Chemical compound CC(C)CCCCCCCCC=C GRWZFPFQSHTXHM-UHFFFAOYSA-N 0.000 description 1
- FIADVASZMLCQIF-UHFFFAOYSA-N 2,2,4,4,6,6,8,8-octamethyl-1,3,5,7,2,4,6,8-tetrazatetrasilocane Chemical compound C[Si]1(C)N[Si](C)(C)N[Si](C)(C)N[Si](C)(C)N1 FIADVASZMLCQIF-UHFFFAOYSA-N 0.000 description 1
- NEBBLNDVSSWJLL-UHFFFAOYSA-N 2,3-bis(2-methylprop-2-enoyloxy)propyl 2-methylprop-2-enoate Chemical compound CC(=C)C(=O)OCC(OC(=O)C(C)=C)COC(=O)C(C)=C NEBBLNDVSSWJLL-UHFFFAOYSA-N 0.000 description 1
- OAYXUHPQHDHDDZ-UHFFFAOYSA-N 2-(2-butoxyethoxy)ethanol Chemical compound CCCCOCCOCCO OAYXUHPQHDHDDZ-UHFFFAOYSA-N 0.000 description 1
- VXQBJTKSVGFQOL-UHFFFAOYSA-N 2-(2-butoxyethoxy)ethyl acetate Chemical compound CCCCOCCOCCOC(C)=O VXQBJTKSVGFQOL-UHFFFAOYSA-N 0.000 description 1
- WMDZKDKPYCNCDZ-UHFFFAOYSA-N 2-(2-butoxypropoxy)propan-1-ol Chemical compound CCCCOC(C)COC(C)CO WMDZKDKPYCNCDZ-UHFFFAOYSA-N 0.000 description 1
- FPZWZCWUIYYYBU-UHFFFAOYSA-N 2-(2-ethoxyethoxy)ethyl acetate Chemical compound CCOCCOCCOC(C)=O FPZWZCWUIYYYBU-UHFFFAOYSA-N 0.000 description 1
- DJCYDDALXPHSHR-UHFFFAOYSA-N 2-(2-propoxyethoxy)ethanol Chemical compound CCCOCCOCCO DJCYDDALXPHSHR-UHFFFAOYSA-N 0.000 description 1
- XYVAYAJYLWYJJN-UHFFFAOYSA-N 2-(2-propoxypropoxy)propan-1-ol Chemical compound CCCOC(C)COC(C)CO XYVAYAJYLWYJJN-UHFFFAOYSA-N 0.000 description 1
- BEWCNXNIQCLWHP-UHFFFAOYSA-N 2-(tert-butylamino)ethyl 2-methylprop-2-enoate Chemical compound CC(=C)C(=O)OCCNC(C)(C)C BEWCNXNIQCLWHP-UHFFFAOYSA-N 0.000 description 1
- JDSQBDGCMUXRBM-UHFFFAOYSA-N 2-[2-(2-butoxypropoxy)propoxy]propan-1-ol Chemical compound CCCCOC(C)COC(C)COC(C)CO JDSQBDGCMUXRBM-UHFFFAOYSA-N 0.000 description 1
- HZBGBOWFTGSNLM-UHFFFAOYSA-N 2-[2-(2-methoxyethoxy)ethoxy]ethyl carbamate Chemical compound COCCOCCOCCOC(N)=O HZBGBOWFTGSNLM-UHFFFAOYSA-N 0.000 description 1
- WAEVWDZKMBQDEJ-UHFFFAOYSA-N 2-[2-(2-methoxypropoxy)propoxy]propan-1-ol Chemical compound COC(C)COC(C)COC(C)CO WAEVWDZKMBQDEJ-UHFFFAOYSA-N 0.000 description 1
- TXBCBTDQIULDIA-UHFFFAOYSA-N 2-[[3-hydroxy-2,2-bis(hydroxymethyl)propoxy]methyl]-2-(hydroxymethyl)propane-1,3-diol Chemical compound OCC(CO)(CO)COCC(CO)(CO)CO TXBCBTDQIULDIA-UHFFFAOYSA-N 0.000 description 1
- NECRQCBKTGZNMH-UHFFFAOYSA-N 3,5-dimethylhex-1-yn-3-ol Chemical class CC(C)CC(C)(O)C#C NECRQCBKTGZNMH-UHFFFAOYSA-N 0.000 description 1
- FMGBDYLOANULLW-UHFFFAOYSA-N 3-isocyanatopropyl(trimethoxy)silane Chemical compound CO[Si](OC)(OC)CCCN=C=O FMGBDYLOANULLW-UHFFFAOYSA-N 0.000 description 1
- SJECZPVISLOESU-UHFFFAOYSA-N 3-trimethoxysilylpropan-1-amine Chemical compound CO[Si](OC)(OC)CCCN SJECZPVISLOESU-UHFFFAOYSA-N 0.000 description 1
- UUEWCQRISZBELL-UHFFFAOYSA-N 3-trimethoxysilylpropane-1-thiol Chemical compound CO[Si](OC)(OC)CCCS UUEWCQRISZBELL-UHFFFAOYSA-N 0.000 description 1
- WXNZTHHGJRFXKQ-UHFFFAOYSA-N 4-chlorophenol Chemical compound OC1=CC=C(Cl)C=C1 WXNZTHHGJRFXKQ-UHFFFAOYSA-N 0.000 description 1
- HRPVXLWXLXDGHG-UHFFFAOYSA-N Acrylamide Chemical compound NC(=O)C=C HRPVXLWXLXDGHG-UHFFFAOYSA-N 0.000 description 1
- 229920001817 Agar Polymers 0.000 description 1
- 239000005995 Aluminium silicate Substances 0.000 description 1
- NLXLAEXVIDQMFP-UHFFFAOYSA-N Ammonium chloride Substances [NH4+].[Cl-] NLXLAEXVIDQMFP-UHFFFAOYSA-N 0.000 description 1
- VHUUQVKOLVNVRT-UHFFFAOYSA-N Ammonium hydroxide Chemical compound [NH4+].[OH-] VHUUQVKOLVNVRT-UHFFFAOYSA-N 0.000 description 1
- 241000416162 Astragalus gummifer Species 0.000 description 1
- IJGRMHOSHXDMSA-UHFFFAOYSA-N Atomic nitrogen Chemical compound N#N IJGRMHOSHXDMSA-UHFFFAOYSA-N 0.000 description 1
- 235000016068 Berberis vulgaris Nutrition 0.000 description 1
- 241000335053 Beta vulgaris Species 0.000 description 1
- WUIRGUDWPKRGHJ-UHFFFAOYSA-N CC(=C)C(O)=O.CC(=C)C(O)=O.CC(=C)C(O)=O.CC(=C)C(O)=O.CC(=C)C(O)=O.OCC(CO)(CO)CO Chemical compound CC(=C)C(O)=O.CC(=C)C(O)=O.CC(=C)C(O)=O.CC(=C)C(O)=O.CC(=C)C(O)=O.OCC(CO)(CO)CO WUIRGUDWPKRGHJ-UHFFFAOYSA-N 0.000 description 1
- OYPRJOBELJOOCE-UHFFFAOYSA-N Calcium Chemical compound [Ca] OYPRJOBELJOOCE-UHFFFAOYSA-N 0.000 description 1
- BVKZGUZCCUSVTD-UHFFFAOYSA-L Carbonate Chemical compound [O-]C([O-])=O BVKZGUZCCUSVTD-UHFFFAOYSA-L 0.000 description 1
- 206010007269 Carcinogenicity Diseases 0.000 description 1
- 241000195649 Chlorella <Chlorellales> Species 0.000 description 1
- 241000206575 Chondrus crispus Species 0.000 description 1
- 229920002307 Dextran Polymers 0.000 description 1
- GDFCSMCGLZFNFY-UHFFFAOYSA-N Dimethylaminopropyl Methacrylamide Chemical compound CN(C)CCCNC(=O)C(C)=C GDFCSMCGLZFNFY-UHFFFAOYSA-N 0.000 description 1
- VGGSQFUCUMXWEO-UHFFFAOYSA-N Ethene Chemical compound C=C VGGSQFUCUMXWEO-UHFFFAOYSA-N 0.000 description 1
- 239000001856 Ethyl cellulose Substances 0.000 description 1
- ZZSNKZQZMQGXPY-UHFFFAOYSA-N Ethyl cellulose Chemical compound CCOCC1OC(OC)C(OCC)C(OCC)C1OC1C(O)C(O)C(OC)C(CO)O1 ZZSNKZQZMQGXPY-UHFFFAOYSA-N 0.000 description 1
- 239000005977 Ethylene Substances 0.000 description 1
- 229920002907 Guar gum Polymers 0.000 description 1
- 229920000569 Gum karaya Polymers 0.000 description 1
- UFHFLCQGNIYNRP-UHFFFAOYSA-N Hydrogen Chemical compound [H][H] UFHFLCQGNIYNRP-UHFFFAOYSA-N 0.000 description 1
- 239000004354 Hydroxyethyl cellulose Substances 0.000 description 1
- 229920000663 Hydroxyethyl cellulose Polymers 0.000 description 1
- WOBHKFSMXKNTIM-UHFFFAOYSA-N Hydroxyethyl methacrylate Chemical compound CC(=C)C(=O)OCCO WOBHKFSMXKNTIM-UHFFFAOYSA-N 0.000 description 1
- 239000005909 Kieselgur Substances 0.000 description 1
- 229920006370 Kynar Polymers 0.000 description 1
- 239000004166 Lanolin Substances 0.000 description 1
- 229920000161 Locust bean gum Polymers 0.000 description 1
- FYYHWMGAXLPEAU-UHFFFAOYSA-N Magnesium Chemical compound [Mg] FYYHWMGAXLPEAU-UHFFFAOYSA-N 0.000 description 1
- ZOKXTWBITQBERF-UHFFFAOYSA-N Molybdenum Chemical compound [Mo] ZOKXTWBITQBERF-UHFFFAOYSA-N 0.000 description 1
- 239000000020 Nitrocellulose Substances 0.000 description 1
- 229910003849 O-Si Inorganic materials 0.000 description 1
- BKQMNPVDJIHLPD-UHFFFAOYSA-N OS(=O)(=O)[Se]S(O)(=O)=O Chemical class OS(=O)(=O)[Se]S(O)(=O)=O BKQMNPVDJIHLPD-UHFFFAOYSA-N 0.000 description 1
- 240000007594 Oryza sativa Species 0.000 description 1
- 235000007164 Oryza sativa Nutrition 0.000 description 1
- 241000192497 Oscillatoria Species 0.000 description 1
- 229910003872 O—Si Inorganic materials 0.000 description 1
- 229930040373 Paraformaldehyde Natural products 0.000 description 1
- 108091005804 Peptidases Proteins 0.000 description 1
- NBIIXXVUZAFLBC-UHFFFAOYSA-N Phosphoric acid Chemical group OP(O)(O)=O NBIIXXVUZAFLBC-UHFFFAOYSA-N 0.000 description 1
- 229920003171 Poly (ethylene oxide) Polymers 0.000 description 1
- 229920002518 Polyallylamine hydrochloride Polymers 0.000 description 1
- 229920002367 Polyisobutene Polymers 0.000 description 1
- 239000004372 Polyvinyl alcohol Substances 0.000 description 1
- 239000004365 Protease Substances 0.000 description 1
- 102100037486 Reverse transcriptase/ribonuclease H Human genes 0.000 description 1
- PMZURENOXWZQFD-UHFFFAOYSA-L Sodium Sulfate Chemical compound [Na+].[Na+].[O-]S([O-])(=O)=O PMZURENOXWZQFD-UHFFFAOYSA-L 0.000 description 1
- 244000061456 Solanum tuberosum Species 0.000 description 1
- 235000002595 Solanum tuberosum Nutrition 0.000 description 1
- 238000000944 Soxhlet extraction Methods 0.000 description 1
- 241000934878 Sterculia Species 0.000 description 1
- 229920001615 Tragacanth Polymers 0.000 description 1
- OKKRPWIIYQTPQF-UHFFFAOYSA-N Trimethylolpropane trimethacrylate Chemical compound CC(=C)C(=O)OCC(CC)(COC(=O)C(C)=C)COC(=O)C(C)=C OKKRPWIIYQTPQF-UHFFFAOYSA-N 0.000 description 1
- 235000021307 Triticum Nutrition 0.000 description 1
- 244000098338 Triticum aestivum Species 0.000 description 1
- 239000013504 Triton X-100 Substances 0.000 description 1
- 229920004890 Triton X-100 Polymers 0.000 description 1
- 229920004896 Triton X-405 Polymers 0.000 description 1
- 230000006750 UV protection Effects 0.000 description 1
- QYKIQEUNHZKYBP-UHFFFAOYSA-N Vinyl ether Chemical class C=COC=C QYKIQEUNHZKYBP-UHFFFAOYSA-N 0.000 description 1
- 240000008042 Zea mays Species 0.000 description 1
- 235000005824 Zea mays ssp. parviglumis Nutrition 0.000 description 1
- 235000002017 Zea mays subsp mays Nutrition 0.000 description 1
- MCMNRKCIXSYSNV-UHFFFAOYSA-N ZrO2 Inorganic materials O=[Zr]=O MCMNRKCIXSYSNV-UHFFFAOYSA-N 0.000 description 1
- FJWGYAHXMCUOOM-QHOUIDNNSA-N [(2s,3r,4s,5r,6r)-2-[(2r,3r,4s,5r,6s)-4,5-dinitrooxy-2-(nitrooxymethyl)-6-[(2r,3r,4s,5r,6s)-4,5,6-trinitrooxy-2-(nitrooxymethyl)oxan-3-yl]oxyoxan-3-yl]oxy-3,5-dinitrooxy-6-(nitrooxymethyl)oxan-4-yl] nitrate Chemical compound O([C@@H]1O[C@@H]([C@H]([C@H](O[N+]([O-])=O)[C@H]1O[N+]([O-])=O)O[C@H]1[C@@H]([C@@H](O[N+]([O-])=O)[C@H](O[N+]([O-])=O)[C@@H](CO[N+]([O-])=O)O1)O[N+]([O-])=O)CO[N+](=O)[O-])[C@@H]1[C@@H](CO[N+]([O-])=O)O[C@@H](O[N+]([O-])=O)[C@H](O[N+]([O-])=O)[C@H]1O[N+]([O-])=O FJWGYAHXMCUOOM-QHOUIDNNSA-N 0.000 description 1
- GJWAPAVRQYYSTK-UHFFFAOYSA-N [(dimethyl-$l^{3}-silanyl)amino]-dimethylsilicon Chemical compound C[Si](C)N[Si](C)C GJWAPAVRQYYSTK-UHFFFAOYSA-N 0.000 description 1
- GQPVFBDWIUVLHG-UHFFFAOYSA-N [2,2-bis(hydroxymethyl)-3-(2-methylprop-2-enoyloxy)propyl] 2-methylprop-2-enoate Chemical compound CC(=C)C(=O)OCC(CO)(CO)COC(=O)C(C)=C GQPVFBDWIUVLHG-UHFFFAOYSA-N 0.000 description 1
- JUDXBRVLWDGRBC-UHFFFAOYSA-N [2-(hydroxymethyl)-3-(2-methylprop-2-enoyloxy)-2-(2-methylprop-2-enoyloxymethyl)propyl] 2-methylprop-2-enoate Chemical compound CC(=C)C(=O)OCC(CO)(COC(=O)C(C)=C)COC(=O)C(C)=C JUDXBRVLWDGRBC-UHFFFAOYSA-N 0.000 description 1
- QUEDYRXQWSDKKG-UHFFFAOYSA-M [O-2].[O-2].[V+5].[OH-] Chemical compound [O-2].[O-2].[V+5].[OH-] QUEDYRXQWSDKKG-UHFFFAOYSA-M 0.000 description 1
- HZEWFHLRYVTOIW-UHFFFAOYSA-N [Ti].[Ni] Chemical compound [Ti].[Ni] HZEWFHLRYVTOIW-UHFFFAOYSA-N 0.000 description 1
- RQVFGTYFBUVGOP-UHFFFAOYSA-N [acetyloxy(dimethyl)silyl] acetate Chemical compound CC(=O)O[Si](C)(C)OC(C)=O RQVFGTYFBUVGOP-UHFFFAOYSA-N 0.000 description 1
- TVJPBVNWVPUZBM-UHFFFAOYSA-N [diacetyloxy(methyl)silyl] acetate Chemical compound CC(=O)O[Si](C)(OC(C)=O)OC(C)=O TVJPBVNWVPUZBM-UHFFFAOYSA-N 0.000 description 1
- 238000002835 absorbance Methods 0.000 description 1
- 150000001242 acetic acid derivatives Chemical class 0.000 description 1
- 150000007513 acids Chemical class 0.000 description 1
- 125000005396 acrylic acid ester group Chemical group 0.000 description 1
- 229910052768 actinide Inorganic materials 0.000 description 1
- 150000001255 actinides Chemical class 0.000 description 1
- 230000009471 action Effects 0.000 description 1
- 230000002411 adverse Effects 0.000 description 1
- 239000008272 agar Substances 0.000 description 1
- 235000010419 agar Nutrition 0.000 description 1
- 238000004220 aggregation Methods 0.000 description 1
- 238000013019 agitation Methods 0.000 description 1
- 150000004781 alginic acids Chemical class 0.000 description 1
- 150000001335 aliphatic alkanes Chemical class 0.000 description 1
- 125000001931 aliphatic group Chemical group 0.000 description 1
- 150000001340 alkali metals Chemical class 0.000 description 1
- 150000001336 alkenes Chemical class 0.000 description 1
- 125000005370 alkoxysilyl group Chemical group 0.000 description 1
- 125000002947 alkylene group Chemical group 0.000 description 1
- XYLMUPLGERFSHI-UHFFFAOYSA-N alpha-Methylstyrene Chemical compound CC(=C)C1=CC=CC=C1 XYLMUPLGERFSHI-UHFFFAOYSA-N 0.000 description 1
- 230000004075 alteration Effects 0.000 description 1
- 235000012211 aluminium silicate Nutrition 0.000 description 1
- 150000001408 amides Chemical class 0.000 description 1
- 125000004103 aminoalkyl group Chemical group 0.000 description 1
- 235000011114 ammonium hydroxide Nutrition 0.000 description 1
- 229920006318 anionic polymer Polymers 0.000 description 1
- 238000002048 anodisation reaction Methods 0.000 description 1
- 230000002547 anomalous effect Effects 0.000 description 1
- 239000003242 anti bacterial agent Substances 0.000 description 1
- 239000008346 aqueous phase Substances 0.000 description 1
- 238000000429 assembly Methods 0.000 description 1
- 230000000712 assembly Effects 0.000 description 1
- 238000000089 atomic force micrograph Methods 0.000 description 1
- DMLAVOWQYNRWNQ-UHFFFAOYSA-N azobenzene Chemical compound C1=CC=CC=C1N=NC1=CC=CC=C1 DMLAVOWQYNRWNQ-UHFFFAOYSA-N 0.000 description 1
- 235000013871 bee wax Nutrition 0.000 description 1
- 239000012166 beeswax Substances 0.000 description 1
- 230000002902 bimodal effect Effects 0.000 description 1
- 230000033228 biological regulation Effects 0.000 description 1
- RSOILICUEWXSLA-UHFFFAOYSA-N bis(1,2,2,6,6-pentamethylpiperidin-4-yl) decanedioate Chemical compound C1C(C)(C)N(C)C(C)(C)CC1OC(=O)CCCCCCCCC(=O)OC1CC(C)(C)N(C)C(C)(C)C1 RSOILICUEWXSLA-UHFFFAOYSA-N 0.000 description 1
- 229910052797 bismuth Inorganic materials 0.000 description 1
- WMWLMWRWZQELOS-UHFFFAOYSA-N bismuth(iii) oxide Chemical compound O=[Bi]O[Bi]=O WMWLMWRWZQELOS-UHFFFAOYSA-N 0.000 description 1
- 150000001642 boronic acid derivatives Chemical class 0.000 description 1
- 230000001680 brushing effect Effects 0.000 description 1
- 239000004566 building material Substances 0.000 description 1
- 238000012662 bulk polymerization Methods 0.000 description 1
- XGZGKDQVCBHSGI-UHFFFAOYSA-N butyl(triethoxy)silane Chemical compound CCCC[Si](OCC)(OCC)OCC XGZGKDQVCBHSGI-UHFFFAOYSA-N 0.000 description 1
- SXPLZNMUBFBFIA-UHFFFAOYSA-N butyl(trimethoxy)silane Chemical compound CCCC[Si](OC)(OC)OC SXPLZNMUBFBFIA-UHFFFAOYSA-N 0.000 description 1
- 239000011575 calcium Substances 0.000 description 1
- 229910052791 calcium Inorganic materials 0.000 description 1
- 239000004204 candelilla wax Substances 0.000 description 1
- 235000013868 candelilla wax Nutrition 0.000 description 1
- 229940073532 candelilla wax Drugs 0.000 description 1
- 229910052799 carbon Inorganic materials 0.000 description 1
- 239000006229 carbon black Substances 0.000 description 1
- 235000019241 carbon black Nutrition 0.000 description 1
- 239000002041 carbon nanotube Substances 0.000 description 1
- 229910021393 carbon nanotube Inorganic materials 0.000 description 1
- 125000003178 carboxy group Chemical group [H]OC(*)=O 0.000 description 1
- 239000001768 carboxy methyl cellulose Substances 0.000 description 1
- 150000001733 carboxylic acid esters Chemical class 0.000 description 1
- 125000002057 carboxymethyl group Chemical group [H]OC(=O)C([H])([H])[*] 0.000 description 1
- 230000007670 carcinogenicity Effects 0.000 description 1
- 231100000260 carcinogenicity Toxicity 0.000 description 1
- 239000004203 carnauba wax Substances 0.000 description 1
- 235000013869 carnauba wax Nutrition 0.000 description 1
- 229920006317 cationic polymer Polymers 0.000 description 1
- 239000000919 ceramic Substances 0.000 description 1
- 229910000422 cerium(IV) oxide Inorganic materials 0.000 description 1
- 125000001309 chloro group Chemical group Cl* 0.000 description 1
- XSDCTSITJJJDPY-UHFFFAOYSA-N chloro-ethenyl-dimethylsilane Chemical compound C[Si](C)(Cl)C=C XSDCTSITJJJDPY-UHFFFAOYSA-N 0.000 description 1
- 229910052570 clay Inorganic materials 0.000 description 1
- 239000011538 cleaning material Substances 0.000 description 1
- 238000004581 coalescence Methods 0.000 description 1
- 229910052681 coesite Inorganic materials 0.000 description 1
- 229940075614 colloidal silicon dioxide Drugs 0.000 description 1
- 230000002301 combined effect Effects 0.000 description 1
- 239000012141 concentrate Substances 0.000 description 1
- 238000009833 condensation Methods 0.000 description 1
- 230000005494 condensation Effects 0.000 description 1
- 238000006482 condensation reaction Methods 0.000 description 1
- 238000007334 copolymerization reaction Methods 0.000 description 1
- SEVNKWFHTNVOLD-UHFFFAOYSA-L copper;3-(4-ethylcyclohexyl)propanoate;3-(3-ethylcyclopentyl)propanoate Chemical compound [Cu+2].CCC1CCC(CCC([O-])=O)C1.CCC1CCC(CCC([O-])=O)CC1 SEVNKWFHTNVOLD-UHFFFAOYSA-L 0.000 description 1
- 235000005822 corn Nutrition 0.000 description 1
- 239000011557 critical solution Substances 0.000 description 1
- 239000003431 cross linking reagent Substances 0.000 description 1
- 150000001925 cycloalkenes Chemical class 0.000 description 1
- URYYVOIYTNXXBN-UPHRSURJSA-N cyclooctene Chemical compound C1CCC\C=C/CC1 URYYVOIYTNXXBN-UPHRSURJSA-N 0.000 description 1
- 239000004913 cyclooctene Substances 0.000 description 1
- 230000003247 decreasing effect Effects 0.000 description 1
- 238000013461 design Methods 0.000 description 1
- 230000006866 deterioration Effects 0.000 description 1
- 230000001627 detrimental effect Effects 0.000 description 1
- AQEFLFZSWDEAIP-UHFFFAOYSA-N di-tert-butyl ether Chemical compound CC(C)(C)OC(C)(C)C AQEFLFZSWDEAIP-UHFFFAOYSA-N 0.000 description 1
- SWXVUIWOUIDPGS-UHFFFAOYSA-N diacetone alcohol Natural products CC(=O)CC(C)(C)O SWXVUIWOUIDPGS-UHFFFAOYSA-N 0.000 description 1
- YLJJAVFOBDSYAN-UHFFFAOYSA-N dichloro-ethenyl-methylsilane Chemical compound C[Si](Cl)(Cl)C=C YLJJAVFOBDSYAN-UHFFFAOYSA-N 0.000 description 1
- GYWBHBXGYTYXRG-UHFFFAOYSA-N dichloro-methyl-octadecylsilane Chemical compound CCCCCCCCCCCCCCCCCC[Si](C)(Cl)Cl GYWBHBXGYTYXRG-UHFFFAOYSA-N 0.000 description 1
- QHBMMABVNRSRHW-UHFFFAOYSA-N dichloro-methyl-octylsilane Chemical compound CCCCCCCC[Si](C)(Cl)Cl QHBMMABVNRSRHW-UHFFFAOYSA-N 0.000 description 1
- 238000001938 differential scanning calorimetry curve Methods 0.000 description 1
- NZZFYRREKKOMAT-UHFFFAOYSA-N diiodomethane Chemical compound ICI NZZFYRREKKOMAT-UHFFFAOYSA-N 0.000 description 1
- FSBVERYRVPGNGG-UHFFFAOYSA-N dimagnesium dioxido-bis[[oxido(oxo)silyl]oxy]silane hydrate Chemical compound O.[Mg+2].[Mg+2].[O-][Si](=O)O[Si]([O-])([O-])O[Si]([O-])=O FSBVERYRVPGNGG-UHFFFAOYSA-N 0.000 description 1
- 239000000539 dimer Substances 0.000 description 1
- JJQZDUKDJDQPMQ-UHFFFAOYSA-N dimethoxy(dimethyl)silane Chemical compound CO[Si](C)(C)OC JJQZDUKDJDQPMQ-UHFFFAOYSA-N 0.000 description 1
- LIKFHECYJZWXFJ-UHFFFAOYSA-N dimethyldichlorosilane Chemical compound C[Si](C)(Cl)Cl LIKFHECYJZWXFJ-UHFFFAOYSA-N 0.000 description 1
- YYLGKUPAFFKGRQ-UHFFFAOYSA-N dimethyldiethoxysilane Chemical compound CCO[Si](C)(C)OCC YYLGKUPAFFKGRQ-UHFFFAOYSA-N 0.000 description 1
- 230000003292 diminished effect Effects 0.000 description 1
- 229910001873 dinitrogen Inorganic materials 0.000 description 1
- SZXQTJUDPRGNJN-UHFFFAOYSA-N dipropylene glycol Chemical compound OCCCOCCCO SZXQTJUDPRGNJN-UHFFFAOYSA-N 0.000 description 1
- AFOSIXZFDONLBT-UHFFFAOYSA-N divinyl sulfone Chemical compound C=CS(=O)(=O)C=C AFOSIXZFDONLBT-UHFFFAOYSA-N 0.000 description 1
- WNAHIZMDSQCWRP-UHFFFAOYSA-N dodecane-1-thiol Chemical compound CCCCCCCCCCCCS WNAHIZMDSQCWRP-UHFFFAOYSA-N 0.000 description 1
- 125000003438 dodecyl group Chemical group [H]C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])* 0.000 description 1
- 229920001971 elastomer Polymers 0.000 description 1
- 230000005684 electric field Effects 0.000 description 1
- 238000004070 electrodeposition Methods 0.000 description 1
- 230000008030 elimination Effects 0.000 description 1
- 238000003379 elimination reaction Methods 0.000 description 1
- 229910001651 emery Inorganic materials 0.000 description 1
- 238000005516 engineering process Methods 0.000 description 1
- 239000003623 enhancer Substances 0.000 description 1
- 230000003628 erosive effect Effects 0.000 description 1
- WNMORWGTPVWAIB-UHFFFAOYSA-N ethenyl 2-methylpropanoate Chemical compound CC(C)C(=O)OC=C WNMORWGTPVWAIB-UHFFFAOYSA-N 0.000 description 1
- MEGHWIAOTJPCHQ-UHFFFAOYSA-N ethenyl butanoate Chemical compound CCCC(=O)OC=C MEGHWIAOTJPCHQ-UHFFFAOYSA-N 0.000 description 1
- GLVVKKSPKXTQRB-UHFFFAOYSA-N ethenyl dodecanoate Chemical compound CCCCCCCCCCCC(=O)OC=C GLVVKKSPKXTQRB-UHFFFAOYSA-N 0.000 description 1
- LZWYWAIOTBEZFN-UHFFFAOYSA-N ethenyl hexanoate Chemical compound CCCCCC(=O)OC=C LZWYWAIOTBEZFN-UHFFFAOYSA-N 0.000 description 1
- AFSIMBWBBOJPJG-UHFFFAOYSA-N ethenyl octadecanoate Chemical compound CCCCCCCCCCCCCCCCCC(=O)OC=C AFSIMBWBBOJPJG-UHFFFAOYSA-N 0.000 description 1
- QBDADGJLZNIRFQ-UHFFFAOYSA-N ethenyl octanoate Chemical compound CCCCCCCC(=O)OC=C QBDADGJLZNIRFQ-UHFFFAOYSA-N 0.000 description 1
- UIWXSTHGICQLQT-UHFFFAOYSA-N ethenyl propanoate Chemical compound CCC(=O)OC=C UIWXSTHGICQLQT-UHFFFAOYSA-N 0.000 description 1
- FWDBOZPQNFPOLF-UHFFFAOYSA-N ethenyl(triethoxy)silane Chemical compound CCO[Si](OCC)(OCC)C=C FWDBOZPQNFPOLF-UHFFFAOYSA-N 0.000 description 1
- MBGQQKKTDDNCSG-UHFFFAOYSA-N ethenyl-diethoxy-methylsilane Chemical compound CCO[Si](C)(C=C)OCC MBGQQKKTDDNCSG-UHFFFAOYSA-N 0.000 description 1
- ZLNAFSPCNATQPQ-UHFFFAOYSA-N ethenyl-dimethoxy-methylsilane Chemical compound CO[Si](C)(OC)C=C ZLNAFSPCNATQPQ-UHFFFAOYSA-N 0.000 description 1
- JEWCZPTVOYXPGG-UHFFFAOYSA-N ethenyl-ethoxy-dimethylsilane Chemical compound CCO[Si](C)(C)C=C JEWCZPTVOYXPGG-UHFFFAOYSA-N 0.000 description 1
- NUFVQEIPPHHQCK-UHFFFAOYSA-N ethenyl-methoxy-dimethylsilane Chemical compound CO[Si](C)(C)C=C NUFVQEIPPHHQCK-UHFFFAOYSA-N 0.000 description 1
- ZZRGHKUNLAYDTC-UHFFFAOYSA-N ethoxy(methyl)silane Chemical class CCO[SiH2]C ZZRGHKUNLAYDTC-UHFFFAOYSA-N 0.000 description 1
- RSIHJDGMBDPTIM-UHFFFAOYSA-N ethoxy(trimethyl)silane Chemical compound CCO[Si](C)(C)C RSIHJDGMBDPTIM-UHFFFAOYSA-N 0.000 description 1
- UHESRSKEBRADOO-UHFFFAOYSA-N ethyl carbamate;prop-2-enoic acid Chemical class OC(=O)C=C.CCOC(N)=O UHESRSKEBRADOO-UHFFFAOYSA-N 0.000 description 1
- 235000019325 ethyl cellulose Nutrition 0.000 description 1
- 229920001249 ethyl cellulose Polymers 0.000 description 1
- SBRXLTRZCJVAPH-UHFFFAOYSA-N ethyl(trimethoxy)silane Chemical compound CC[Si](OC)(OC)OC SBRXLTRZCJVAPH-UHFFFAOYSA-N 0.000 description 1
- 150000002194 fatty esters Chemical class 0.000 description 1
- 230000002349 favourable effect Effects 0.000 description 1
- 239000011518 fibre cement Substances 0.000 description 1
- 239000005357 flat glass Substances 0.000 description 1
- 239000012530 fluid Substances 0.000 description 1
- 125000006162 fluoroaliphatic group Chemical group 0.000 description 1
- XPBBUZJBQWWFFJ-UHFFFAOYSA-N fluorosilane Chemical compound [SiH3]F XPBBUZJBQWWFFJ-UHFFFAOYSA-N 0.000 description 1
- 239000011888 foil Substances 0.000 description 1
- HKIOYBQGHSTUDB-UHFFFAOYSA-N folpet Chemical compound C1=CC=C2C(=O)N(SC(Cl)(Cl)Cl)C(=O)C2=C1 HKIOYBQGHSTUDB-UHFFFAOYSA-N 0.000 description 1
- 238000013467 fragmentation Methods 0.000 description 1
- 238000006062 fragmentation reaction Methods 0.000 description 1
- 238000010528 free radical solution polymerization reaction Methods 0.000 description 1
- 230000008014 freezing Effects 0.000 description 1
- 238000007710 freezing Methods 0.000 description 1
- 230000002538 fungal effect Effects 0.000 description 1
- 239000007789 gas Substances 0.000 description 1
- 238000001879 gelation Methods 0.000 description 1
- 239000012362 glacial acetic acid Substances 0.000 description 1
- 125000005417 glycidoxyalkyl group Chemical group 0.000 description 1
- 230000005484 gravity Effects 0.000 description 1
- 235000010417 guar gum Nutrition 0.000 description 1
- 239000000665 guar gum Substances 0.000 description 1
- 229960002154 guar gum Drugs 0.000 description 1
- 210000004209 hair Anatomy 0.000 description 1
- 238000003306 harvesting Methods 0.000 description 1
- IUJAMGNYPWYUPM-UHFFFAOYSA-N hentriacontane Chemical compound CCCCCCCCCCCCCCCCCCCCCCCCCCCCCCC IUJAMGNYPWYUPM-UHFFFAOYSA-N 0.000 description 1
- CZWLNMOIEMTDJY-UHFFFAOYSA-N hexyl(trimethoxy)silane Chemical compound CCCCCC[Si](OC)(OC)OC CZWLNMOIEMTDJY-UHFFFAOYSA-N 0.000 description 1
- 238000004128 high performance liquid chromatography Methods 0.000 description 1
- 229920001519 homopolymer Polymers 0.000 description 1
- 150000002430 hydrocarbons Chemical group 0.000 description 1
- 239000001257 hydrogen Substances 0.000 description 1
- 229910052739 hydrogen Inorganic materials 0.000 description 1
- 235000019447 hydroxyethyl cellulose Nutrition 0.000 description 1
- 229910052588 hydroxylapatite Inorganic materials 0.000 description 1
- 229920013818 hydroxypropyl guar gum Polymers 0.000 description 1
- 239000001866 hydroxypropyl methyl cellulose Substances 0.000 description 1
- 235000010979 hydroxypropyl methyl cellulose Nutrition 0.000 description 1
- 229920003088 hydroxypropyl methyl cellulose Polymers 0.000 description 1
- UFVKGYZPFZQRLF-UHFFFAOYSA-N hydroxypropyl methyl cellulose Chemical compound OC1C(O)C(OC)OC(CO)C1OC1C(O)C(O)C(OC2C(C(O)C(OC3C(C(O)C(O)C(CO)O3)O)C(CO)O2)O)C(CO)O1 UFVKGYZPFZQRLF-UHFFFAOYSA-N 0.000 description 1
- 238000007654 immersion Methods 0.000 description 1
- 238000005470 impregnation Methods 0.000 description 1
- 230000000977 initiatory effect Effects 0.000 description 1
- 150000002484 inorganic compounds Chemical class 0.000 description 1
- 229910010272 inorganic material Inorganic materials 0.000 description 1
- 229920000592 inorganic polymer Polymers 0.000 description 1
- 125000003010 ionic group Chemical group 0.000 description 1
- 150000002500 ions Chemical class 0.000 description 1
- DCYOBGZUOMKFPA-UHFFFAOYSA-N iron(2+);iron(3+);octadecacyanide Chemical compound [Fe+2].[Fe+2].[Fe+2].[Fe+3].[Fe+3].[Fe+3].[Fe+3].N#[C-].N#[C-].N#[C-].N#[C-].N#[C-].N#[C-].N#[C-].N#[C-].N#[C-].N#[C-].N#[C-].N#[C-].N#[C-].N#[C-].N#[C-].N#[C-].N#[C-].N#[C-] DCYOBGZUOMKFPA-UHFFFAOYSA-N 0.000 description 1
- 230000001788 irregular Effects 0.000 description 1
- 230000002427 irreversible effect Effects 0.000 description 1
- IQPQWNKOIGAROB-UHFFFAOYSA-N isocyanate group Chemical group [N-]=C=O IQPQWNKOIGAROB-UHFFFAOYSA-N 0.000 description 1
- 229910052622 kaolinite Inorganic materials 0.000 description 1
- 235000010494 karaya gum Nutrition 0.000 description 1
- 239000000231 karaya gum Substances 0.000 description 1
- 229940039371 karaya gum Drugs 0.000 description 1
- 235000019388 lanolin Nutrition 0.000 description 1
- 229910052747 lanthanoid Inorganic materials 0.000 description 1
- 150000002602 lanthanoids Chemical class 0.000 description 1
- 229910052746 lanthanum Inorganic materials 0.000 description 1
- CDOSHBSSFJOMGT-UHFFFAOYSA-N linalool Chemical compound CC(C)=CCCC(C)(O)C=C CDOSHBSSFJOMGT-UHFFFAOYSA-N 0.000 description 1
- 238000010550 living polymerization reaction Methods 0.000 description 1
- 235000010420 locust bean gum Nutrition 0.000 description 1
- 239000000711 locust bean gum Substances 0.000 description 1
- 230000005923 long-lasting effect Effects 0.000 description 1
- 239000001095 magnesium carbonate Substances 0.000 description 1
- 229910000021 magnesium carbonate Inorganic materials 0.000 description 1
- 239000000391 magnesium silicate Substances 0.000 description 1
- 229910052919 magnesium silicate Inorganic materials 0.000 description 1
- 235000019792 magnesium silicate Nutrition 0.000 description 1
- 229910052748 manganese Inorganic materials 0.000 description 1
- 239000011572 manganese Substances 0.000 description 1
- WPBNNNQJVZRUHP-UHFFFAOYSA-L manganese(2+);methyl n-[[2-(methoxycarbonylcarbamothioylamino)phenyl]carbamothioyl]carbamate;n-[2-(sulfidocarbothioylamino)ethyl]carbamodithioate Chemical compound [Mn+2].[S-]C(=S)NCCNC([S-])=S.COC(=O)NC(=S)NC1=CC=CC=C1NC(=S)NC(=O)OC WPBNNNQJVZRUHP-UHFFFAOYSA-L 0.000 description 1
- 239000011159 matrix material Substances 0.000 description 1
- QSHDDOUJBYECFT-UHFFFAOYSA-N mercury Chemical compound [Hg] QSHDDOUJBYECFT-UHFFFAOYSA-N 0.000 description 1
- 229910052753 mercury Inorganic materials 0.000 description 1
- 229910001507 metal halide Inorganic materials 0.000 description 1
- 150000005309 metal halides Chemical class 0.000 description 1
- 229910052976 metal sulfide Inorganic materials 0.000 description 1
- 229910052752 metalloid Inorganic materials 0.000 description 1
- 150000002738 metalloids Chemical class 0.000 description 1
- 125000005397 methacrylic acid ester group Chemical group 0.000 description 1
- 125000005395 methacrylic acid group Chemical group 0.000 description 1
- MYWUZJCMWCOHBA-VIFPVBQESA-N methamphetamine Chemical compound CN[C@@H](C)CC1=CC=CC=C1 MYWUZJCMWCOHBA-VIFPVBQESA-N 0.000 description 1
- OKKJLVBELUTLKV-UHFFFAOYSA-N methanol Natural products OC OKKJLVBELUTLKV-UHFFFAOYSA-N 0.000 description 1
- 125000000956 methoxy group Chemical group [H]C([H])([H])O* 0.000 description 1
- IYICSMKIBSQCKK-UHFFFAOYSA-N methoxy(methyl)silane Chemical class CO[SiH2]C IYICSMKIBSQCKK-UHFFFAOYSA-N 0.000 description 1
- POPACFLNWGUDSR-UHFFFAOYSA-N methoxy(trimethyl)silane Chemical compound CO[Si](C)(C)C POPACFLNWGUDSR-UHFFFAOYSA-N 0.000 description 1
- 229920000609 methyl cellulose Polymers 0.000 description 1
- 239000005055 methyl trichlorosilane Substances 0.000 description 1
- 239000001923 methylcellulose Substances 0.000 description 1
- 235000010981 methylcellulose Nutrition 0.000 description 1
- JLUFWMXJHAVVNN-UHFFFAOYSA-N methyltrichlorosilane Chemical compound C[Si](Cl)(Cl)Cl JLUFWMXJHAVVNN-UHFFFAOYSA-N 0.000 description 1
- BFXIKLCIZHOAAZ-UHFFFAOYSA-N methyltrimethoxysilane Chemical compound CO[Si](C)(OC)OC BFXIKLCIZHOAAZ-UHFFFAOYSA-N 0.000 description 1
- 239000010445 mica Substances 0.000 description 1
- 229910052618 mica group Inorganic materials 0.000 description 1
- 230000003641 microbiacidal effect Effects 0.000 description 1
- 230000002906 microbiologic effect Effects 0.000 description 1
- 238000001000 micrograph Methods 0.000 description 1
- 230000005012 migration Effects 0.000 description 1
- 238000013508 migration Methods 0.000 description 1
- 230000003278 mimic effect Effects 0.000 description 1
- 239000002480 mineral oil Substances 0.000 description 1
- 235000010446 mineral oil Nutrition 0.000 description 1
- 230000004048 modification Effects 0.000 description 1
- 238000012986 modification Methods 0.000 description 1
- 229910052750 molybdenum Inorganic materials 0.000 description 1
- 239000011733 molybdenum Substances 0.000 description 1
- 125000000896 monocarboxylic acid group Chemical group 0.000 description 1
- 238000010068 moulding (rubber) Methods 0.000 description 1
- WKWOFMSUGVVZIV-UHFFFAOYSA-N n-bis(ethenyl)silyl-n-trimethylsilylmethanamine Chemical compound C[Si](C)(C)N(C)[SiH](C=C)C=C WKWOFMSUGVVZIV-UHFFFAOYSA-N 0.000 description 1
- 125000004108 n-butyl group Chemical group [H]C([H])([H])C([H])([H])C([H])([H])C([H])([H])* 0.000 description 1
- 125000001280 n-hexyl group Chemical group C(CCCCC)* 0.000 description 1
- QNILTEGFHQSKFF-UHFFFAOYSA-N n-propan-2-ylprop-2-enamide Chemical compound CC(C)NC(=O)C=C QNILTEGFHQSKFF-UHFFFAOYSA-N 0.000 description 1
- 239000011234 nano-particulate material Substances 0.000 description 1
- 150000004767 nitrides Chemical class 0.000 description 1
- 229920001220 nitrocellulos Polymers 0.000 description 1
- 229940079938 nitrocellulose Drugs 0.000 description 1
- 239000012299 nitrogen atmosphere Substances 0.000 description 1
- 239000002736 nonionic surfactant Substances 0.000 description 1
- FTMKAMVLFVRZQX-UHFFFAOYSA-N octadecylphosphonic acid Chemical compound CCCCCCCCCCCCCCCCCCP(O)(O)=O FTMKAMVLFVRZQX-UHFFFAOYSA-N 0.000 description 1
- YTJSFYQNRXLOIC-UHFFFAOYSA-N octadecylsilane Chemical compound CCCCCCCCCCCCCCCCCC[SiH3] YTJSFYQNRXLOIC-UHFFFAOYSA-N 0.000 description 1
- MSRJTTSHWYDFIU-UHFFFAOYSA-N octyltriethoxysilane Chemical compound CCCCCCCC[Si](OCC)(OCC)OCC MSRJTTSHWYDFIU-UHFFFAOYSA-N 0.000 description 1
- 150000002894 organic compounds Chemical class 0.000 description 1
- 125000000962 organic group Chemical group 0.000 description 1
- 239000005416 organic matter Substances 0.000 description 1
- 150000001451 organic peroxides Chemical class 0.000 description 1
- NDLPOXTZKUMGOV-UHFFFAOYSA-N oxo(oxoferriooxy)iron hydrate Chemical compound O.O=[Fe]O[Fe]=O NDLPOXTZKUMGOV-UHFFFAOYSA-N 0.000 description 1
- 238000012856 packing Methods 0.000 description 1
- 238000010422 painting Methods 0.000 description 1
- 229910052763 palladium Inorganic materials 0.000 description 1
- KDLHZDBZIXYQEI-UHFFFAOYSA-N palladium Substances [Pd] KDLHZDBZIXYQEI-UHFFFAOYSA-N 0.000 description 1
- 239000011236 particulate material Substances 0.000 description 1
- 239000001814 pectin Substances 0.000 description 1
- 235000010987 pectin Nutrition 0.000 description 1
- 229920001277 pectin Polymers 0.000 description 1
- XYJRXVWERLGGKC-UHFFFAOYSA-D pentacalcium;hydroxide;triphosphate Chemical compound [OH-].[Ca+2].[Ca+2].[Ca+2].[Ca+2].[Ca+2].[O-]P([O-])([O-])=O.[O-]P([O-])([O-])=O.[O-]P([O-])([O-])=O XYJRXVWERLGGKC-UHFFFAOYSA-D 0.000 description 1
- RGSFGYAAUTVSQA-UHFFFAOYSA-N pentamethylene Natural products C1CCCC1 RGSFGYAAUTVSQA-UHFFFAOYSA-N 0.000 description 1
- 230000000737 periodic effect Effects 0.000 description 1
- 230000035699 permeability Effects 0.000 description 1
- 150000002978 peroxides Chemical class 0.000 description 1
- JRKICGRDRMAZLK-UHFFFAOYSA-L peroxydisulfate Chemical compound [O-]S(=O)(=O)OOS([O-])(=O)=O JRKICGRDRMAZLK-UHFFFAOYSA-L 0.000 description 1
- 125000001997 phenyl group Chemical group [H]C1=C([H])C([H])=C(*)C([H])=C1[H] 0.000 description 1
- NBIIXXVUZAFLBC-UHFFFAOYSA-K phosphate Chemical compound [O-]P([O-])([O-])=O NBIIXXVUZAFLBC-UHFFFAOYSA-K 0.000 description 1
- 239000010452 phosphate Substances 0.000 description 1
- 125000002467 phosphate group Chemical group [H]OP(=O)(O[H])O[*] 0.000 description 1
- ACVYVLVWPXVTIT-UHFFFAOYSA-N phosphinic acid Chemical group O[PH2]=O ACVYVLVWPXVTIT-UHFFFAOYSA-N 0.000 description 1
- ABLZXFCXXLZCGV-UHFFFAOYSA-N phosphonic acid group Chemical group P(O)(O)=O ABLZXFCXXLZCGV-UHFFFAOYSA-N 0.000 description 1
- 150000003013 phosphoric acid derivatives Chemical class 0.000 description 1
- 238000000016 photochemical curing Methods 0.000 description 1
- 230000000704 physical effect Effects 0.000 description 1
- 238000004375 physisorption Methods 0.000 description 1
- 239000011120 plywood Substances 0.000 description 1
- 229920001983 poloxamer Polymers 0.000 description 1
- 229920002493 poly(chlorotrifluoroethylene) Polymers 0.000 description 1
- 229920001483 poly(ethyl methacrylate) polymer Polymers 0.000 description 1
- 229920000212 poly(isobutyl acrylate) Polymers 0.000 description 1
- 229920000205 poly(isobutyl methacrylate) Polymers 0.000 description 1
- 229920001495 poly(sodium acrylate) polymer Polymers 0.000 description 1
- 229920002037 poly(vinyl butyral) polymer Polymers 0.000 description 1
- 229920001748 polybutylene Polymers 0.000 description 1
- 239000005023 polychlorotrifluoroethylene (PCTFE) polymer Substances 0.000 description 1
- 229920000867 polyelectrolyte Polymers 0.000 description 1
- 229920000129 polyhexylmethacrylate Polymers 0.000 description 1
- 229920001228 polyisocyanate Polymers 0.000 description 1
- 239000005056 polyisocyanate Substances 0.000 description 1
- 229920002959 polymer blend Polymers 0.000 description 1
- 229920006254 polymer film Polymers 0.000 description 1
- 229920000307 polymer substrate Polymers 0.000 description 1
- 239000003505 polymerization initiator Substances 0.000 description 1
- 229920005651 polypropylene glycol dimethacrylate Polymers 0.000 description 1
- 229920000909 polytetrahydrofuran Polymers 0.000 description 1
- 239000011118 polyvinyl acetate Substances 0.000 description 1
- 229920002689 polyvinyl acetate Polymers 0.000 description 1
- 229920002451 polyvinyl alcohol Polymers 0.000 description 1
- 235000019422 polyvinyl alcohol Nutrition 0.000 description 1
- 239000004800 polyvinyl chloride Substances 0.000 description 1
- 229920000915 polyvinyl chloride Polymers 0.000 description 1
- 229920000036 polyvinylpyrrolidone Polymers 0.000 description 1
- 239000001267 polyvinylpyrrolidone Substances 0.000 description 1
- 235000013855 polyvinylpyrrolidone Nutrition 0.000 description 1
- 239000011148 porous material Substances 0.000 description 1
- 229910021426 porous silicon Inorganic materials 0.000 description 1
- 238000001556 precipitation Methods 0.000 description 1
- XOFYZVNMUHMLCC-ZPOLXVRWSA-N prednisone Chemical compound O=C1C=C[C@]2(C)[C@H]3C(=O)C[C@](C)([C@@](CC4)(O)C(=O)CO)[C@@H]4[C@@H]3CCC2=C1 XOFYZVNMUHMLCC-ZPOLXVRWSA-N 0.000 description 1
- 230000037452 priming Effects 0.000 description 1
- 238000012545 processing Methods 0.000 description 1
- QQONPFPTGQHPMA-UHFFFAOYSA-N propylene Natural products CC=C QQONPFPTGQHPMA-UHFFFAOYSA-N 0.000 description 1
- 125000004805 propylene group Chemical group [H]C([H])([H])C([H])([*:1])C([H])([H])[*:2] 0.000 description 1
- 239000011253 protective coating Substances 0.000 description 1
- 230000001681 protective effect Effects 0.000 description 1
- 229960003351 prussian blue Drugs 0.000 description 1
- 239000013225 prussian blue Substances 0.000 description 1
- 239000008262 pumice Substances 0.000 description 1
- 230000001698 pyrogenic effect Effects 0.000 description 1
- 229910052903 pyrophyllite Inorganic materials 0.000 description 1
- 239000002516 radical scavenger Substances 0.000 description 1
- 150000003254 radicals Chemical class 0.000 description 1
- 239000011342 resin composition Substances 0.000 description 1
- 230000027756 respiratory electron transport chain Effects 0.000 description 1
- 230000026041 response to humidity Effects 0.000 description 1
- 230000035440 response to pH Effects 0.000 description 1
- MHOVAHRLVXNVSD-UHFFFAOYSA-N rhodium atom Chemical compound [Rh] MHOVAHRLVXNVSD-UHFFFAOYSA-N 0.000 description 1
- 235000009566 rice Nutrition 0.000 description 1
- 239000005060 rubber Substances 0.000 description 1
- 238000001878 scanning electron micrograph Methods 0.000 description 1
- 230000003678 scratch resistant effect Effects 0.000 description 1
- 238000005201 scrubbing Methods 0.000 description 1
- 125000000467 secondary amino group Chemical group [H]N([*:1])[*:2] 0.000 description 1
- 150000003346 selenoethers Chemical class 0.000 description 1
- 239000013545 self-assembled monolayer Substances 0.000 description 1
- FZHAPNGMFPVSLP-UHFFFAOYSA-N silanamine Chemical compound [SiH3]N FZHAPNGMFPVSLP-UHFFFAOYSA-N 0.000 description 1
- SCPYDCQAZCOKTP-UHFFFAOYSA-N silanol Chemical compound [SiH3]O SCPYDCQAZCOKTP-UHFFFAOYSA-N 0.000 description 1
- 125000005372 silanol group Chemical group 0.000 description 1
- 239000000741 silica gel Substances 0.000 description 1
- 229910002027 silica gel Inorganic materials 0.000 description 1
- RMAQACBXLXPBSY-UHFFFAOYSA-N silicic acid Chemical compound O[Si](O)(O)O RMAQACBXLXPBSY-UHFFFAOYSA-N 0.000 description 1
- 239000010944 silver (metal) Substances 0.000 description 1
- 125000003808 silyl group Chemical group [H][Si]([H])([H])[*] 0.000 description 1
- ADCFFIGZZVYRNB-UHFFFAOYSA-N silyl propanoate Chemical class CCC(=O)O[SiH3] ADCFFIGZZVYRNB-UHFFFAOYSA-N 0.000 description 1
- 239000000779 smoke Substances 0.000 description 1
- 235000010413 sodium alginate Nutrition 0.000 description 1
- 239000000661 sodium alginate Substances 0.000 description 1
- 229940005550 sodium alginate Drugs 0.000 description 1
- 235000019812 sodium carboxymethyl cellulose Nutrition 0.000 description 1
- 229920001027 sodium carboxymethylcellulose Polymers 0.000 description 1
- NNMHYFLPFNGQFZ-UHFFFAOYSA-M sodium polyacrylate Chemical compound [Na+].[O-]C(=O)C=C NNMHYFLPFNGQFZ-UHFFFAOYSA-M 0.000 description 1
- XNRNJIIJLOFJEK-UHFFFAOYSA-N sodium;1-oxidopyridine-2-thione Chemical compound [Na+].[O-]N1C=CC=CC1=S XNRNJIIJLOFJEK-UHFFFAOYSA-N 0.000 description 1
- 239000002689 soil Substances 0.000 description 1
- 238000001179 sorption measurement Methods 0.000 description 1
- 238000005507 spraying Methods 0.000 description 1
- 239000010935 stainless steel Substances 0.000 description 1
- 229910001220 stainless steel Inorganic materials 0.000 description 1
- 229920003179 starch-based polymer Polymers 0.000 description 1
- 239000004628 starch-based polymer Substances 0.000 description 1
- 230000003068 static effect Effects 0.000 description 1
- 125000004079 stearyl group Chemical group [H]C([*])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])[H] 0.000 description 1
- 230000000638 stimulation Effects 0.000 description 1
- 238000003756 stirring Methods 0.000 description 1
- 229910052682 stishovite Inorganic materials 0.000 description 1
- 238000013517 stratification Methods 0.000 description 1
- 125000000626 sulfinic acid group Chemical group 0.000 description 1
- 125000003375 sulfoxide group Chemical group 0.000 description 1
- 150000003467 sulfuric acid derivatives Chemical class 0.000 description 1
- QAOWNCQODCNURD-UHFFFAOYSA-N sulfuric acid group Chemical group S(O)(O)(=O)=O QAOWNCQODCNURD-UHFFFAOYSA-N 0.000 description 1
- 238000010557 suspension polymerization reaction Methods 0.000 description 1
- 239000000454 talc Substances 0.000 description 1
- 229910052623 talc Inorganic materials 0.000 description 1
- IROINLKCQGIITA-UHFFFAOYSA-N terbutryn Chemical compound CCNC1=NC(NC(C)(C)C)=NC(SC)=N1 IROINLKCQGIITA-UHFFFAOYSA-N 0.000 description 1
- WMOVHXAZOJBABW-UHFFFAOYSA-N tert-butyl acetate Chemical compound CC(=O)OC(C)(C)C WMOVHXAZOJBABW-UHFFFAOYSA-N 0.000 description 1
- YLQBMQCUIZJEEH-UHFFFAOYSA-N tetrahydrofuran Natural products C=1C=COC=1 YLQBMQCUIZJEEH-UHFFFAOYSA-N 0.000 description 1
- 239000004753 textile Substances 0.000 description 1
- 238000010257 thawing Methods 0.000 description 1
- 238000002076 thermal analysis method Methods 0.000 description 1
- 238000001757 thermogravimetry curve Methods 0.000 description 1
- 229920005992 thermoplastic resin Polymers 0.000 description 1
- 125000003396 thiol group Chemical group [H]S* 0.000 description 1
- 235000010487 tragacanth Nutrition 0.000 description 1
- 239000000196 tragacanth Substances 0.000 description 1
- 229940116362 tragacanth Drugs 0.000 description 1
- 230000009466 transformation Effects 0.000 description 1
- 230000007704 transition Effects 0.000 description 1
- 229910052723 transition metal Inorganic materials 0.000 description 1
- 229910000314 transition metal oxide Inorganic materials 0.000 description 1
- 150000003624 transition metals Chemical class 0.000 description 1
- GQIUQDDJKHLHTB-UHFFFAOYSA-N trichloro(ethenyl)silane Chemical compound Cl[Si](Cl)(Cl)C=C GQIUQDDJKHLHTB-UHFFFAOYSA-N 0.000 description 1
- PYJJCSYBSYXGQQ-UHFFFAOYSA-N trichloro(octadecyl)silane Chemical compound CCCCCCCCCCCCCCCCCC[Si](Cl)(Cl)Cl PYJJCSYBSYXGQQ-UHFFFAOYSA-N 0.000 description 1
- RCHUVCPBWWSUMC-UHFFFAOYSA-N trichloro(octyl)silane Chemical compound CCCCCCCC[Si](Cl)(Cl)Cl RCHUVCPBWWSUMC-UHFFFAOYSA-N 0.000 description 1
- 229910052905 tridymite Inorganic materials 0.000 description 1
- DENFJSAFJTVPJR-UHFFFAOYSA-N triethoxy(ethyl)silane Chemical compound CCO[Si](CC)(OCC)OCC DENFJSAFJTVPJR-UHFFFAOYSA-N 0.000 description 1
- CPUDPFPXCZDNGI-UHFFFAOYSA-N triethoxy(methyl)silane Chemical compound CCO[Si](C)(OCC)OCC CPUDPFPXCZDNGI-UHFFFAOYSA-N 0.000 description 1
- JCVQKRGIASEUKR-UHFFFAOYSA-N triethoxy(phenyl)silane Chemical compound CCO[Si](OCC)(OCC)C1=CC=CC=C1 JCVQKRGIASEUKR-UHFFFAOYSA-N 0.000 description 1
- NBXZNTLFQLUFES-UHFFFAOYSA-N triethoxy(propyl)silane Chemical compound CCC[Si](OCC)(OCC)OCC NBXZNTLFQLUFES-UHFFFAOYSA-N 0.000 description 1
- JXUKBNICSRJFAP-UHFFFAOYSA-N triethoxy-[3-(oxiran-2-ylmethoxy)propyl]silane Chemical compound CCO[Si](OCC)(OCC)CCCOCC1CO1 JXUKBNICSRJFAP-UHFFFAOYSA-N 0.000 description 1
- QQQSFSZALRVCSZ-UHFFFAOYSA-N triethoxysilane Chemical compound CCO[SiH](OCC)OCC QQQSFSZALRVCSZ-UHFFFAOYSA-N 0.000 description 1
- 125000004205 trifluoroethyl group Chemical group [H]C([H])(*)C(F)(F)F 0.000 description 1
- UWSYCPWEBZRZNJ-UHFFFAOYSA-N trimethoxy(2,4,4-trimethylpentyl)silane Chemical compound CO[Si](OC)(OC)CC(C)CC(C)(C)C UWSYCPWEBZRZNJ-UHFFFAOYSA-N 0.000 description 1
- NMEPHPOFYLLFTK-UHFFFAOYSA-N trimethoxy(octyl)silane Chemical compound CCCCCCCC[Si](OC)(OC)OC NMEPHPOFYLLFTK-UHFFFAOYSA-N 0.000 description 1
- HQYALQRYBUJWDH-UHFFFAOYSA-N trimethoxy(propyl)silane Chemical compound CCC[Si](OC)(OC)OC HQYALQRYBUJWDH-UHFFFAOYSA-N 0.000 description 1
- 239000005051 trimethylchlorosilane Substances 0.000 description 1
- QHUNJMXHQHHWQP-UHFFFAOYSA-N trimethylsilyl acetate Chemical compound CC(=O)O[Si](C)(C)C QHUNJMXHQHHWQP-UHFFFAOYSA-N 0.000 description 1
- GPRLSGONYQIRFK-MNYXATJNSA-N triton Chemical compound [3H+] GPRLSGONYQIRFK-MNYXATJNSA-N 0.000 description 1
- WFKWXMTUELFFGS-UHFFFAOYSA-N tungsten Chemical compound [W] WFKWXMTUELFFGS-UHFFFAOYSA-N 0.000 description 1
- 229910052721 tungsten Inorganic materials 0.000 description 1
- 239000010937 tungsten Substances 0.000 description 1
- 238000009281 ultraviolet germicidal irradiation Methods 0.000 description 1
- 229910052720 vanadium Inorganic materials 0.000 description 1
- 229910001935 vanadium oxide Inorganic materials 0.000 description 1
- 239000002966 varnish Substances 0.000 description 1
- 239000005050 vinyl trichlorosilane Substances 0.000 description 1
- UKRDPEFKFJNXQM-UHFFFAOYSA-N vinylsilane Chemical class [SiH3]C=C UKRDPEFKFJNXQM-UHFFFAOYSA-N 0.000 description 1
- 238000001429 visible spectrum Methods 0.000 description 1
- 230000000007 visual effect Effects 0.000 description 1
- 239000012855 volatile organic compound Substances 0.000 description 1
- 229920001285 xanthan gum Polymers 0.000 description 1
- 235000010493 xanthan gum Nutrition 0.000 description 1
- 239000000230 xanthan gum Substances 0.000 description 1
- 229940082509 xanthan gum Drugs 0.000 description 1
- 229910052724 xenon Inorganic materials 0.000 description 1
- FHNFHKCVQCLJFQ-UHFFFAOYSA-N xenon atom Chemical compound [Xe] FHNFHKCVQCLJFQ-UHFFFAOYSA-N 0.000 description 1
- 238000004383 yellowing Methods 0.000 description 1
- PIMBTRGLTHJJRV-UHFFFAOYSA-L zinc;2-methylprop-2-enoate Chemical compound [Zn+2].CC(=C)C([O-])=O.CC(=C)C([O-])=O PIMBTRGLTHJJRV-UHFFFAOYSA-L 0.000 description 1
- RNWHGQJWIACOKP-UHFFFAOYSA-N zinc;oxygen(2-) Chemical class [O-2].[Zn+2] RNWHGQJWIACOKP-UHFFFAOYSA-N 0.000 description 1
- XKMZOFXGLBYJLS-UHFFFAOYSA-L zinc;prop-2-enoate Chemical compound [Zn+2].[O-]C(=O)C=C.[O-]C(=O)C=C XKMZOFXGLBYJLS-UHFFFAOYSA-L 0.000 description 1
- 229910052726 zirconium Inorganic materials 0.000 description 1
- 229910000859 α-Fe Inorganic materials 0.000 description 1
Classifications
-
- C—CHEMISTRY; METALLURGY
- C09—DYES; PAINTS; POLISHES; NATURAL RESINS; ADHESIVES; COMPOSITIONS NOT OTHERWISE PROVIDED FOR; APPLICATIONS OF MATERIALS NOT OTHERWISE PROVIDED FOR
- C09D—COATING COMPOSITIONS, e.g. PAINTS, VARNISHES OR LACQUERS; FILLING PASTES; CHEMICAL PAINT OR INK REMOVERS; INKS; CORRECTING FLUIDS; WOODSTAINS; PASTES OR SOLIDS FOR COLOURING OR PRINTING; USE OF MATERIALS THEREFOR
- C09D5/00—Coating compositions, e.g. paints, varnishes or lacquers, characterised by their physical nature or the effects produced; Filling pastes
- C09D5/16—Antifouling paints; Underwater paints
- C09D5/1681—Antifouling coatings characterised by surface structure, e.g. for roughness effect giving superhydrophobic coatings or Lotus effect
-
- C—CHEMISTRY; METALLURGY
- C04—CEMENTS; CONCRETE; ARTIFICIAL STONE; CERAMICS; REFRACTORIES
- C04B—LIME, MAGNESIA; SLAG; CEMENTS; COMPOSITIONS THEREOF, e.g. MORTARS, CONCRETE OR LIKE BUILDING MATERIALS; ARTIFICIAL STONE; CERAMICS; REFRACTORIES; TREATMENT OF NATURAL STONE
- C04B41/00—After-treatment of mortars, concrete, artificial stone or ceramics; Treatment of natural stone
- C04B41/009—After-treatment of mortars, concrete, artificial stone or ceramics; Treatment of natural stone characterised by the material treated
-
- C—CHEMISTRY; METALLURGY
- C04—CEMENTS; CONCRETE; ARTIFICIAL STONE; CERAMICS; REFRACTORIES
- C04B—LIME, MAGNESIA; SLAG; CEMENTS; COMPOSITIONS THEREOF, e.g. MORTARS, CONCRETE OR LIKE BUILDING MATERIALS; ARTIFICIAL STONE; CERAMICS; REFRACTORIES; TREATMENT OF NATURAL STONE
- C04B41/00—After-treatment of mortars, concrete, artificial stone or ceramics; Treatment of natural stone
- C04B41/45—Coating or impregnating, e.g. injection in masonry, partial coating of green or fired ceramics, organic coating compositions for adhering together two concrete elements
- C04B41/4584—Coating or impregnating of particulate or fibrous ceramic material
-
- C—CHEMISTRY; METALLURGY
- C04—CEMENTS; CONCRETE; ARTIFICIAL STONE; CERAMICS; REFRACTORIES
- C04B—LIME, MAGNESIA; SLAG; CEMENTS; COMPOSITIONS THEREOF, e.g. MORTARS, CONCRETE OR LIKE BUILDING MATERIALS; ARTIFICIAL STONE; CERAMICS; REFRACTORIES; TREATMENT OF NATURAL STONE
- C04B41/00—After-treatment of mortars, concrete, artificial stone or ceramics; Treatment of natural stone
- C04B41/45—Coating or impregnating, e.g. injection in masonry, partial coating of green or fired ceramics, organic coating compositions for adhering together two concrete elements
- C04B41/46—Coating or impregnating, e.g. injection in masonry, partial coating of green or fired ceramics, organic coating compositions for adhering together two concrete elements with organic materials
- C04B41/48—Macromolecular compounds
- C04B41/483—Polyacrylates
-
- C—CHEMISTRY; METALLURGY
- C04—CEMENTS; CONCRETE; ARTIFICIAL STONE; CERAMICS; REFRACTORIES
- C04B—LIME, MAGNESIA; SLAG; CEMENTS; COMPOSITIONS THEREOF, e.g. MORTARS, CONCRETE OR LIKE BUILDING MATERIALS; ARTIFICIAL STONE; CERAMICS; REFRACTORIES; TREATMENT OF NATURAL STONE
- C04B41/00—After-treatment of mortars, concrete, artificial stone or ceramics; Treatment of natural stone
- C04B41/60—After-treatment of mortars, concrete, artificial stone or ceramics; Treatment of natural stone of only artificial stone
- C04B41/61—Coating or impregnation
- C04B41/62—Coating or impregnation with organic materials
- C04B41/63—Macromolecular compounds
-
- C—CHEMISTRY; METALLURGY
- C09—DYES; PAINTS; POLISHES; NATURAL RESINS; ADHESIVES; COMPOSITIONS NOT OTHERWISE PROVIDED FOR; APPLICATIONS OF MATERIALS NOT OTHERWISE PROVIDED FOR
- C09D—COATING COMPOSITIONS, e.g. PAINTS, VARNISHES OR LACQUERS; FILLING PASTES; CHEMICAL PAINT OR INK REMOVERS; INKS; CORRECTING FLUIDS; WOODSTAINS; PASTES OR SOLIDS FOR COLOURING OR PRINTING; USE OF MATERIALS THEREFOR
- C09D133/00—Coating compositions based on homopolymers or copolymers of compounds having one or more unsaturated aliphatic radicals, each having only one carbon-to-carbon double bond, and at least one being terminated by only one carboxyl radical, or of salts, anhydrides, esters, amides, imides, or nitriles thereof; Coating compositions based on derivatives of such polymers
- C09D133/04—Homopolymers or copolymers of esters
- C09D133/06—Homopolymers or copolymers of esters of esters containing only carbon, hydrogen and oxygen, the oxygen atom being present only as part of the carboxyl radical
-
- C—CHEMISTRY; METALLURGY
- C09—DYES; PAINTS; POLISHES; NATURAL RESINS; ADHESIVES; COMPOSITIONS NOT OTHERWISE PROVIDED FOR; APPLICATIONS OF MATERIALS NOT OTHERWISE PROVIDED FOR
- C09D—COATING COMPOSITIONS, e.g. PAINTS, VARNISHES OR LACQUERS; FILLING PASTES; CHEMICAL PAINT OR INK REMOVERS; INKS; CORRECTING FLUIDS; WOODSTAINS; PASTES OR SOLIDS FOR COLOURING OR PRINTING; USE OF MATERIALS THEREFOR
- C09D133/00—Coating compositions based on homopolymers or copolymers of compounds having one or more unsaturated aliphatic radicals, each having only one carbon-to-carbon double bond, and at least one being terminated by only one carboxyl radical, or of salts, anhydrides, esters, amides, imides, or nitriles thereof; Coating compositions based on derivatives of such polymers
- C09D133/04—Homopolymers or copolymers of esters
- C09D133/14—Homopolymers or copolymers of esters of esters containing halogen, nitrogen, sulfur or oxygen atoms in addition to the carboxy oxygen
-
- C—CHEMISTRY; METALLURGY
- C09—DYES; PAINTS; POLISHES; NATURAL RESINS; ADHESIVES; COMPOSITIONS NOT OTHERWISE PROVIDED FOR; APPLICATIONS OF MATERIALS NOT OTHERWISE PROVIDED FOR
- C09D—COATING COMPOSITIONS, e.g. PAINTS, VARNISHES OR LACQUERS; FILLING PASTES; CHEMICAL PAINT OR INK REMOVERS; INKS; CORRECTING FLUIDS; WOODSTAINS; PASTES OR SOLIDS FOR COLOURING OR PRINTING; USE OF MATERIALS THEREFOR
- C09D133/00—Coating compositions based on homopolymers or copolymers of compounds having one or more unsaturated aliphatic radicals, each having only one carbon-to-carbon double bond, and at least one being terminated by only one carboxyl radical, or of salts, anhydrides, esters, amides, imides, or nitriles thereof; Coating compositions based on derivatives of such polymers
- C09D133/04—Homopolymers or copolymers of esters
- C09D133/14—Homopolymers or copolymers of esters of esters containing halogen, nitrogen, sulfur or oxygen atoms in addition to the carboxy oxygen
- C09D133/16—Homopolymers or copolymers of esters containing halogen atoms
-
- C—CHEMISTRY; METALLURGY
- C09—DYES; PAINTS; POLISHES; NATURAL RESINS; ADHESIVES; COMPOSITIONS NOT OTHERWISE PROVIDED FOR; APPLICATIONS OF MATERIALS NOT OTHERWISE PROVIDED FOR
- C09D—COATING COMPOSITIONS, e.g. PAINTS, VARNISHES OR LACQUERS; FILLING PASTES; CHEMICAL PAINT OR INK REMOVERS; INKS; CORRECTING FLUIDS; WOODSTAINS; PASTES OR SOLIDS FOR COLOURING OR PRINTING; USE OF MATERIALS THEREFOR
- C09D5/00—Coating compositions, e.g. paints, varnishes or lacquers, characterised by their physical nature or the effects produced; Filling pastes
-
- C—CHEMISTRY; METALLURGY
- C09—DYES; PAINTS; POLISHES; NATURAL RESINS; ADHESIVES; COMPOSITIONS NOT OTHERWISE PROVIDED FOR; APPLICATIONS OF MATERIALS NOT OTHERWISE PROVIDED FOR
- C09D—COATING COMPOSITIONS, e.g. PAINTS, VARNISHES OR LACQUERS; FILLING PASTES; CHEMICAL PAINT OR INK REMOVERS; INKS; CORRECTING FLUIDS; WOODSTAINS; PASTES OR SOLIDS FOR COLOURING OR PRINTING; USE OF MATERIALS THEREFOR
- C09D5/00—Coating compositions, e.g. paints, varnishes or lacquers, characterised by their physical nature or the effects produced; Filling pastes
- C09D5/02—Emulsion paints including aerosols
- C09D5/024—Emulsion paints including aerosols characterised by the additives
-
- C—CHEMISTRY; METALLURGY
- C09—DYES; PAINTS; POLISHES; NATURAL RESINS; ADHESIVES; COMPOSITIONS NOT OTHERWISE PROVIDED FOR; APPLICATIONS OF MATERIALS NOT OTHERWISE PROVIDED FOR
- C09D—COATING COMPOSITIONS, e.g. PAINTS, VARNISHES OR LACQUERS; FILLING PASTES; CHEMICAL PAINT OR INK REMOVERS; INKS; CORRECTING FLUIDS; WOODSTAINS; PASTES OR SOLIDS FOR COLOURING OR PRINTING; USE OF MATERIALS THEREFOR
- C09D5/00—Coating compositions, e.g. paints, varnishes or lacquers, characterised by their physical nature or the effects produced; Filling pastes
- C09D5/14—Paints containing biocides, e.g. fungicides, insecticides or pesticides
-
- B—PERFORMING OPERATIONS; TRANSPORTING
- B05—SPRAYING OR ATOMISING IN GENERAL; APPLYING FLUENT MATERIALS TO SURFACES, IN GENERAL
- B05D—PROCESSES FOR APPLYING FLUENT MATERIALS TO SURFACES, IN GENERAL
- B05D5/00—Processes for applying liquids or other fluent materials to surfaces to obtain special surface effects, finishes or structures
- B05D5/04—Processes for applying liquids or other fluent materials to surfaces to obtain special surface effects, finishes or structures to obtain a surface receptive to ink or other liquid
-
- B—PERFORMING OPERATIONS; TRANSPORTING
- B05—SPRAYING OR ATOMISING IN GENERAL; APPLYING FLUENT MATERIALS TO SURFACES, IN GENERAL
- B05D—PROCESSES FOR APPLYING FLUENT MATERIALS TO SURFACES, IN GENERAL
- B05D5/00—Processes for applying liquids or other fluent materials to surfaces to obtain special surface effects, finishes or structures
- B05D5/08—Processes for applying liquids or other fluent materials to surfaces to obtain special surface effects, finishes or structures to obtain an anti-friction or anti-adhesive surface
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08F—MACROMOLECULAR COMPOUNDS OBTAINED BY REACTIONS ONLY INVOLVING CARBON-TO-CARBON UNSATURATED BONDS
- C08F220/00—Copolymers of compounds having one or more unsaturated aliphatic radicals, each having only one carbon-to-carbon double bond, and only one being terminated by only one carboxyl radical or a salt, anhydride ester, amide, imide or nitrile thereof
- C08F220/02—Monocarboxylic acids having less than ten carbon atoms; Derivatives thereof
- C08F220/10—Esters
- C08F220/12—Esters of monohydric alcohols or phenols
- C08F220/16—Esters of monohydric alcohols or phenols of phenols or of alcohols containing two or more carbon atoms
- C08F220/18—Esters of monohydric alcohols or phenols of phenols or of alcohols containing two or more carbon atoms with acrylic or methacrylic acids
- C08F220/1804—C4-(meth)acrylate, e.g. butyl (meth)acrylate, isobutyl (meth)acrylate or tert-butyl (meth)acrylate
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08K—Use of inorganic or non-macromolecular organic substances as compounding ingredients
- C08K3/00—Use of inorganic substances as compounding ingredients
- C08K3/18—Oxygen-containing compounds, e.g. metal carbonyls
- C08K3/20—Oxides; Hydroxides
- C08K3/22—Oxides; Hydroxides of metals
- C08K2003/2237—Oxides; Hydroxides of metals of titanium
- C08K2003/2241—Titanium dioxide
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08L—COMPOSITIONS OF MACROMOLECULAR COMPOUNDS
- C08L2201/00—Properties
- C08L2201/50—Aqueous dispersion, e.g. containing polymers with a glass transition temperature (Tg) above 20°C
Definitions
- the present invention particularly relates to stimuli responsive self-cleaning aqueous coating compositions for masonry and other substrates involving coatings that are durable, storage stable, ambient curing and recoatable comprising of smart polymers, and more particularly, relates to said coatings exhibiting reversible super-hydrophobic and super-hydrophi!ic behaviour in response to external stimuli based variable environmental conditions such as variations in at least one temperature, pH, humidity and light; also exhibiting low contact angle hysteresis of ⁇ 25 degrees which reversibly switches to a contact angle hysteresis of 25 degrees with external stimuli.
- aqueous coating composition Comprising a polymeric dispersion of either acrylic, fluorinated and amine backbone or acrylic, fluorinated and acid backbone in combination with hydrophobic and hydrophilic silicones and at least partially surface treated particles also including particulate mixture of micro and nano particles having balance of hydrophobicity and hydrophilicity, advantageously facilitates the attainment of contact angle hysteresis of ⁇ 25 degrees by the said self cleaning coating composition.
- said attainment of such low contact angle hysteresis ( ⁇ 25 degrees) and its subsequent reversible wettability switching by the aqueous coating composition/ stimuli responsive self-cleaning coatings enables the flow of rain water or forced air/ wind to clean the dirt from said coating surface
- said coating can be formed by simple mixing of the components and applied by simple techniques such as brush, spray and dip coating application.
- the coating exhibits good dirt pickup resistance and resistance to dirt lines, streaks and water spots and also exhibits good anti microbial resistance to microorganisms, particularly algae.
- the coating displays enhanced photocatalytic activity under diffused sunlight with reduced chalking and shows good resistance to organic dirt and deposits such as bird droppings and oily stains.
- The- coating also has a reduced; tendency for spreading of water containing rust.
- the surface of exterior coatings on exposure to outdoor environments is contaminated by organic and inorganic dust, clay particles, smoke, exhausts gas, rust, bird droppings and discoloured by ultraviolet rays.
- the wetting of surfaces with water results in the retention of water droplets on the surface and their evaporation, with the solids suspended or dissolved in the water remaining as unsightly residues (water marks, smears or spots) on the surface.
- the wetting of a surface with water is frequently also a trigger for its degradation or for infestation with microorganisms and the growth of algae, fungi, lichen, mosses, bacteria etc. Therefore, it is particularly required for such outdoor coatings to show physical properties such as self-cleaning, weather resistance and anti microbial properties with long-term reliability.
- Wettability of the surface can be measured by the contact angle which depends on the surface material and the surface texture.
- the wettability of a surface can be represented by the roll-off angle or Contact angle hysteresis.
- the contact angle ⁇ is the : angle at which a liquid/vapor interface meets the solid surface wherein Fig 1 reveals contact angle measurement showing a hydrophobic and hydrophilic drop *
- Hydrophilic When the contact angle is ⁇ 90° the surface is said to be hydrophilic.
- Oleophobic When the contact angle is >90° with oil/ oil like substance, the surface is said to be Oleophobic.
- CAH Contact angle hysteresis
- Figure 2 reveals contact angle measurement showing the advancing and the receding angle wherein the Advancing angle: The contact angle which is a result of increasing volume of water on a surface is called the advancing angle and wherein the Receding angle: When water with a syringe is removed from a surface the contact angle is called the receding angle.
- hydrophilic liquids may be reduced by a hydrophobic coating of the surface. Furthermore, it has proven favorable to structure hydrophobic surfaces.
- the structuring reduces the adhesion of the surface for polar liquids such as water and leads to reduced adhesion of solid deposits such as dirt particles on the surface. Given appropriate structuring, the dirt particles are flushed from the surface by moving water. The particulate solids may also be removed very easily by means of forced air or wind.
- U.S. 2002/0048679 describes surfaces having a smooth, extremely hydrophobic polymer (for example, polytetrafluoroethylene) film and surfaces having a smooth extremely hydrophilic polymer film as examples of surfaces on which water and dirt can run off without forming droplets.
- U.S. 2002/0048679 further describes how a long-term hydrophobic coating may be formed by applying certain silane derivatives underneath a hydrophobic coating on a surface.
- Other self-cleaning surfaces are described in U.S. Patent Application numbers US 2002/0150723, US 2002/0150724, US 2002/0150725, US 2002/0150726, US 2003/0013795 and US 2003/0147932.
- Oil and water repellent compositions based on fluoropolymers are described by Linert, et al, in WO 199700230, describe a composition comprising a fluoroaliphatic groups, carboxyl containing groups, oxyalkene groups and optionally silyl groups which provide repellency to oil- and water-based stains for porous substrates.
- Random fluorinated copolymers prepared by radical copolymerisation of monomers in solution in a water-miscible organic solvent using peroxides or azo compounds as initiators have been described (see, for example, EP 542598, EP 1106630 and US 2004026053), together with their hydrophobic and oleophobic properties on various substrates.
- US 7781027 teaches durable, weatherable and scratch -resistant hydrophobic coatings comprising a fluorinated component and an adhesion promoter compound.
- the adhesion promoter compound is said to include an alkoxy group, a furfuryl-containing ring structure, and a reactive group.
- US 4859754 teaches a water and oil repellent polymer having desoiling properties composed of a polyfiuorinated group-containing copolymer obtained by copolymerizing high levels (65-95%) of a polyfiuorinated group-containing monomer and an amphipathic monomer (5-35%) having a hydrophilic moiety and a lipophilic moiety.
- US 20110111659 teaches a method of providing oil repellency, water repellency, and dynamic water repellency to a substrate by coating with a polymer composition comprising hydrophobized nanoparticles and one or more monomers of Ci. to .
- Ci8 straight, branched, or cyclic alkyl (meth)acrylate> one or more monomers selected from the group consisting of N- methylol (meth)acrylamide and a second polymer comprising high quantities of fluorinated monomer.
- EP2210921 teaches a hydrophobic composite coating which comprises a plurality of carbon nanotubes dispersed in a hydrophobic polymer resulting in a coating which has a water contact angle of least about 120°.
- Super-hydrophobicity is frequently also referred to as the "lotus effect" because it was first observed on the leaves of the lotus plant.
- Super-hydrophobicity of a surface results in particularly easy dripping or rolling off of water from the surface, which supports fast drying of the surface and hampers the adhesion of dirt to the surface.
- wenzel state the drop of fluid completely covers the surface.
- hydrophobicity of a hydrophobic surface rises.
- contact angle of a hydrophilic surface decreases with increasing roughness.
- a hydrophobic surface can be turned into a super-hydrophobic surface, and a hydrophilic surface can be turned into a super-hydrophilic surface by increasing the surface roughness.
- the Cassie- Baxter state furthermore considers that air bubbles are trapped between the drop and the rough surface.
- the leaves of the lotus plant have elevations made from a wax, and these elevations lower the contact area with water.
- WO 00/58410 describes these structures and claims the formation of the same by spray-application of hydrophobic alcohols, such as 10- nonacosanol, or of alkanediols, such as 5, 10-nonacosanediol.
- hydrophobic alcohols such as 10- nonacosanol
- alkanediols such as 5, 10-nonacosanediol.
- the separations of the elevations in the structures are in the range from 0.1 to 200 pm and the heights of the elevations are from 0.1 to 100 Mm.
- US 7544411 teaches a self-cleaning paint coating that exhibits the "Lotus Effect”.
- the paint coating comprises a paint base coat and a topcoat.
- the topcoat includes structure-forming particles and is at least partially hydrophobic.
- the structure-forming particles have an average particle diameter of less than 100 nanometers (nm) and form elevations in the topcoat surface that are interspaced at a distance of less than 50 nm.
- the topcoat of the paint coating is formed from the agent which contains a liquid medium, a hydrophobating agent dissolved in the liquid medium, and structure-forming particles suspended in the medium.
- US7196043 targets a composition for producing a self-cleaning coating on a surface, the composition comprising: an aqueous mixture comprising water, metaJ oxide nanoparticles present at between 0.001 percent to 10 percent by weight of the mixture and having a particle size- of less than 300 nanometers and a fluorinated water-soluble hydrophobic surface modifier of between 0.001 percent and 10 percent by weight of the mixture wherein the surface modifier can produce an unstructured surface having a surface energy below 30 dynes per centimeter, which fluorinated water-soluble hydrophobic surface includes fluorinated acrylic polymers.
- CH-PS-26 82 58 describes water-repellent surfaces which exhibit a contact angle with water of more than 120°.
- US 4061503 teaches a composition comprising titanium dioxide particles containing on their surfaces a silane, in an amount sufficient to improve the dispersibility of said particles in a resin or plastic medium.
- hydrophobic film or coating comprising primary particles covalently bonded with secondary particles, adhering to the surface of the primary particles and having an average diameter that is smaller than the average diameter of the primary particles, and a hydrophobic layer covering at least partly the surface of the secondary particles and adhering to that surface.
- US8202614 discloses an additive particle comprising a carrier microparticle, a plurality of nanoparticles adhered to and substantially covering a surface of the microparticle, and a hydrophobic surface treatment material in contact with exposed surfaces of the carrier microparticle and/or the nanoparticles sufficient to impart a superhydrophobic contact angle to the additive particle of greater than 150°; wherein the hydrophobic surface treatment material comprises a perfluorinated alkyl silane.
- US8147607, US7964244 and US20050118433 relates to a hydrophobic coating composition for forming a coating on a substrate, consisting of hydrophobic particles, a solvent and water.
- the hydrophobic particles comprise one or more metal oxides that are treated with an organosilane, alkylsilane, fluorinated silane, and/or disilazane.
- US6683126 provides a coating composition for producing difficult-to-wet surfaces, comprising at least one finely divided powder whose particles have a hydrophobic surface and a porous structure characterized by a BEET surface area of at least 1 m 2 /g, and at least one film-forming binder characterized by a surface tension ⁇ 50 mN/m, the weight ratio of powder to binder being at least 1 :4.
- a hydrophilic surface may also produce good dirt shedding.
- the hydrophilic coating resists dirt pickup since water can wet the surface and flow off more easily allowing dirt to be easily washed off by rainwater.
- US20060058490 claims a hydrophilic coating (contact angle ⁇ 30°) formed using a hydrophilic silane and mixture of hydrophilic particles. They mention that if an organic binder is used, it may undergo degradation if photo catalytic particles are present in their system. Solgel network formation leads to a coating film with a hydrophilic surface where dirt streak marks can be avoided.
- US20040082494 teaches hydrophobic-hydrophilic block copolymers to increase the water affinity of low energy surfaces using high contents of hydrophilic monomers (0 to 95%).
- Ti0 2 Under UV irradiation Ti0 2 (Titanium dioxide) is photocatalytically active and can produce super-wetting effects as a result of water hydrolysis effects.
- US6048910 teaches a coating composition comprising of an emulsion of a silicone resin, typically a water-insoluble, silanol group-bearing silicone resin dispersed in water, and contains photocatalyst particles, typically titanium oxide which forms a hydrophilic film having a water-drop contact angle of no more than 60° when irradiated with UV-containing light. Films obtained by applying this coating composition to the surface of substrates have excellent weathering resistance and antifouling properties due to a self-cleaning effect arising from the photocatalytic action of the particles.
- titanium dioxide exhibit photocatalytic activity, of which, only anatase and rutile have potential commercial applicability as photocatalysts.
- Rutile titanium dioxide has band gap energy of 3.05 eV, which corresponds to approximately 420 nm wavelength of light, and anatase titanium dioxide has a band gap of 3.2 eV (approximately 385 nm).
- anatase titanium dioxide has established a reputation as generally superior to rutile titanium dioxide.
- a titanium dioxide crystal absorbs a photon of sufficient energy (from natural radiation, sun or from an artificial radiation exposure, lamp), an electron is promoted to the conduction band, and a positive hole (h+) is generated in the valence band.
- This "electron- hole pair" generates highly oxidizing hydroxyl and superoxide radicals at the crystal surface that are capable of oxidizing practically all organic matter to C0 2 and H 2 0.
- Photocata lytic oxides are known to have antifouling, antimicrobial, soil-degrading, deodorizing, air-cleaning, antifouling, antimicrobial, water-cleaning, superhydrophilicizing and/or antifogging effects. Due to photocata lysis the organic dirt even when deposited on the surface of the coating, are unstable and hence easily removed, permitting the surface to be kept clean.
- US6337129 teaches an antifouling coating composition with both a hydrophobic portion, and a hydrophilic portion induced by a photocatalyst, both portions being present in a microscopically dispersed and exposed state on the surface.
- the content of the photocata lytic oxide in the surface layer is also not particularly limited.
- the content of the photocatalytic oxide in the surface layer is preferably about 10 to 80% by weight, more preferably about 20 to 50% by weight. Thus high contents of photocatalysts are being used.
- US7521039 describe photocatalytically active rutile titanium dioxide which demonstrates enhanced activity in the visible light spectrum. It is produced by neutralizing the rutile seed to a high pH prior to washing and thermally treating the seed.
- US7955430 teaches a coating material comprising a binding agent and at least one filler including particles having a size and/or surface roughness of about 100 microns or less, and a photocatalytically active agent about 2 to about 15 weight percent of the coating material.
- the binding agent may be at least partially decomposed by a photocatalytic action, and a microstructured, self-cleaning surface may be formed.
- the photocatalytically active agent used is an oxide of titanium, zinc, iron, manganese, molybdenum and/or tungsten, which comprises at least one additive selected from C, N, S and/or from a transition metal oxide and/or transition metal halide.
- the selected additive(s) may enable the stimulation of the catalytic property even with wave lengths e.g. in the visible range of the solar spectrum.
- the desired self-cleaning properties may advantageously be achieved if the coating material is formulated using an excess of binding agent.
- coating materials for plastics based on inorganic binding agents and fillers are described.
- the coating materials set forth in the known publication may additionally contain photocatalytically active agents for self-cleaning by means of decomposition of organic soiling and superhydrophilics, while the binding agents used in the manufacture of the known materials are stable to the photocata lytic effect of the photocatalytically active agents.
- US20100311572 discloses a coating material comprising at least one binder and at least one photocatalytically active particle comprising a core composed of at least one first substance having a diameter of 0.1 to 1 pm and at least one envelope at least partly surrounding the core and composed of at least one second substance having an average layer thickness of 0.1 to 10 nm.
- the active particles are generally present in the coating material in an amount ranging from 0.1% to 10% by weight.
- Useful metals or semimetals whose oxides are present in the core of the photocatalytically active particles are generally selected from the group consisting of elements of groups I to XV of the periodic table (in accordance with IUPAC), lanthanides, actinides and mixtures thereof, more preferably from the group consisting of V, Ti, Zr, Ce, Mo, Bi, Zn, Mn, Si, Ba, Au, Ag, Pd, Pt, u, Rh, La and mixtures thereof.
- Very particularly preferred metal or semimetal oxides present in the at least one envelope of the particles are Si0 2 , ZnO, Ce0 2 , Ti0 2 SnO or mixtures thereof.
- JP 11 181339 A discloses a hydrophilic coating comprising photocatalytically active titanium' dioxide particles.
- the titanium dioxide particles have a particle diameter of 1 to 100 nm, comprise particles of Sn0 2 and are silica and/or silicone coated.
- the coating material prevents soiling of a surface coated with this material.
- a surface thus treated further has self-cleaning properties when rained upon.
- JP 2006-233343 discloses a liquid photocatalyst composition for coating textiles which comprise photocatalyst particles having a preset particle diameter and a coating. The size of these photocatalyst particles is 0.5 to 10 pm.
- the core of the photocatalyst particle consists of titanium dioxide and the envelope consists of silica.
- JP 11228873 A discloses a paint composition comprising titanium dioxide particles coated with porous silica and having photocatalytic properties.
- the composition further comprises titanium dioxide pigment and an organic resin binder and does not teach any particle size of said photocatalytically active particles.
- US20110129204 discloses a photocatalytic coating composition
- a photocatalytic coating composition comprising a hydroxyapatite-coated photocatalyst, a silica-based binder, an acrylic emulsion, a thickener and water.
- US 20110313095 relates to a mixture, comprising at least one thermoplastic polymer as component (A) and at least one photocatalytically active particle, comprising a non-porous core comprising at least one metal oxide or semimetal oxide with a diameter of from 0.1 nm to l.mu.m, and, at least to some extent surrounding the core, at least one porous outer layer comprising at least one further metal oxide or further semimetal oxide with an average layer thickness of from 0.1 to 10 nm, as component (B), to a process for the production of this mixture, via mixing of components (A) and (B), to the use of the mixture as photocatalytically active surface, to moldings, comprising this mixture, and to the use of this mixture for the production of moldings.
- a mixture comprising at least one thermoplastic polymer as component (A) and at least one photocatalytically active particle, comprising a non-porous core comprising at least one metal oxide or semimetal oxide with a diameter of from 0.1
- EP1955767 Al discloses a mixture comprising photocatalysts and organic polymers, such as polyacetals, polyethylene, polypropylene ' , polystyrene, polyvinyl chloride, nylon-type polymers, polycarbonates, etc.
- the photocatalysts used preferably comprise titanium dioxide particles, which have a coating composed of non-porous silicon dioxide.
- JP2000-017096 discloses foamed foils composed of thermoplastic resins, which have titanium dioxide as antibacterial coating. Silanes have been applied as surface modifier on the surface of the titanium dioxide.
- JP2000-204194 discloses a process for the production of polymer compositions which comprise titanium dioxide as photocatalyst.
- the titanium dioxide has, for example, a coating of silicon dioxide.
- the ratio of polymer to titanium dioxide is from 97: 3 to 20:80.
- JP 09/225321 A discloses a photocatalytically active body composed of titanium dioxide in the anatase form.
- the titanium dioxide used in that composition is present between two porous layers of another inorganic compound, such as silicon dioxide.
- JP 2005-097608 A discloses a polyoiefin film which comprises photocatalyst particles, where the photocatalyst particles are mixed crystalline oxides comprising titanium dioxide and silicon dioxide.
- US20110312065 A teaches a substrate or coating that includes a protease which is capable of enzymatically degrading of one or more components of the biological stain to facilitate biological stain removal from the substrate or said coating.
- UV light refers to light of a wavelength below about 400 nm.
- light from the visible spectrum refers to light of a wavelength from about 400 nm to about 800 nm.
- photocatalytic paints comprise silicone type mineral binders and not binders based on organic products, so as to prevent the binder itself from being photocatalytically degraded. There is also a need to develop an economical photocatalytic coating composition that can minimize degradation of the organic binder.
- WO2007044784 teaches a self-decontaminating surface coating resistant to spores which when cured has a partially hydrophobic surface.
- the coating comprises a polymeric coating resin which, when cured, is hydrophilic.
- the coating also comprises a biocide, a germinating agent, and a hydrophobic micro/nano particulate material.
- the coating is suitable for application to ceramics, metals, and polymer substrates.
- Polymers that have anti-microbial properties are disclosed by the following patent applications: DE10024270, DE10022406, PCT/EP 00/06501, DE10014726, and DE 10008177.
- the antimicrobial properties are attributable to the contact of bacteria with ' the surface.
- European patent application EP0862858 discloses that copolymers of tert- butylaminoethyl methacrylate, a methacrylate with a secondary amino function have microbicidal properties.
- Acid functional polymers are pH responsive with the polymer becoming hydrophilic with pH>7 and hydrophobic with pH ⁇ 7. Polymers with sufficient hydrophilicity in their backbone can exhibit response to humidity with higher humidity conditions leading to water absorption and hence the surfaces behaving hydrophilic and vice versa.
- the most commonly utilised stimulus is temperature and polyacrylamide based copolymers are well known to create temperature-stimulus-sensitive surfaces as taught by Cheng, X.; Canavan, ⁇ . ⁇ .; Stein, M. J.; Hull, J. R.; Kweskin, S. J.; Wagner, M. S.; Somorjai, G. A.; Castner, D. G.; Ratner, B. D.
- LCST lower critical solution temperature
- PNiPAm poly-n-isopropylacrylamide
- LCST critical solution temperature
- PNiPAm poly-n-isopropylacrylamide
- Below the LCST PNiPAm expands and becomes hydrophilic (swollen coil conformation) while above the LCST it shrinks becoming hydrophobic (globular conformation) as disclosed in Benee, L. S.; Snowden, M. J., Chowdhry B. Z. Langmuir, 2002, 18, 6025.
- Copolymerization with hydrophobic monomers are known to decrease the LCST whereas with hydrophilic monomers increases the LCST.
- Aqueous polymeric dispersions are taught in US5173523, using a two stage monomer addition process followed by stripping of the co-solvent.
- US6277953 teaches preparation of an aqueous polymeric dispersion which is substantially free of an emulsifier, having an acid value in the range of from about 10 to about 40 with a particle size of not more than about 300 nm and the aqueous dispersion has less than about 2 weight percent organic solvent, at least about 30 weight percent solids.
- US4616058, US5817370 also teach the preparation of polymeric dispersions.
- US5319019 teaches a process for making acrylic polymer ammonium salt/organic solvent/water mixture, the organic solvent and water being in amounts effective for forming an azeotrope; resulting in a polymer having less than 2 weight percent of organic solvent.
- Responsive colloidal systems Reversible aggregation and fabrication of superhydrophobic surfaces by Mikhail Motornov, Journal of Colloid and Interface Science 310 (2007) 481-488, reported a method of fabricating stimuli-responsive core-shell nanoparticles using block copolymers of poly(styrene-/?-2-vinylpyridine-£>-ethylene oxide) and poly(styrene-£»-4- vinylpyridine) covalently bound to silica nanoparticles with two different diameters: colloidal silica 200 nm in diameter and fumed silica 15 nm in diameter.
- the S0 3 groups of the SDBS hydrogen bonds with the COOH groups on the bead, creating a hydrophobic surface state.
- the acid groups are deprotonated the COO- and the S0 3 - groups repel each other and provide a hydrophilic surface state.
- US7695814 and US7923106 are directed to a responsive coated substrate comprising at least one silicone-based, substantially hydrophobic polymer and at least one substantially hydrophilic polymer comprising polyethylenimine wherein said responsive coating substrate is in a first state; and methods of coating the same.
- US6919398 teaches a hydrophobic coating with a contact angle of greater than 130° after 3 minutes equilibrium consisting of a hydrophobic polymer and a bimodal particle size distribution of particles more than 5 microns particle size and less than 3 microns. They also teach about their formulation containing an insufficient amount of hydrophilic components.
- a paint described in DE2352242, particularly for protecting facades such as concrete facades, which paint contains pigment and as a binder copolymerisates of methacrylic acid esters has in the binder, in order to increase the water-repellent properties, a content of lower molecular weight methyl, phenyl or methylphenyl siloxanes in an amount of 10-50% by weight of the copolymerisate, and preferably also a catalyst for the siloxanes, e.g. an organotin compound.
- the siloxanes are stated to polymerise to form higher molecular weight compounds, which improves the water-repellency.
- a water-repellent coating composition consisting of an inert carrier liquid, a resin-like binder and one or more metal or metalloid oxides the surface of which has been rendered hydrophobic with an organic silicon compound.
- a coating formed by such a composition must be expected to be surface-hydrophobic and hence give a good water-repellency.
- US5584921 discloses coatings for building materials, in the form of an aqueous dispersion comprising: (a) 10 to 60 wt % of at least one polysiloxane (an aqueous emulsion of a hydroxyl-functional methyl silicone resin); (b) 5 to 65 wt % of at least one colloidal silica in the form of silica sol; (c) 5 to 80 wt % of at least one pigment or unreactive filler; and (d) 0 to 30 wt % of at least one further varnish or paint additive.
- aqueous dispersion comprising: (a) 10 to 60 wt % of at least one polysiloxane (an aqueous emulsion of a hydroxyl-functional methyl silicone resin); (b) 5 to 65 wt % of at least one colloidal silica in the form of silica sol; (c) 5 to 80 wt % of at least one pigment or unreactive filler
- Coatings containing silicates have been shown to provide superior stain resistance. Such coatings are described, for example in EP0942052, JP2002294154, and JP01172389. Organosilicates have also been used with fluoropolymer resins to produce desirable coating compositions as described in JP02003775, JP2003020450, U.S. Pat. No. 6,635,341 and EP1035184. An organosilicate works by a mechanism of stratification first, and then hydrolysis with rain water in the surface increasing the hydrophilicity. The book "Silicone resins and their combinations" Wernfried Heilen, Vincentz Network, 2005 teaches the use of silicone containing binders to enhance durability of architectural paints.
- the binder in silicone resin emulsion paints comprises 50 % mineral-based silicone resin and 50 .% polymer emulsion.
- silicone resin binders build up stable three-dimensional silicone resin networks which are chemically bound to the mineral substrate.
- the water repellency arises from the organic group on the silicone resin while the inorganic portion of the silicone resin bonds with the filler and the pigment together, creating permanently water-repellent capillaries and pores.
- US5759980 teaches a composition, which comprises a surfactant package consisting of a silicone-based surfactant and a polymer which is capable of bonding to a surface to make a hydrophilic film which eliminates the problem of water marks.
- this hydrophilic coating may tend to be removed from the surface by a single water rinse.
- German publication DE-A2161591 also describes a composition for cleaning cars wherein the surface is again made hydrophilic by using amino functional polymers. This coating also tends to be rinsed off from a single rinse.
- waxes and other products available in the market for attempting to retain this spot free finish.
- certain wax containing hydrophobic surfaces result in water marks when the water dries giving an unaesthetic appearance.
- US20060110542 discloses a composition for forming a detachable and renewable protective coating produced by making a highly concentrated dispersion of hydrophobically modified silica particles in the presence of a disilazane derivative under high shear conditions.
- Examples of commercially available materials which attempt to produce this "Lotus" cleaning effect are products sold under the trade name of MINCOR available from BASF, and TEGTOP available from Degussa. As stated in US20090018249 these products, when tested for their ability to protect various surfaces from the appearance of water marks, corrosion, and dirt repellency and while maintaining the water repellency, were deemed unsuitable.
- the coating is initially super hydrophobic and may remain so for long periods indoors; however, when exposed to outdoor UV light, rubbed even slightly, or in general exposed to weather, the coating loses super hydrophobicity and becomes less hydrophobic within days or even hydrophilic and hence less useful for the object of the present invention. Examination under the microscope after a week of exposure on a panel in a UV cabinet reveals that a coating made from fumed silica and at least one film forming binder as taught in US6683126 disintegrates.
- US20030013795 teaches a self-regenerating, self-cleaning hydrophobic surface formed when particles are secured on a carrier, resulting in a long lasting self cleaning action.
- WO2008071957 relates to novel block copolymers for use in coating surfaces to provide self-cleaning surfaces and capture/release materials wherein the copolymers comprises a first hydrophobic block and a second hydrophilic block comprising a non-fluorinated acrylate or methacrylate monomer. Although some of the above-mentioned surfaces may have excellent self-cleaning properties, the attachment or colonisation of microorganisms can impair these properties.
- micro-organisms fungi, algae, bacteria
- fungi, algae, bacteria may colonise a self-cleaning surface which may impair, or may entirely remove, the self-cleaning properties of the surface.
- organic dirt and deposits such as bird droppings and oily stains further mar the surface of these self cleaning coatings.
- Coatings containing photocata lytic particles use high contents of nano Titania or are doped with expensive materials to enhance activity under visible light which is not a feasible proposition.
- a further requirement of new generation smart surfaces in addition to the abovesaid, is the ability to dispose off the dirt deposited on the surface by a combination of hydrophobic and hydrophilic cleaning, to eventually not only prevent the deposition of the dirt but also prevent the infestation and growth of unwanted organisms, such as microorganisms, algae, lichen, and mosses.
- Such a substrate comprising substantially hydrophobic coating as abovesaid which when exposed to at least one external stimulus such as UV, pH, humidity or temperature would cause the substrate to switch from the first substantially hydrophobic state to a second substantially hydrophilic state, with a provision to produce such smart coatings by facile, economical and simple techniques for manufacturing on a large scale and applied by conventional techniques such as brush and spray application that would thus exhibit superhydrophobicity under certain environmental conditions of high temperature/ low pH/ low humidity/ low intensity of light and superhydrophilicity at low temperature/ high pH/ high humidity/ high intensity of light.
- Another object of the present invention is to provide for a stimuli responsive self-cleaning aqueous paint/ coating compositions and self-cleanable surfaces which would be oleophobic and exhibit the desired reversible hydrophilic-hydrophobic switching to provide for a water contact angle hysteresis of ⁇ 25° under certain environmental conditions with variations in at least one or more of temperature, humidity, pH, light that would be switchable to a water contact angle hysteresis of >25° under said environmental conditions offering benefits of both superhydrophobic and superhydrophilic cleaning, water marks, good organic and inorganic dirt pickup resistance, resistance to dirt streaks, dirt lines, water spotting, good cleanability with water and forced air or wind, reduced chalking, good resistance to organic dirt and deposits such as bird droppings and oily Stains, and reduced tendency for spreading of water containing rust.
- low contact angle hysteresis ⁇ 25 degrees, preferably ⁇ 10 degrees and more preferably ⁇ 5 degrees
- Still another object of the present invention is to provide a super hydrophobic coating, which would be so hydrophobic that water and even muddy water will bounce off the surface of nominally horizontal coated plates, and which would also be preferably styrene free to give a coating with reduced chalking behavior and high durability.
- Another object of the present invention is to provide said coatings that are practical in use, that do not degrade rapidly in sunlight and have enough resistance to abrasion to survive for a practical length of time, preferably for several months or even several years depending upon environmental conditions to which the coating is exposed, which would not lose said hydrophobic-hydrophilic switching properties even over a period of time. It is another object of the present invention to provide for said self-cleaning coating composition that cures by evaporation of the water and does not require any special treatment, such as heating or exposing to IR or UV light to cure.
- an aqueous coating composition exhibiting reversible hydrophilic-hydrophobic switching with the variations in temperature, humidity, pH and or light comprising:
- oligomeric or polymeric dispersion/binder comprising acrylic, fluorinated and hydrophilic (acid/amine/ionic/non-ionic) backbone;
- a particulate mixture comprising a plurality of particles at least partially surface treated (with a balance of hydrophobicity and hydrophilicity) with a particle size less than or equal to 100 microns, preferably less than or equal to 325 mesh (44 microns)
- Optionally contains one or more nano particles exhibiting photocatalytic properties, less than 1% and preferably less than 0.5% of total formulation especially for coatings containing particles equal to and over 325 mesh to selectively provide a balance of hydrophobicity and hydrophilicity such as to provide the desired reversible hydrophilic- hydrophobic switching.
- said aqueous coating composition is oleophobic and has a water contact angle hysteresis of ⁇ 25° under certain environmental conditions with variations in at least one or more of temperature, humidity, pH, light which is reversibly switchable to a water contact angle hysteresis of >25° under said environmental conditions offering benefits of both superhydrophobic and superhydrophilic cleaning, good organic and inorganic dirt pickup resistance, resistance to dirt streaks, dirt lines, water spotting, water marks, good cleanability with water and forced air or wind, reduced chalking, good resistance to organic dirt and deposits such as bird droppings and oily stains, and reduced tendency for spreading of water containing rust.
- Said coating has excellent resistance to microbial growth, particularly algae and also exhibits enhanced photocatalytic activity for even rutile grades of Titania, even under diffused sunlight.
- an aqueous coating composition exhibiting reversible hydrophilic-hydrophobic switching under variable environmental conditions including variations in at least one or more of temperature, pH, humidity or other external stimuli including light can be thus achieved with low contact angle hysteresis of ⁇ 25 degrees that is reversibly switchable to a contact angle hysteresis of >25°.
- the method of preparation of said aqueous coating composition is simple and industrially facile which coating composition in being adapted for both brush or spray type applications is also thus attractive to the consumers.
- said silicones both hydrophobic and hydrophilic when combined with the abovesaid ingredients including the particles with a balance of hydrophobicity and hydrophilicity, leads to a surface similar to desert beetle with the protrusions which arise from a plurality of particles being either hydrophobic or hydrophilic.
- Said silicones in the composition while advantageously facilitates to retain the stimuli responsive behavior and reversible wettability switching upon long exposures to environmental conditions, also provides for better weather durability and improved water resistance of the dried film.
- the effect of the silicone crosslinking further enables the coating to maintain a high contact angle conducive for self cleaning even after weathering of the coating over a period of time.
- the coating composition of the present invention thus also exhibits good resistance to organic dirt and deposits such as bird droppings and oily stains even with lovy contents of fluoromonomers ( ⁇ 5%), wherein enhancement in photocata lytic activity are also exhibited even under diffused sunlight with low contents of nano anatase titania or even micronized rutile grades of Ti0 2 with negligible degradation of the organic binder, subject to the selectively high particle to binder ratio with better dispersion of the photocata lytic particles due to the involvement of hydrophobic and hydrophilic silicones in a selective polymeric dispersion.
- said aqueous coating composition reversibly switches from superhydrophobic (contact angle hysteresis ⁇ 25° and water droplet rolling effect) to superhydrophilic (contact angle hysteresis >25° and complete wetting) for at least 2000 hours of accelerated exterior exposure (Atlas and QUV testing), preferably the contact angle hysteresis values are ⁇ 20°, more preferably ⁇ 10° and most preferably ⁇ 5° in one state and higher in the switched state for a deposited 0.02 mL water droplet, wherein preferably the contact angle exhibited in one state is >100° with water, more preferably >120°, even more preferably superhydrophobic having contact angle >150° and contact angle ⁇ 90° in the switched state for a deposited 0.02 mL droplet; and wherein said switching takes place within a varying period of time from a few seconds to several minutes dependent on a particular stimuli.
- said aqueous coating composition has low surface energy of ⁇ 40 mN/m and preferably ⁇ 20 mN/m and most preferably ⁇ 10 mN/m even with the second coat and hence recoatable to enhance film ' build-up to attain desired opacity/ translucency/ hiding/ whiteness.
- said aqueous coating composition comprising at least one or more wetting and dispersing agents, encapsulated or free antimicrobial agents selected from fungicide, algaecide, in-can preservative and optionally involving, one or more nanoparticles exhibiting photocata lytic activity.
- said aqueous coating composition especially favours photocatalytic activity even under diffused sunlight even with cost effective grades of micronized particles such as Titania while maintaining its durability together also exhibiting antimicrobial properties towards microorganisms such as algae, fungi and bacteria.
- said aqueous coating composition wherein said polymeric dispersion comprises dispersion polymers involving primary or secondary dispersion in water or in a water/ co-solvent mixture with or without emulsion/latex polymers.
- said polymeric dispersion comprises dispersion polymers with or without emulsion/ latex polymers that is preferably styrene free and is a free radical polymerization or controlled radical polymerization product of hydrophobic monomers C1-C8 straight, branched or cyclic esters of methacrylic acid/ acrylic acid selected from methyl methacrylate, ethyl methacrylate, butyl Methacrylate, isobutyl ethacrylate, tertiary butyl methacrylate, isobornyl methacrylate, 2-ethylhexyl methacrylate and various isomers and their corresponding acrylates also involving >C8 esters of methacrylic acid/ acrylic acid selected from lauryl acrylate, stearyl acrylate.
- said polymeric dispersion comprises dispersion polymers with or without emulsion/ latex polymers and has monomers selected such that the resulting polymer has an average octanol-water partition coefficient (log Kow) greater than 0.6, preferably greater than 1.5 and more preferably greater than 2.
- said aqueous coating composition wherein said polymeric dispersion including dispersion polymers with or without emulsion/ latex polymers in being styrene free involves styrene or alkylstyrene group as part of the polymer or as a part of additives including opacifiers in less than 15% of the ' film forming composition favouring reduced chalking of said composition on UV exposure.
- said polymeric dispersion comprising dispersion polymers and emulsion/ latex polymers is in the ratio range of dispersion to emulsion polymers of 95:5 to 5:95.
- said polymeric dispersion comprising dispersion polymers and emulsion/ latex polymers is preferably in the ratio range of 30:70.
- said aqueous coating composition wherein said polymeric dispersion comprising dispersion polymers includes primary or secondary dispersion in water or in a water/ co-solvent mixture involves co-solvent that is VOC exempt under Green Seal standard GS-11 with resulting coating VOC's in the range of 1 to 100 grams/liter.
- said polymeric dispersion comprising dispersion polymers with or without emulsion/ latex polymers includes polymers selected from natural polymer, a neoprene polymeri a nitrile polymer, a vinyl acrylic polymer, acrylic polymer; styrene acrylic polymer, styrene butadiene polymer, copolymer of vinyl acetate and butyl acrylate or veova monomers or ethylene/ propylene/ butylene, butadiene, isoprene involving crosslinkers selected from diacrylates, triacrylates, multifunctional acrylates, silane functional monomers, acetoacetate functional monomers, monomers with aziridine, hydrazine, carbodiimide or allylic functionality.
- said aqueous coating composition wherein said polymeric dispersion comprising dispersion polymers with or without emulsion/ latex polymers includes polymers of fluorinated monomers comprising 2 to 20 carbon atoms and polymeric fluorine content in the range of 0.1 to 50% on monomer solids and preferably in the range of less than 5% on monomer solids resulting in economic benefits and a surface energy of ⁇ 50 mN/m , preferably ⁇ 30 mN/m and more preferably ⁇ 20 mN/m
- said polymeric dispersion comprising fluorinated backbone comprises fluorinated monomers including esters of acrylic or methacrylic acid with a linear or branched perfluoroalkyl functional group preferably involving 1-6 fluorinated carbons selected from trifluoroethyl ' meth(acrylate), pentafluoropropyl meth (acrylate), heptafluorobutyl meth(acrylate).
- said aqueous coating composition comprises said ionic or ionizable or non- ionic hydrophilic monomers selected from acrylic acid, methacrylic acid and their aklali metal or quaternary ammonium ion salts; monoolefinic sulphonic acid compounds and their alkali metal salts including 2-acrylamido-2-methylpropanesulphonic acid; acrylates or methacrylates of aminoalcohols, acrylamides selected from ⁇ , ⁇ - dimethylaminopropylacrylamide or N, N-Dimethylaminoethylmethacrylate, polyalkylene oxide group containing monomers or a polymeric chain prepared by polymerizing a polyalkylene glycol (meth)acrylate, hydroxyl alkyl (ethyl, propyl or butyl) (meth)acrylates, wherein the hydrophilic component comprises at least one oxygen, sulfur, phosphorus and/or nitrogen atom.
- the hydrophilic component comprises at least one oxygen, sulfur,
- said aqueous coating composition wherein said acrylic, fluorinated and amine backbone or acrylic, fluorinated and acid backbone of the primary or secondary polymeric dispersion comprises sufficient content of acid or amine monomer including ionic or non-ionic external surfactants adapted for acid or amine value in the range 5 to 100 mg KOH/g.
- said aqueous coating composition wherein said particles comprising either micron sized particles or including particulate mixture of both micron sized and nano sized particles (mixture of micron sized and nano sized particles) and polymeric dispersion are present in selective ratios of 0.9 to 2.5 wherein higher ratios closer to 2 is preferred for a two coat application.
- Said aqueous coating composition including said particles comprises either micron sized particles or includes particulate mixture of both micron sized and nano sized particles, and includes particulate pigments wherein the pigment volume concentration (PVC) is in the range of about 40 to 90 % and preferably in the range of 60 to 80% for the coating and preferably the coating PVC is lower than the practical critical pigment volume concentration (CPVC).
- PVC pigment volume concentration
- said particles comprising either micron sized particles or including particulate mixture of both micron sized and nano sized particles comprises microhized and nanosized particles present in selective ratios of 3.5 to 8 with ratios closer to 8 being preferred for a two coat application.
- said at least partially surface treated particles involve organic and/or inorganic particles having size ranging from 1 nm-100 microns includes surface treated nanometer sized organic and/or inorganic particles have size ranging from about 1 nm to 300 nm, and wherein said particles preferably has mean particle size less than 325 mesh (44 microns) and even more preferably less than 40 microns.
- said aqueous coating composition wherein the mass ratio of said hydrophobic organosilicone to hydrophilic organosilicone is in the range 10/90 to 60/40.
- said hydrophilic organosilicone/silane having a contact angle hysteresis of >10 degrees based on the average of readings for 10 and 20 pL of water when applied on an aerated cement block is present in the range of 10% to 90% by weight of dispersion solids in said composition.
- said hydrophobic organosilicone/silane having a contact angle hysteresis of ⁇ 10 degrees based on the average of readings for 10 and 20 ⁇ _ of water when applied on an aerated cement block is present in the range of 5% to 80% by weight of dispersion solids in said composition.
- said hydrophobic and hydrophilic organosilicone content present as a part of the continuous phase in said composition is 10- 90 % of the weight of the dispersion/binder.
- said aqueous coating composition wherein said wetting agent and dispersing agent is balanced by having said dispersing agent upto 1% and preferably in the range of 0.1 to 0.5% and the wetting agent upto 2% and preferably in the range of 0.2 to 1% by weight in said composition favouring accelerated storage stability for atleast 30 days at 55°C and a composition that passes at least 5 cycles of freeze thaw stability test (-15°C to 30°C).
- Aqueous coating composition as claimed in anyone of the preceding claims including photocatalytically active agents comprising one or more nano particles exhibiting photocatalytic properties in amounts less than 1% and preferably less than 0.5% of total formulation especially for coatings containing particles more than or equal to 325 mesh, preferably comprise nano titania of the anatase or rutile grade in the range of 0.1 to 1% of the formulation with a particle size of 1 to 300 nm and more preferably comprise titania in anatase form adapted for enhancement of photocatalytic activity of said composition (while - maintaining its durability) against organic pollutants of atleast >5%, more preferably >20% and most preferably >80% of the activity of the control.
- Enhanced photocatalytic activity of coating with good durability is also demonstrated by even micron sized particles (>300 nm) such as Titania (anatase or rutile).
- Said aqueous coating composition including photocatalytically active agents comprising one or more nano particles exhibiting photocatalytic properties in amounts less than 1% and preferably less than 0.5% of total formulation especially for coatings containing particles more than or equal to 325 mesh, preferably comprise nano titania of the anatase or rutile grade in the range of 0.1 to 1% of the formulation with a particle size of 1 to 300 nm and more preferably comprise titania in anatase form adapted for enhancement of photocata lytic activity of said composition while maintaining its durability by even micron sized particles (>300 nm) of Titania (anatase or rutile) against organic pollutants of atleast >5%, more preferably >20% and most preferably >80% of the activity of the control.
- photocatalytically active agents comprising one or more nano particles exhibiting photocatalytic properties in amounts less than 1% and preferably less than 0.5% of total formulation especially for coatings containing particles more than or equal to 325 mesh, preferably comprise
- said at least partially surface treated particles are selected from substantially spherical particles, nodular particles, platy particles, cubical paraticles, various irregularly shaped particles, and mixtures thereof.
- said aqueous coating composition wherein said at least partially surface treated particles comprise at least one material selected from the group of silicates, doped silicates, minerals, metal oxides, silicas and metal powders and are selected from the group of aluminum oxides (alumina), titanium oxide, zirconium oxide, silver, nickel, nickel oxide, iron oxide, and alloys, polystyrene particles, (meth)acrylate particles, PTFE particles, silica particles, polyolefin particles, polycarbonate particles, polysiloxane particles, silicone particles, polyester particles, polyamide particles, polyurethane particles, ethylenically unsaturated polymer particles, polyanhydride particles, biodegradable particles, polycaprolactone, nanofibers, nanotube
- said aqueous coating composition wherein said at least partially surface treated particles comprise particles that are surface treated with compounds selected from silanes (alkylsilanes, perfluoroalkylsilanes, and alkyldisilazanes), silicones and fluorinated compounds or fluorinated acrylics or other organic/ inorganic treatments to achieve a balance between hydrophobicity and hydrophilicity.
- silanes alkylsilanes, perfluoroalkylsilanes, and alkyldisilazanes
- silicones and fluorinated compounds or fluorinated acrylics or other organic/ inorganic treatments to achieve a balance between hydrophobicity and hydrophilicity.
- said at least partially surface treated particles comprise preferably externally surface treated particles with selective hydrophobic and hydrophilic balance and are obtained of sol-gel treatment to prevent any colour change or graying of white particles present in said coating composition, especially where fluorosilanes or fluorosilicones are used for treatment or is alternatively in-situ treated while preparation of the coating with said mixture of hydrophobic and hydrophilic silicones/silanes.
- said aqueous coating composition is flowable comprising at least one organic diluent or solvent, wherein the solvent is selected from the group comprising of water, an alcohol, an ether, a ketone, an ester, a glycol, a glycol ether, an alkylene carbonate, a C 5 - QL 8 aliphatic hydrocarbon, a C 6 - Ci 8 aromatic hydrocarbon, various polar/non-polar solvents and mixtures thereof.
- the solvent is selected from the group comprising of water, an alcohol, an ether, a ketone, an ester, a glycol, a glycol ether, an alkylene carbonate, a C 5 - QL 8 aliphatic hydrocarbon, a C 6 - Ci 8 aromatic hydrocarbon, various polar/non-polar solvents and mixtures thereof.
- said polymeric dispersion comprises thermoplastic or thermosetting polymers including a single polymer or a blend of more than one polymer or self crosslinking polymers selected from acrylics, polyesters, polyurethanes, polycarbonates, polyolefins, alkyds, epoxies, polyamides, fluoropolymers, silicone polymers and their hybrids, wherein said fluoropolymers include polyperfluoropolyethers, and a polymer having one or more monomer repeat units selected from the group consisting of ethylene, propylene, styrene, tetrafluoroethylene, vinylidene fluoride, hexafluoropropylene, perfluoro(methyl vinyl ether), perfluoro(ethyl vinyl ether), and perfluoro(propyl vinyl ether) and/ or includes a colloid, a latex, or a suspension of a fluoropolymer selected from a group comprising of poly
- said polymeric dispersion comprising polymers have a Tg of -30 to 100 deg C or preferably Tg in the range of 0 to 50°C. More preferably, said aqueous coating composition wherein said polymeric dispersion comprising polymers have surface energy of ⁇ 50 ml ⁇ l/m and preferably less than 30 mN/m and more preferably ⁇ 20 mN/m.
- Said aqueous coating composition comprise components that are multifunctional or having individual functions selected from thickeners, biocides, wetting and dispersing agents, fluorosurfactants, defoamers, opacifying polymers, cosolvents, coalescents, plasticizers, pigments, special effect pigments, extenders, colorants, freeze thaw stabilizers, buffers, fire retardants, uv absorbers, organic fiber material, and inorganic fiber materials, flow and levelling agents, adhesion promoters, oil and water repellents and other components known to be provided in paint formulations.
- components that are multifunctional or having individual functions selected from thickeners, biocides, wetting and dispersing agents, fluorosurfactants, defoamers, opacifying polymers, cosolvents, coalescents, plasticizers, pigments, special effect pigments, extenders, colorants, freeze thaw stabilizers, buffers, fire retardants, uv absorbers, organic fiber material, and inorganic fiber
- said aqueous coating composition is ambient curing including air curing, infra-red curing and thermal curing.
- a dry film of said coating has a dry film thickness of 1 to 200 microns and preferably 40 to 120 microns and withstands at least 30 to 1000 cycles and preferably 100 to 3000 cycles in a wet scrub resistance test with a scrub brush and a 0.45 kg load.
- Said aqueous coating composition is adapted for applications comprising self cleaning, easy cleaning, anti-graffiti, stain resistant, soil-degrading, deodorizing, air-cleaning, antifouling, antimicrobial, water-cleaning, superhydrophobicizing, superhydrophilicizing and/or antifogging effects.
- a process for the preparation of said aqueous coating composition comprising the steps of:
- oligomeric or polymeric dispersion/binder comprising acrylic, fluorinated and hydrophilic (acid/amine/ionic/non-ionic) backbone;
- said at least partially surface treated organic or inorganic particles is provided by subjecting the particles to sol-gel treatment with compounds selected from silicones, fluorinated compounds to prevent color change of the particles.
- aqueous coating composition that is adapted for substrates selected from masonary, concrete, cementitious, plaster, baked clay tiles, celiulosic, wood, one or more polymer, dry or damp surfaces, brick, tile, stone, grout, mortar, composite materials, gypsum board, , porous and non porous surfaces, interior surfaces or surfaces exposed to weathering on at least one surface of the substrate, which surface is preferably pretreated for better adhesion of the aqueous coating composition with various types of surface treatments.
- a commercial or industrial material a structure or building or a substrate comprising said aqueous coating reversible switchable composition exhibiting reversible hydrophilic-hydrophobic switching under variable environmental conditions including variations in at least one or more of temperature, pH, humidity or other external stimuli including light.
- aqueous coating of said reversible switchable composition selectively applying to atleast a portion of commercial or industrial material, a structure or building or a substrate requiring said " reversible coating characteristic such as to exhibit reversible hydrophilic- hydrophobic switching under variable environmental conditions including variations in at least one or more of temperature, pH, humidity or other external stimuli including light.
- Fig 1 reveals contact angle measurement showing a hydrophobic and hydrophilic drop
- Fig 2 reveals contact angle measurement showing the advancing and the receding angle
- Fig 3 reveals the image of untreated Ti02;
- Fig 4 reveals the image of treated " ⁇ 02;
- Fig 5 reveals Consecutive images of 5 pL incrementing volumes of water droplet from the syringe of the contact angle meter, with 45 pL forming a droplet having a contact angle of 143°;
- Fig 6 reveals rheogram (temperature sweep at constant shear rate) for the water based pigmented coating system
- Fig 7 reveals differential scanning calorimetry thermogram for the dried pigmented coating
- Fig 8 reveals scanning electron micrograph of the paint surface showing 2 levels of topography
- Fig 9 reveals an AFM picture of the paint composition
- Fig 10 reveals an AFM picture showing the thermoresponsive behaviour of the paint (left: hot condition, right: cold condition)
- Fig 11 reveals a photograph of the paint exposed outdoors for a period of 6 months showing its resistance to dirt streaks and water marks; (left side: paint according to present invention, right side: commercially available self cleaning paint).
- the present invention provides for stimuli responsive self- cleaning water-borne pigmented aqueous coating composition responsive to at least one or more variations of temperature, pH, humidity or other external stimuli including light under variable environmental conditions that exhibits reversible switchable super hydrophilic- super hydrophobic characteristics.
- the said coating in comprising one or more oligomeric or polymeric dispersion/binder comprising acrylic, fluorinated and hydrophilic (acid/amipe/ionic/non-ionic) backbone selectively present in combination with at least ; partially surface treated particles, hydrophobic and hydrophilic silanes is thus adapted for a stimuli responsive self-cleaning aqueous paint/ coating compositions and self-cleanable surfaces, which is oleophobic and exhibit the desired reversible hydrophilic-hydrophobic switching to provide for a water contact angle hysteresis of ⁇ 25° under certain environmental conditions with variations in at least one or more of temperature, humidity, pH, light that is switchable to a water contact angle hysteresis of >25° under said environmental conditions offering benefits of both superhydrophobic and superhydrophilic cleaning, good organic and inorganic dirt pickup resistance, resistance to dirt streaks, dirt lines, water spotting, water marks, good cleanability with water and forced air or wind, reduced chalking
- Said particles comprising at least partially surface treated particles are functionalized pigments or extenders present in selective sizes and ratios involving particulate mixture in selective ratios of micro and nano sized particles in said polymeric dispersion. More specifically, said pigments are surface treated/ functionalized either in-situ or prior to incorporation in the coatings and are present in said polymeric dispersion under a selective binder to pigment/ filler ratio such as to favour the desired contact angle hysteresis of ⁇ 25 degrees.
- said attainment of low contact angle hysteresis ( ⁇ 25 degrees) and subsequent reversible wettability switching by the aqueous coating composition/ stimuli responsive self-cleaning coatings of the present invention enables the flow of rain water or forced air to clean the dirt from said coating/ paint surface wherein said coating can be formed by simple mixing of the components and applied by simple techniques such as brush and spray application.
- the present invention in providing for at least partially surface treated particles including particles based on a mixture of micro and nanosized particles with a balance of hydrophobicity and hydrophilicity, a combination of silicones both hydrophobic and hydrophilic in combination with a suitable dispersion polymer leads to a surface similar to the desert beetle, with the protrusions which arise from a plurality of particles being either hydrophobic or hydrophilic.
- the hydrophobic or hydrophilic polymer coating shall be understood as a polymer coating which can change reversibly from a hydrophobic state into a hydrophilic state.
- the surface is therefore "switchable” and reversibly changed from one state to the other as a result of the influence of an external parameter.
- This external stimulus includes temperature change or humidity change or change in the intensity of UV light or change in the pH value.
- the polymer coating is hydrophobic and at a pH value of greater than approximately 7.0 it is hydrophilic.
- the coating changes from a hydrophobic state into a hydrophilic state and vice versa at another suitable pH value, which preferably ranges between 3.0 and 10.0.
- the coating is hydrophobic at a temperature greater than ambient temperature (approximately 30° C) and is hydrophilic at a temperature of less than approximately 30° C.
- the coating also changes from a hydrophobic state to a hydrophilic state at another suitable temperature, which preferably ranges between 5° C and 60° C.
- the polymer coating is hydrophobic and at a humidity value of greater than approximately 50% it is hydrophilic.
- the coating also changes from a hydrophobic state into a hydrophilic state and vice versa at another suitable humidity value, which preferably ranges between 5 to 100%.
- the polymer coating is hydrophilic and under low intensity light the polymer coating is hydrophobic.
- the coating can also change from a hydrophobic state into a hydrophilic state and vice versa under a range of light between 200 to 800 nm.
- said coating also exhibits enhanced photocatalytic activity even under diffused sunlight with negligible degradation of the organic polymer due to the presence of a combination of hydrophilic and hydrophobic organosilicones and titania, thereby showing improved resistance to organic dirt and deposits such as bird droppings and oily stains allowing them to degrade with UV exposure and finally get cleaned from the surface with rain or wind.
- the ; perfluorinated groups and silicone groups cover the surface of the coating.
- the hydrophilic moiety contained in the copolymer and the silicone appears on the surface of the coating to provide wettability and thereby to improve the desoiling properties. This is believed to be the reason why the water repellency and desoiling properties can be obtained simultaneously that is further assisted by the selectively high particle to polymer ratio.
- the applied switchable hydrophobic or hydrophilic coating comprises at least one oligomer or polymer and a plurality of treated particles (with a balance of hydrophobicity and hydrophilicity) which is suited to reversibly switch the coating from a hydrophobic state to a hydrophilic state as a result of the influence of an external parameters.
- Particles treated with a balance of hydrophobicity and hydrophilicity imply that the surface of the particles are functionalized with a combination of hydrophobic and hydrophilic groups.
- the formulation of the present invention is thus economical in involving cost effective photocatalysts wherein even micron sized rutile or anatase titanium dioxide photocatalyst in the selective polymeric dispersion involving silicones exhibits enhanced photocatalytic activity to be activated by visible light and even diffused sunlight.
- the deposited dirt on the rough surface of the smart paint of the present invention is able to get cleaned by this combination of hydrophobic and hydrophilic cleaning.
- the coating not only prevents the deposition of dirt from the water but also prevents the infestation and growth of unwanted organisms, such as microorganisms, algae, lichen, and mosses.
- the silicone containing components are present in a concentration of 10 to 90% of the organic binder while maintaining the stimuli responsive behavior and also maintaining a clean surface that is free from dirt lines, streaks, water spots and algae, wherein said coating composition of the present invention displays enhanced photocatalytic activity under diffused sunlight with reduced chalking and also exhibits good resistance to organic dirt and deposits such as bird droppings and oily stains. Further said coating also has a reduced tendency for spreading of water containing rust.
- the coated substrate loses the hydrophobic property over the course of approximately one second to approximately one day, preferably over trie course of approximately one minute to approximately 10 minutes.
- the switching may happen within a varying period of time from a few seconds to several minutes which are stimuli dependent.
- binders are meant, polymers, oligomers and low molecular mass substances which form a solid film on a surface.
- the binders serve, for example, to fix the particles on the surface of the substrate to be coated.
- the binder should be stable when exposed outdoors to strong UV light, rain, wind, etc. for a minimum time of one month to several years.
- Preferred binders used in the invention have a surface energy ⁇ 50 mN/m, preferably ⁇ 30 mN/m and in particular ⁇ 20 mN/m.
- the binders generally comprise thermoplastic polymers or organic prepolymers which undergo crosslinking by a thermal, oxidative or photochemical curing process.
- the fluoropolymer could be any polymer having fluorine functionality.
- examples of such polymers include, but are not limited to fluoroolefins, vinylidene difluoride-based polymers, fluoroethylene vinyl ethers.
- the fluoropolymer may be a thermoplastic or thermoset polymer. Fluoropolymers are available under the trade name Kynar and lumiflon and may be blended with the preferred binder. Further, to the polyfluorinated copolymer, other polymer blends may be incorporated. Further self crosslinking polymers can also be used.
- the polymers can be acrylics, polyesters, polyurethanes, alkyds, epoxies, urethane acrylates, homopolymers and copolymers of ethylenically unsaturated monomers, and acrylic acid/maleic anhydride copolymers known in the art.
- co-binders are natural waxes such as beeswax, carnauba wax, wool wax, candelilla wax, and also synthetic waxes such as montanic acid waxes, montanic ester waxes, amide waxes, and also waxlike polymers of ethylene and of propylene (polyethylene wax, polypropylene wax).
- natural waxes such as beeswax, carnauba wax, wool wax, candelilla wax
- synthetic waxes such as montanic acid waxes, montanic ester waxes, amide waxes, and also waxlike polymers of ethylene and of propylene (polyethylene wax, polypropylene wax).
- polymerization initiating source various polymerization initiators such as an organic peroxide, an azo compound and a persulfate may be employed.
- an emulsifier for emulsion polymerization almost all emulsifiers including anionic, cationic and non-ionic emulsifiers may be used. Catalyst concentration can be about 0.1 to 2 percent based on the weight of total monomers.
- the number average molecular weight of the binder polymers may vary over a wide range and is generally in the range from 1000 to 1 million g/mol and preferably in the range from 2500 to 0.5 million g/mol, in particular 10,000 to 0.5 million, g/mol (determined by GPC).
- the polymer has a Tg of -30 to 100 deg C. Alternatively a blend of polymers having different Tg's can be used. Most preferably the Tg is in the range of 0 to 50°C.
- the aqueous polymer dispersions have a solid content of 25% to 70% by weight, preferably 40% to 65% by weight.
- the polymer can be made by Free radical polymerization or controlled radical polymerization (CRP).
- CRP / living polymerization can be used to achieve a desired molecular weight and a narrow weight distribution or polydispersity.
- CRP provides a maximum degree of control for the synthesis of polymers with predictable and well-defined structures.
- Atom transfer radical polymerization (ATRP), nitroxide-mediated polymerization (NMP), reversible addition fragmentation transfer polymerization (RAFT), activated regenerated by electron transfer (ARGET) are some of the techniques of CRP.
- copolymer of the present invention various systems and conditions for the polymerization reaction may be employed, and any of various polymerization systems such as bulk, solution, suspension or emulsion polymerization may be used.
- Examples of preferred hydrophobic monomers are C 2 -C 24 olefins, such as ethylene, propylene, n-butene, isobutene, n-hexene, n-octene, ispoctene, n-decene, isotridecene, C 5 - C 8 cycloolefins such as cyclopentene, cyclopentadiene, cyclooctene, vinyl aromatic monomers, such as styrene and alpha-methylstyrene, and also fluoroolefins and fluorochloroolefins such as vinylidene fluoride, chlorotrifluoroethylene, tetrafluoroethylene, vinyl esters of linear or branched alkane carboxylic acids having 2 to 36 carbon atoms, e.g., vinyl acetate, vinyl propionate, vinyl n-butyrate, vinyl isobutyrate, vinyl hexanoate
- sub.36 alkanols e.g., ethyl (meth)acrylate, n-butyl (meth)acrylate, tert-butyl (meth)acrylate, n-hexyl (meth)acrylate, 2-ethylhexyl (meth)acrylate, 2-propylheptyl (meth)acrylate, lauryl (meth>acrylate and stearyl (meth)acrylate and also vinyl ethers and allyl ethers of C.sub.2 -C.sub.36 alkanols, such as n-butyl vinyl ether and octadecyl vinyl ether, fluorinated monomers and also monomers containing poiysiloxane groups.
- Typical binder polymers of this type are polyethylene, polypropylene, polyisobutene, polychlorotrifluoroethylene, polytetrafluoroethylene, polyvinyl acetate, polyethyl methacrylate, poly-n-butyl methacrylate, polyisobutyl methacrylate, poly-tert-butyl methacrylate, polyhexyl methacrylate, poly(2-ethylhexyl methacrylate), polyethyl acrylate, poly-n-butyl acrylate, polyisobutyl acrylate, poly-tert-butyl acrylate, poly(2-ethylhexyl acrylate), and copolymers of maleic acid with at least one hydrophobic monomer selected from C.sub.3 -C.sub.6 olefins, C.sub.l -C.
- sub.36 alkyl vinyl ethers the vinyl esters of aliphatic C.sub.l -C. sub.36 carboxylic acids.
- Further suitable binders are poly-C.sub.l -C.sub.4 -alkylene oxides, such as polyoxymethylene, polypropylene oxide and polybutylene oxide, polytetrahydrofuran and also polycaprolactone, polycarbonates, polyvinylbutyral, polyvinylformal, and also linear or branched polydialkylsiloxanes such as polydimethylsiloxane (silicones).
- the polymer contains fluorinated monomers (comprising from 2 to 20 carbon atoms). Further the polymer contains fluorine content in the range of 0.1 to 50% on monomer solids and preferably in the range of less than 5% on monomer solids thus offering economic benefits.
- the fluorinated monomers can be esters of acrylic or methacrylic acid with a linear or branched perfluoroalkyl functional group.
- the fluoromonomer or fluoromonomers contain up to six fluorinated carbons such as trifluoroethyl meth(acrylate), pentafluoropropyl meth(acrylate), heptafluorobutyl meth(acrylate) among others.
- the hydrophilic moiety a non-ionic group such as a polyoxyethylene chain, a polyoxypropylene chain, a polyoxybutylene chain, an anionic group such as a sulfonic acid group or a carboxylic acid group, or a cationic group such as an ammonium salt or an amine salt.
- a non-ionic group such as a polyoxyethylene chain, a polyoxypropylene chain, a polyoxybutylene chain, an anionic group such as a sulfonic acid group or a carboxylic acid group, or a cationic group such as an ammonium salt or an amine salt.
- an amphipathic monomer having a hydrophilic- lipophilic balance (HUB) of hydrophilic moiety/lipophilic moiety being from 5 to 15 is selected.
- the polymer wherein the ionic or ionizable hydrophilic monomer or monomers are selected from: acrylic acid, methacrylic acjd, maleic acid and their alkali metal or quaternary ammonium ions salts; monoolefinic sulphonic acid compounds and their alkali metal salts including 2-acrylamido-2-methylpropanesulphonic acid; acrylates or methacrylates of aminoalcohols, acrylamides such as ⁇ , ⁇ -dimethylaminopropylacrylamide or N,N- Dimethylaminoethylmethacrylate, polyalkylene oxide group containing monomers or a polymeric chain prepared by polymerizing a polyalkylene glycol (meth)acrylate, hydroxyl alkyl (ethyl, propyl or butyl) (meth)acrylates.
- the hydrophilic component comprises at least one oxygen, sulfur, phosphorus and/or nitrogen atom.
- the polymeric dispersion comprises acid or amine monomer (in the range 5 to 100 .mg KOH/g acid or amine value) and may contain external surfactants;
- the amount of hydrophilic monomer is in the range of 2 to 20%. If the hydrophilic monomer content is high (>20%), the coating will have very poor water resistance, exhibit yellowing (where hydrophilic monomer is amine) and hence poor durability. Further, it will not be of any commercial importance as a coating.
- one or more additional monomers such as a monomer having a cross-linking group or a chelating group to the substrate, a high Tg monomer for improving the film-forming property, or a low Tg monomer for improving the flexibility of the coating film, may be incorporated without any particular restriction.
- the polymer is preferably styrene free with the preferred hydrophobic monomers being CI to C8 straight, branched or cyclic esters of methacrylic acid/ acrylic acid such as methyl methacrylate, ethyl methacrylate, butyl Methacrylate, isobutyl Methacrylate, tertiary butyl methacrylate, isobornyl methacrylate, 2-ethylhexyl methacrylate and various isomers and their corresponding acrylates.
- methacrylic acid/ acrylic acid such as methyl methacrylate, ethyl methacrylate, butyl Methacrylate, isobutyl Methacrylate, tertiary butyl methacrylate, isobornyl methacrylate, 2-ethylhexyl methacrylate and various isomers and their corresponding acrylates.
- Styrene or alkylstyrene may be used as part of the polymer or through additives such as opacifiers such that it is under 15% of the film forming composition to reduce chalking tendency on UV exposure.
- opacifiers such that it is under 15% of the film forming composition to reduce chalking tendency on UV exposure.
- Other long chain monomers, >C8 esters of methacrylic acid/ acrylic acid such as lauryl acrylate, stearyl acrylate etc. can also be used.
- the polymer is selected from the group consisting of a natural polymer; a neoprene polymer; a nitrile polymer; a vinyl acrylic polymer; a acrylic polymer; a styrene acrylic polymer; a styrene butadiene polymer; a copolymer of vinyl acetate and butyl acrylate or veova monomers or ethylene/ propylene/ butylenes, butadiene, isoprene etc.
- Crosslinkers such as diacrylates, triacrylates, multifunctional acrylates, silane functional monomers, acetoacetate functional- monomers, monomers with aziridine, hydrazine, carbodiimide, allylic functionality, divinyl benzene, divinylsulfone, thallyl phosphate, zinc diacrylate, zinc dimethacrylate, diallyphthalate, diallylacrylamide, polyethylene glycol dimethacrylate, polypropylene glycol dimethacrylate, pentaerythritol tetramethacrylate, pentaerythritol trimethacrylate, pentaerythritol dimethacrylate, trimethylolpropane -tri methacrylate, dipentaerythritol hexamethacrylate, di pentaerythritol pentamethacrylate, glycerol trimethacrylate or a mixture thereof can also be used.
- the monomers are selected such that the resulting polymer has an octanol-water partition coefficient (log Kow) greater than 0.6, preferably greater than 1.5 and more preferably greater than 2 (calculated as a weighted average of the monomers used to prepare the polymer).
- the octanol/water partition coefficient is defined as the ratio of a chemical's concentration in the octanol phase to its concentration in the aqueous phase of a two-phase octanol/water system and can be calculated by methods known in the art such as UV/visible absorbance, use of separatory funnels, high performance liquid chromatography etc.
- An aqueous coating composition wherein said polymeric dispersion comprises dispersion polymers with or without emulsion/latex polymers wherein the ratio of dispersion to emulsion polymers is preferably in the range of 95: 5 to 5:95.
- amines can be used as the neutralizing agents for the anionic polymers such as liquor ammonia, morpholine, triethyl amine, vantex T, AMP-95, Advantex etc.
- acids can be used such as acetic acid etc. for the neutralization of the cationic polymers.
- particle is intended to include any discrete particle, primary particle, aggregate and/or aggregated collection of primary particles, agglomerate and/or agglomerated ' collection of aggregates, colloidally dispersed particles, loose assemblies of particulate materials, and combinations thereof.
- the particles that may be used include organic or inorganic particles currently used and known to one skilled in the art.
- the particles preferably have an average particle diameter of between 1 nm to 44 microns, preferably being of inorganic nature. Depending on the size and material of the particles used, it is possible to obtain translucent or opaque self- cleaning surfaces.
- Surface treated micron sized particles involve organic or inorganic particles lower than 44 microns and wherein said surface treated nanometer sized organic or inorganic particles in the size range of about 1 nm to 300 nm.
- micron sized particles can be used or a combination of micron and nano sized particles can be used to prepare the aqueous coating composition. Only nano sized particles may be used, however they increase the cost of the coating.
- the surface of particles should preferably have a high oil absorption value or an irregular structure. Examples of such materials include clays from Hoffmann (Silitin and Aktisil grades), high oil absorption value titanium dioxide from DuPont e.g. Ti02 R931, calcium carbonate grades from Omya and Jorcal grades form Jordan carbonate Co.
- extenders prepared using the ground process which leads to rougher surfaces as compared to the precipitation process. Cristobalite and respirable crystalline silica are not desired due to their carcinogenicity.
- Preferred fillers include gypsum, magnesium silicate, magnesium or calcium carbonate, pyrophyllite, mica, pumice, silica, diatomite, barium sulphate, alumina or mixtures thereof.
- the filler also serves to improve the film durability, abrasion resistance, and lower cost.
- the filler may be present in the aqueous composition in a range of from about 2.0 percent by weight to about 50.0 percent by weight, preferably from about 5.0 percent by weight to about 40.0 percent by weight based on the total weight of the composition.
- micronized to nanosized particles is in selective ratios of 3.5 to 8 with ratios closer to 8 being preferred for a two coat application.
- the particulate mixture (micron and nano meter sized) and polymeric dispersion is in selective ratios of 0.9 to 2.5 wherein higher ratios closer to 2 being preferred for a two coat application.
- the micron sized and nano sized particles may be of the same chemistry or different chemistries.
- the nano sized particle maybe for example silicon dioxide, calcium carbonate or Teflon and the micron sized particle maybe silicon dioxide, calcium carbonate or titanium oxide.
- Nanoparticles that can be used to make the coatings of this invention are generally from the class of fumed silica's, titania's, zinc oxides, aluminium oxides, zirconium dioxide and tin dioxide.
- the average particle size may be from about 1 nm to 300 nm.
- inorganic substances there may be mentioned by way of example: metal oxides, mixed oxides, silicates, sulfates, phosphates, borates, metal sulfides, oxosulfides, selenides and sulfoselenides, metal nitrides and oxide-nitrides and metal powders.
- organic structure-forming particles there may be mentioned by way of example carbon blacks and nanoscale organic polymeric particles, and among these fluorine-containing polymers.
- finely divided organic powders with a hydrophobic surface are finely divided polymer powders, e.g., polytetrafluoroethylene powders or C2-C4 polyolefin powders, examples being the polyethylene powders and polypropylene powders.
- the structure-forming particles used are preferably those which comprise at least one material selected from silicates, doped silicates, minerals, metal oxides, silicas, and polymers.
- the structure-forming particles are particularly preferably naturally occurring, precipitated or pyrogenically prepared metal oxides.
- Pyrogenic silicas are commercially obtainable with an average primary particle size in the range from about 5 to 40 nm.
- the silicon dioxide utilized is a colloidal silicon dioxide.
- Such products are commercially available from a number of sources, including Cabot Corporation, (under the trade name CAB-O-SIL) and Degussa, Inc., (under the trade name AEROSIL).
- a plurality of particles of different particle sizes can be used for the coating composition with different hydrophobic and hydrophilic silicone treatments provided they are incorporated with the polymer in certain ratios which are discussed below.
- Hydrophobic Titanium dioxide e.g. R101, R102, R103, R104 and R105 grade (0.31 microns average particle size) is available from companies such as DuPont, which are recommended for use in plastics products. These grades being hydrophobic are difficult to disperse into waterborne systems. Even if dispersed with high quantity of surfactants, they lead to poor hydrophobicity (low water contact angles because of high quantity of surfactants).
- Aktisil grades are also available with different silane treatments Aktisil EM (epoxysilane), AM (aminosilane) etc.
- Micronized PTFE is available from Micro Powders, Inc. Some examples are Fluo 300 (mean particle size 5 to 6 microns), Fluo 625F-H (13 to 21 microns), Fluo HT-LS (2 to 3 microns).
- the photocatalytically active agent comprises titanium dioxide in a form selected from the group consisting of amorphous form, partially crystalline form, and anatase form.
- the photocatalyst may be selected from titanium dioxide (anatase and rutile form), zinc oxide, zirconium oxide, tin oxide, vanadium trioxide, ferric oxide, dibismuth trioxide, tungsten trioxide, and-strontium titanate and combinations thereof. Titanium dioxide is preferred because of its high photocatalytic activity. It is known that the photocatalytic activity of the photocatalysts changes in the order: titanium dioxide>zinc oxide>zirconium oxide>tin oxide>vanadium oxide.
- a suitable source of light is from sunlight, diffused sunlight, a room lamp, a fluorescent lamp, a mercury lamp, an incandescent lamp, a xenon lamp, a high pressure sodium lamp, a metal halide lamp, a BLB lamp and light sources known to the person skilled in the art.
- the photocatalytically active particles present in the mixture according to the invention develop their activity on irradiation by sunlight.
- the preferred photocatalysts are p25 (21 nm, BET surface area 50 m 2 /g / mixed anatase and rutile) from Evonik, VP Aeroperl P25/ 20 (20 pm) and LPX 21530 (30 nm) from BYK.
- rutile grade nanosized or micron sized Ti0 2 may also be used alone or in combination with nano Titania.
- Hydrophobic nanometer sized silica available from Evonik under the trade name Aerosil R972 (primary, particle size 16 nm), Aeroxide LEI (primary particle size 20 nm), LE2 (primary particle size 7 nm) and LE3 from Evonik. can be used.
- hydrophilic grades are available such as Cabosil M5 (200 nm - 300 nm aggregates with a primary particle size of 14 nm in diameter) from Cabot or Aerosil R200 (average particle size of 12 nm) from Evonik that can be either heat treated or treated by sol-gel process.
- the particle be treated to exhibit a balance of hydrophobicity and hydrophilicity.
- the hydrophilic or hydrophobic behavior of a solid is determined essentially by the groups at its surface. For example, particles which have hydroxyl groups at their surface are usually hydrophilic. However, if there are hydrocarbon groups, such as, alkyl groups, and especially longer chain alkyl groups, at the surface of the particle, then the particles have hydrophobic properties.
- hydrophilic or hydrophobic character of inorganic or organic particles This can be accomplished by adsorption of substances on the surface of a particle, especially by chemical reactions with reactive groups, which are at the surface of the particle.
- This balance of hydrophobicity and hydrophilicity may be obtained by using a mixture of hydrophobic and hydrophilic particles which are available commercially, using particles treated with hydrophobic and hydrophilic surface modifiers (organosilanes etc.) by the sol- gel, heat treatment process or other process known in the art, using particles treated with hydrophobic and hydrophilic organosilicones during the paint making process.
- nano sized particles are treated with hydrophobic surface groups and the micron sized particles are treated with hydrophilic surface groups or vice versa.
- both the particles are treated with functional groups such that they exhibit a balance of hydrophobicity and hydrophilicity.
- the modifying agent is used in an amount less than equivalent, so that not all of the reactive groups at the surface of the particles can be modified, it may be assumed that the distribution of the modified groups on the surface follows statistical laws so that the modified particle behaves equally hydrophilic or hydrophobic at any position on its surface.
- the solids with modified surface properties known from the state of the art have hydrophobic or hydrophilic groups, which are located at the surface of the particles in a uniform random distribution. Therefore, it is only possible to hydrophobize a hydrophilic particle more or less pronouncedly. This is shown for by example dispersing treated Ti02 in water and the particles partly float on water and partly disperse in water.
- the surface of the particle may be modified by physisorption, chemisorption or covalent bonding with organic molecules with a variety bf hydrophobic and hydrophilic functional groups.
- the hydrophobic properties are a result of a suitable hydrophobizing treatment, e.g., treatment with at least one compound from the group of the silsesquioxanes, perfluoroacrylic resins, organosilanes, alkylsilanes, fluorinated silanes, disilazanes, waxes, paraffins, fatty esters, fluorinated and/or functionalized alkanes.
- a suitable hydrophobizing treatment e.g., treatment with at least one compound from the group of the silsesquioxanes, perfluoroacrylic resins, organosilanes, alkylsilanes, fluorinated silanes, disilazanes, waxes, paraffins, fatty esters, fluorinated and/or functionalized alkanes.
- Suitable organosilanes include, but are not limited to: alkylchlorosilanes; alkoxysilanes, e.g., ethyltrimethoxysilane, ethyltriethoxysilane, n-propyltrimethoxysilane, n- propyltriethoxysilane, i-propyltrimethoxysilane, i-propyltriethoxysilane, butyltrimethoxysilane, butyltriethoxysilane, hexyltrimethoxysilane, octyltrimethoxysilane, 3- mercaptopropyltrimethoxysilane, n-octyltriethoxysilane, phenyltriethoxysilane, polytriethoxysilane; trialkoxyarylsilanes; isooctyltrimethoxy-silane; N-(
- alkylchlorosilanes include, for example, methyltrichlorosilane, dimethyldichlorosilane, trimethylchlorosilane, octylmethyldichlorosilane, octyltrichlorosilane, octadecylmethyldichlorosilane and octadecyltrichlorosilane.
- Suitable materials include, for example, methylmethoxysilanes such as methyltrimethoxysilane, dimethyldimethoxysilane and trimethylmethoxysilane; methylethoxysilanes such as methyltriethoxysilane, dimethyldiethoxysilane and trimethylethoxysilane; methylacetoxysilanes such as methyltriacetoxysilane, dimethyldiacetoxysilane and trimethylacetoxysilane; vinylsilanes such as vinyltrichlorosilane, vinylmethyldichlorosilane, vinyldimethylchlorosilane, vinyltrimethoxysilane, vinylmethyldimethoxysilane, vinyldimethylmethoxysilane, vinyltriethoxysilane, vinylmethyldiethoxysilane and vinyldimethylethoxysilane.
- methylmethoxysilanes such as methyltrimethoxysilane, dimethyldimethoxys
- Suitable disilazanes include for example, but are not limited to: hexamethyldisilazane, divinyltetramethyldisilazane and bis(3,3-trifluoropropyl)tetramethyldisilazane.
- Cyclosilazanes are also suitable, and include, for example, octamethylcyclotetrasilazane.
- Suitable fluorinated silanes include the fluorinated alkyl-, alkoxy-, aryl- and/or alkylaryl- silanes, and fully perfluorinated alkyl-, alkoxy ⁇ , aryl- and/or alkylaryl-si lanes.
- fluoroalkyl silanes include, but are not limited to: those marketed by Degussa under the trade name of Dynasylan F8261, F8815 etc.
- An example of a suitable fluorinated alkoxy- silane is perfluorooctyl trimethoxysilane.
- oligomeric silicones may also be used for the treatment such as DC 3074 and DC 3037 from Dow Corning, Silres SY 231 from Wacker etc. Oligomers have the benefit of having a very low vapour pressure whereas monomeric corresponding compounds have a comparatively high vapour pressure.
- Hydrophilic organosilanes that may be used include tetraalkoxysilane or silanes having hydrophilic functional groups other than hydroxy groups after hydrolysis. It is preferable to use a silane having one or two hydrophilic functional groups. Particular examples thereof include a glycidoxyalkyl group, aminoalkyl group, isocyanatoalkyl group, etc. Additionally, the hydrophilic functional group may include a thiol group, amine oxide group, sulfoxide group, phosphate group, sulfate group or salts thereof; or hydrophilic groups of polyoxyethyelne or polyoxypropylene.
- hydrophilic silane examples include IM-aminoethyl gamma-aminopropyl trimethoxysilane, N-aminoethyl gamma-aminopropyl triethyoxysilane, gamma-aminopropyl trimethoxysilane, gamma-aminopropyl triethoxysilane, gamma- glycidoxypropyl trimethoxysilane, gamma-glycidoxypropyl triethoxysilane, gamma- isocyanopropyl triethoxysilane, gamma-isocyanopropyl trimethoxysilane or mixtures thereof.
- the organosilane is used to treat the particles to prepare particles with a balance of hydrophobicity and hydrophilicity. They can be used suitably in an amount of 1-50 wt % based on 100 wt % of the particle content. This content varies with the chemistry and surface area of the particles to be treated. Nano sized particles may require higher quantities of silanes while micron sized particles may require lower quantities. Where fluorosilanes are used the slurry or sol-gel treatment may be preferred as compared to the heat treatment to avoid discoloration/ graying of the white particles such as titanium dioxide.
- insitu surface treatment can also be performed.
- organosilicones/ silanes such as Dow Corning 3074, Silres SY 231 from Wacker, Protectosil SC concentrate, Dynasylan F8261, Tegophobe 1500, 1401, 1505 etc from Evonik Degussa can be used to impart hydrophobicity to the pigments and extenders and subsequently to the paint film. Further one or more of the hydrophobic organosilicones/silanes can be used which are described above.
- organosilicones such as Dow Corning QP8 5314, Tegophobe 1650, Wacker BS 45 etc. can be used. Further one or more of the hydrophilic organosilicones/silanes can be used which are described above. These silicones are added during the paint making process to functionalize the particles in-situ.
- the hydrophobic agent may be present in the aqueous composition in a range of from about 5 to 80% by weight of polymeric dispersion.
- the hydrophilic agent may be present in the aqueous composition in a range of from about 10 to about 90% percent by weight of the polymeric dispersion. It is preferred to have a combination of both types of silicones (hydrophilic and hydrophobic) to get a balance of hydrophobicity and hydrophilicity and attain a coating that exhibits good resistance to dirt lines, streaks and water spots.
- the hydrophobic organosilicone/silane has a contact angle hysteresis of ⁇ 10 degrees (average of 2 readings for 10 and 20 pL of water) when applied on an aerated cement block.
- the hydrophilic organosilicone/silane has a contact angle hysteresis of > 10 degrees when applied on an aerated cement block (average of 2 readings for 10 and 20 pL of water).
- One or more organosiloxanes are present as part of said continuous phase, the total organosiloxane being present in an amount of 10 to 90% of the weight of binder.
- the mass ratio of the hydrophobic silicone to the hydrophilic silicone is in the range 10/90 to 60/40.
- PVC pigment-volume concentration
- each particle will be entirely enclosed by binder. This will also be the case with increasing particle concentration up to a certain limit at which the amount of binder ' is just sufficient to enclose all of the particles.
- the point where the amount of binder is exactly sufficient for such enclosure is called the critical pigment-volume concentration, CPVC.
- CPVC critical pigment-volume concentration
- the dry paint film may contain voids and become porous and generally permeability, strength and gloss may hereby be influenced unfavourably; in case of outdoor paints the durability may become unsatisfactory.
- Prior art uses organic silicon compounds in combination with the binder to render paints watertight or hydrophobic.
- paints with only hydrophobicity or superhydrophobicity lead to inefficient and uneven cleaning of dirt form the surface of coatings with rain water. This leads to dirt lines and dirt patches which eventually leads to microbial growth.
- it is important according to the current invention to use a combination of hydrophobic and hydrophilic silicones in a certain ratio with a specific binder, particular PVC and specific particle size which lead to surfaces with very high durability and good aesthetic appearance benefitting from combined superhydrophobic and superhydrophilic cleaning effects.
- the composition has a pigment volume concentration (PVC) in a range of from about 40 to 90 % and preferably in the range of 60 to 80%. Said concentration is under the practical CPVC as tested by the curling test or other suitable tests known in the art, wherein the benefit of operating below the practical CPVC (practical pigment volume concentration) could be derived by careful formulation aided by the use of dispersion polymers as compared to latex polymers in combination with functionalized particles that eventually generates coatings also having high durability and good antimicrobial properties at reduce dosages of biocides.
- PVC pigment volume concentration
- the aqueous composition has a solid content in a range of from about 25 to about 65 percent, preferably in a range of from about 35 to about 55 percent. Solids content greater than about 65 percent may be less desirable in that there would be a difficulty in application and a loss of storage stability.
- the aqueous composition has a pH in a range of from about 5 to about 10, preferably in a range of from about 6 to about 9.0.
- the water-based compositions are preferably free of volatile organic compounds.
- the water-based compositions have a calculated volatile organic content of ⁇ 100 g/liter and more preferably ⁇ 50 g/liter as per Green Seal standard GS-i l.
- volatile organic chemical as described herein is defined as an organic compound having a boiling point less than 280 °C at ambient pressure. Solvents "
- non-aqueous liquids such as ketones, glycol ethers, glycol ether acetates, alcohols, aliphatic hydrocarbon solvents, polydimethylsiloxane, cyclic polydimethylsiloxane, aromatic hydrocarbon solvents, tetrahydrofuran, acetic acid, acetates, and glycols.
- the preferred solvents are acetone and isopropyl alcohol.
- a non-VOC material may be selected or a VOC exempt material may be selected to produce non-VOC hydrophobic particle dispersion in water.
- Example of non VOC solvents are Ecosoft PB form Dow, dimethyl carbonate, tertiary butyl acetate, Optifilm enhancer 300 from Eastman, Triethylene glycol etc.
- the solvent is present at a level of less than 50 percent by weight based on the total weight of the composition, and preferably in an effective amount of between 0.001 to 50 percent by weight, more preferably between 1 to 5 percent by weight based on the weight of the total composition.
- Suitable examples of less volatile organic solvents are those with lower vapor pressures, for example those having a low vapor pressure which include, but are not limited to: dipropylene glycol n-propyl ether, dipropylene glycol t-butyl ether, dipropylene glycol n- butyl ether, tripropylene glycol methyl ether, tripropylene glycol n-butyl ether, diethylene glycol propyl ether, diethylene glycol butyl ether, dipropylene glycol methyl ether acetate, diethylene glycol ethyl ether acetate, and diethylene glycol butyl ether acetate.
- the composition may also comprises one or more organic solvents which are capable of dissolving the dispersion polymer.
- Suitable solvents are, for example, monohydric alcohols having 1 to 4 C atoms and mono-, di- and trialkylene glycol monoalkyl ethers, wherein the alkylene group usually contains 2 or 3 C atoms and the alkyl group 1 to 4 C atoms.
- the aqueous composition may contain at least one co-solvent. When present in the composition, the co-solvent is a means to improve film coalescence at low temperatures of below 10°C, and improve sprayability and substrate wetting.
- cosolvents include Dowanol DPnB, PnP and PPH from Dow Chemical, Texanol from Eastman Chemical, Glycol Ether PB, EB, and DPM from Ashland Chemical.
- Examples of preferred cosolvents include Dowanol DPnB from Dow Chemical and Texanol from Eastman Chemical.
- the co-solvent may be present in the aqueous composition in a range of from about 0.20 percent by weight to about 4.0 percent by weight, preferably from about 0.40 percent by weight to about 2.0 percent by weight based on the total weight of the composition. Alternatively, co-solvent content less than about 0.20 percent by weight may be less desirable in that it would be insufficient for insuring good film formation at low temperatures.
- the aqueous composition may contain at least one UV absorbing component.
- the UV absorbing component is used as a means to provide weather protection for the film forming polymer and antimicrobials used in the product by protecting them against degradation caused by UV radiation.
- Useful UV absorbers include conventional zinc oxide, nano zinc oxide, titanium dioxide etc.
- the nanometer sized zinc oxide particles are broad spectrum UV absorbers (UV-A and UV-B) which is not the case for micro fine Ti02 and organic UV absorbers. It also has anti-bacterial properties and is mildew resistant.
- the UV absorbing component may be present in the aqueous composition in a range of from about 0.1 percent by weight to about 50 percent by weight of the dry composition.
- UV absorbing components useful include Tinuvin 292 1130, and 5151 from Ciba Specialty Chemical, These UV absorbing components may be present in the aqueous composition in a range of from about 0.10 percent by weight to about 5.0 percent by weight of the composition.
- the formulation contains at least one or more biocides which can be in liquid or solid form. Suitable types of incan preservatives, antifungal agents and anti algal agents and their contents can be decided by carrying out tests to determine a level required for long term protection depending upon the degree of exposure. Some biocides which can perform a multifunctional role of antifungal and antialgal can also be used. Antibacterial film properties can be conferred by suitable antibacterial agents such as silver etc. Further, encapsulated biocides can also be used.
- the aqueous composition contains at least one algaecide.
- Useful algaecides include Terbutrin, Rocima 344, Rocima 350, Rozone 2000, etc.
- the algaecide (active content) is present in the aqueous composition in a range of from about 0.05 percent by weight to about 3.0 percent by weight, preferably from about 0.10 percent by weight to about 2.0 percent by weight based on the total weight of the composition.
- the aqueous composition contains at least one fungicide.
- Useful fungicides include Zinc and Sodium Omadine from Arch Chemical, Rozone 2000, Rocima 200, Rocima 63, and Skane M- 8 from Rohm & Haas, Mergal S-90, Polyphase 678, and Mergal S-89 from Troy Chemical, and Fungitrol 720 from ISP Corp.
- the fungicide (active content) is present in the aqueous composition in a range of from about 0.05 percent by weight to about 3.0 percent by weight, preferably from about 0.10 percent by weight to about 2.0 percent by weight based on the total weight of the composition.
- a biocide such as DANTOGARD, TROYSAN 395, Nipacide CFX, Thor MBS etc. can be used as a preservative in the product.
- the preservative provides a useful shelf life to the product.
- the biocide preservative is added in an effective amount to preserve the composition product and ranges from 0.01 to 1.0 percent by weight, and more preferably in a range of from 0.05 to 0.9 percent by weight.
- biocides may be required in other to inhibit the growth of all of them.
- the biocides have low water solubility (less than about 50 parts per million), excellent resistance against hydrolysis, be resistant to degradation by UV where exposed to sunlight (unless sufficient UV absorbers are used) and should be free from formaldehyde.
- Surfactants may also be used to help wet the surface in order to form a uniform coating.
- Surfactants can be nonionic, cationic, amphoteric, or anionic in nature.
- the aqueous composition contains at least one dispersant.
- the dispersant is a means to disperse the pigments and prevent agglomeration on product aging.
- Useful dispersants include BYK 190, 191, 194 from BYK Chemie, AMP-95, Orotan 850, Orotan 731 etc from Dow Chemical Company.
- the dispersant is present in the aqueous composition in a range of from about 0.10 percent by weight to about 1.0 percent by weight, preferably from about 0.20 percent by weight to about 0.5 percent by weight based on the total weight of the composition. Dispersant content greater than about 1.0 percent by weight may be less desirable in that it could adversely affect viscosity stability and water resistance of the dried film. Alternatively, a dispersant content less than about 0.10 percent by weight may be less desirable in that the product would show poor viscosity stability on aging, pigment agglomeration.
- the aqueous composition contains at least one surfactant.
- the surfactant is a means to lower surface tension for improved surface wetting, pigment dispersion and polymer stability.
- Useful surfactants include Triton X-100 and Triton X-405 from Dow Chemical, Surfynol series from Air Products, Cresmer series from Croda.
- the surfactant is present in the composition in a range of from about 0.10 percent by weight to about 2.0 percent by weight, preferably from about 0.20 percent by weight to about 1.0 percent by weight based on the total weight of the composition. Surfactant content greater than about 2.0 percent by weight may be less desirable in that it would hamper the dried film's water resistance and durability.
- a surfactant content less than about 0.10 percent by weight may be less desirable in that it would not sufficiently lower the surface tension for adequate surface wetting and aiding in pigment dispersion.
- suitable surfactants include non-ionic surfactants having an HLB value of from between 9-13, branched ethoxylated alcohols, linear ethoxylated alcohols, and silicone surfactants. These surfactants are sold under the trade names of Tomah, Triton, Surfonic, Igepal, Alfonic, Rhodasurf, Synperonic etc.
- non-ionic wetting agent it is preferred to have certain ratios of the non-ionic wetting agent as compared to the ionic dispersing agent in the composition as shown in example number 56 to achieve a good accelerated storage stability (studied at 55°C for 30 days).
- other polymer and surfactant materials known in the art may also be suitably • employed in the inventive treatment compositions provided that they do not negatively impact the performance of the coatings.
- composition passes at least 5 cycles of freeze thaw stability (-15 deg C to 30 deg C).
- the aqueous composition may contain at least one fluorosurfactant. These are available from DuPont and 3M. It is preferred that the fluorosurfactant contain less than 6 carbon atoms and can be either ionic or non-ionic.
- the aqueous composition may contain at least one rheological agent.
- Useful thickeners include Rheolate 278, Rheolate 244, Rheolate 300 from Elementis Specialties, Acrysol DR-1, Acrysol DR-72, Acrysol DR-73, Acrysol ASE-60, Acrysol TT-615, Acrysol TT-935, Acrysol RM- 5, Acrysol RM-6000, Acrysol RM-5000, Acrysol RM-2020NPR, Acrysol; RM-8W, Acrysol STC- 275, Acrysol RM-825 from Dow, Coapur 6050 form Coatex.
- Inorganic thickeners such as bentone Lt, Laponite SD, laponite RD can also be used.
- the thickener may be present in the aqueous composition in a range of from about 0.10 percent by weight to about 3.0 percent by weight, preferably from about 0.20 percent by weight to about 1.0 percent by weight based on the total weight of the composition.
- the aqueous composition has a viscosity in a range of from about 100 to 1000 grams on the stormer viscometer. A viscosity greater than about 1000 grams may be less desirable in that it would not be easy to handle. Alternatively, a viscosity less than about 100 grams may be less desirable in that it would run down on vertical surfaces providing reduced coat weight and would tend to show separation in the container on standing before it could be applied.
- Suspending agents may optionally be included in the inventive treatment compositions to improve the suspension and/or dispersion properties of the inventive compositions.
- Polymer type suspending agents include anionic, cationic and nonionic polymers. Examples include, but are not limited to: vinyl polymers such as, cross linked acrylic acid polymers, cellulose derivatives and modified cellulose polymers such as, methyl cellulose, ethyl cellulose, hydroxyethyl cellulose, hydroxypropyl methyl cellulose, nitro cellulose, sodium cellulose sulfate, sodium carboxymethyl cellulose, crystalline cellulose, cellulose powder, polyvinylpyrrolidone, polyvinyl alcohol, guar gum, hydroxypropyl guar gum, xanthan gum, arabia gum, tragacanth, galactan, carob gum, karaya gum, carrageen, pectin, agar, starch (rice, corn, potato, wheat), microbiological polymers such as, dextran, succinoglucan, pull
- the aqueous composition may contain at least one defoamer.
- Useful defoamers include mineral oil, silicone defoamers, etc. Examples of preferred defoamers include BYK -012, BYK-020, BYK-022, Dapro-4164, Dapro 7005, Dapro-7010.
- the defoamer may be present in the composition in a range of from about 0.05 percent by weight to about 1.0 percent by weight, preferably from about 0.1 percent by weight to about 0.50 percent by weight based on the total weight of the composition.
- the aqueous composition may contain at least one freeze/thaw stabilizer.
- Useful freeze/thaw stabilizers include ethylene glycol, propylene glycol and triethylene glycol.
- the freeze/thaw stabilizer may be present in the aqueous composition in a range of from about 0.50 percent by weight to about 4.0 percent by weight, preferably from about 1.0 percent by weight to about 3.0 percent by weight based on the total weight of the composition. Freeze/thaw stabilizer content greater than about 4.0 percent by weight may be less desirable in that it would make the composition too water sensitive and the drying time too long and increase the volatile organic content of the aqueous composition.
- Colorants Organic or inorganic colorants can be used. Some examples of colorants suitable for use include naphthol, pyrazalones, quinacridones, phthalocyanines, pigments including titanium nickel yellow, limonites, haematites, magnetites, micaceous oxides of iron, iron ferrites and Prussian blue. Further effect pigments may also be used to create special effect finishes with wettability switching properties e.g. Sumica Pearl (Bright gold) available from Sudarshan Chemical Industries Ltd.
- the coating can have a dry film thickness of 1 to 200 microns and preferably from 40 to 120 microns. If the thickness is less than 1 micron, the film hardness is poor. If the thickness is greater than 200 microns, the coating layer has poor flexibility, resulting in generation of cracking in the coating film.
- the aqueous composition can be formed using conventional blending and mixing techniques. Preferably, water is added first, then the t ickeners, surfactants, dispersants, and defoamer, followed by the particles, silicones, then the polymer and other additives using mechanical agitation.
- the water-based composition can be either spray, brushed, or paint roller applied on the construction site to the construction surfaces, or supplied as an article to the construction site by pre-application to construction panels, such as oriented strand board, plywood, gypsum based sheeting, and other sheeting materials. It can also be applied by dip coating and spin coating.
- the wet-coated substrate formed in the method of the present invention may be dried using conventional techniques including ambient environmental drying, forced air drying, and forced air/ thermal drying.
- the composition is dried under ambient conditions.
- the coating compositions may be processed both at room temperature and at temperatures above or below room temperature, for example, at temperatures in the range from 0 °C to 55 °C. Substrates
- the coating When applied to a surface, the coating has good adhesion to masonry surfaces, painted and many other surfaces such as concrete, cementitious, plaster, cellulosic, wood, one or more polymer, dry or wet surfaces, surfaces exposed to weathering on at least one surface of the substrate.
- the adhesion of the coating to the substrate may be produced by chemical bonding, or else by physical forces.
- the contact angle can be measured using a goniometer and the CAH can be measured using the dynamic sessile drop method. 20 pL drop of water is taken, 10 ⁇ _ of diiodomethane and 15 pL of hexadecane is taken for surface energy measurements by the Owens-Wendt equation.
- the contact angle of water on the coated surface made according to the present invention may be difficult to measure with conventional means because the water droplet bounces or runs off the surface when applied.
- the surface energy for one embodiment of the coating is ⁇ 10 mN/m.
- the self-cleaning surfaces of the invention have a roll-off angle of less than 20 degrees, preferably less than 10 degree, even more preferably less than ⁇ 5 degrees in one state.
- the water contact angles are preferably above 100 degrees, more preferably over 120 degrees, even more preferably superhydrophobic i.e. difficult to measure by conventional techniques.
- the coating has a surface energy of ⁇ 40 mN/m, preferably ⁇ 20 mN/m and most preferably ⁇ 10 mN/m and is recoatable favouring application of a second coat to enhance film build- up to attain desired opacity/ hiding/ whiteness.
- a coating formed from the coating composition of the present invention has good durability when exposed outdoors to extreme weather for a time period of a few months to several years. Stability is determined by observing that the unwettability, by measuring the contact angle of a droplet on the surface, has not diminished over the course time being exposed to the environment in addition to the colour difference after exposure to Atlas and QUV testing after 2000 hours as illustrated in example 41.
- Test methods The following test methods were used to quantify the properties of the inventive water- based compositions.
- a pig bristle brush (DIN specification) [Hog hair brush], (Brush holder, brush, packing mat.) Total weight: 430 +/- 20 gms.
- a film of WFT (wet film thickness) 6 mil was applied on a Leneta P-121 ION dull black plastic panels (165 x 432 x 0.25 mm). The panels are air dried in horizontal position for seven days in a dust free open room.
- Scrub medium consisting of water, thickener, Liquor ammonia, surfactant, Silica 400 mesh and preservative.
- the panel was placed and clamped in the tray ' of washability apparatus.
- the brush was soaked with 1% solution of SLS.
- the brush was removed and shaken vigorously to remove any excess SLS solution.
- the brush was mounted in the holder.
- the scrub medium was stirred with a stainless steel spatula and lOg was spead evenly on the panel.
- the brush was placed at one end of the path and attached to the guide cables.
- the panel was wet with 5 ml of distilled water in the path of the brush.
- Algae inoculum preparation Two species of algae, Chlorella and Oscillatoria, were cultured separately. Aerated Cement Blocks (ACBs) of similar surface profile were used for a set of paints under study. The ACBs were sanded with emery paper ⁇ Ho. 180). Two coats of paint were applied on the ACBs with the help of a brush, on the cut surface barring 1 cm area on the top portion of the side. Also paint was applied on the bottom side of the ACBs. During first coat, 3 ⁇ 0.1 gm was deposited by wet wt.
- ACBs Aerated Cement Blocks
- the specimen condition in the can was examined for settling, gelation, coagulation, lumpiness.
- the paint specimens were stirred and the consistency on stormer viscometer at 30 + 1 °C was determined.
- the paint was applied to a cement concrete block of 6"x6" by brushing.
- the performance of the test specimen with the control was compared in terms of dried film properties, such as gloss and film integrity. The paint will be considered to have passed this test if no significant difference in performance/ behaviour is observed between control and test samples.
- the present invention is illustrated hereunder in greater details in reference to the non- limiting exemplary illustrations and figures.
- Example 1 Surface treatment/ Functionalization of Ti0 2 with Dynasylan F8261 by heat treatment/ sol-gel treatment
- Ti0 2 was both hydrophobic as well as oleophobic as it floats on water and a bed of the Ti02 powder spread on a glass panel is able to repel both water and oil. Heating the pigment with Dynasylan F8261 caused greying of Ti0 2 which was not desired as Ti0 2 is added to paint and coatings for its whiteness and hence the greying is undesirable.
- Example 2 Surface treatment/ Functionalization of Ti0 2 with VTMO by heat treatment/ sol- gel treatment
- Ti0 2 R902 pigment (20 g) was carried out with VTMO (vinyl trimethoxy silane, 10 g) in 20 g of isopropanol and in 1 g of 25% solution of liquor ammonia.
- VTMO vinyl trimethoxy silane
- the functionalization was carried out at room temperature (for 4 hours) using a sol-gel process instead of employing high temperatures as discussed above.
- the results reveal that the treated Ti0 2 pigment showed good hydrophobicity (floating on water) which may be used in the cdating composition.
- Example 3 Surface treatment/ Functionalization of amorphous nanosilica with VTMO and Dynasylan F8261 by sol-gel treatment
- Cabosil M5 amorphous nanosilica (200 nm - 300 nm aggregates with a primary particle size of 14 nm in diameter) (5g) was treated with VTMO (2.5 g) in isopropanol (1.8 g) and 25% liquor ammonia solution (0.7g) using a sol-gel method at room temperature (30°C) for 5 hours and 30 minutes.
- Example 4 Selection of the treatment process of the pigments present in the aqueous coating composition of the present invention, and selection of the material for functionalization to achieve a balance of hydrophobicity and hydrophilicity.
- Dynasylan F8261 is preferred over functionalization with VTMO as VTMO imparts only hydrophobicity whereas Dynasylan F8261 imparts both hydrophobicity and oleophobicity.
- Table 1 below reveals the surface treatments carried out with monomeric and oligomeric silicones for Titanium dioxide and other fillers. These were carried out by heat treatment/ sol gel method. Further they also represent some of the particles and their sizes that may be used in the present invention.
- Fig 3 and 4 clearly shows the image of untreated and treated Ti0 2 with silicone Siires SY231, wherein it is clearly seen in Fig 4 that a fine 27 nm layer of silicone is bonded to the pigment.
- All of the above treated pigments and extenders as per Table 1 were found to be either hydrophilic (disperses in water) or hydrophobic (floating on water) or partly floating and partly dispersing in water.
- the Ti02 treated with 10% 3-glycidoxypropyltrimethoxysilane showed a lower degree of hydrophobicity (the treated materials are getting dispersed in water and not floating on it) as compared to treatment with Siires SY231 and may be considered as a hydrophilic treatment of silicone.
- Other such hydrophilic silanes are those with the amine or isocyanate group.
- nano silica treated with 25% 3-methacryloxypropyltrimethoxysilane the treated silica was not having hydrophobic character (treated for 5h 30 minutes at upto 80°C. However, when checked after a period of 1 month, the particles showed good hydrophobicity (floating on water). Treatment of nanosilica with 2% 3- methacryloxypropyltrimethoxysilane did not lead to hydrophobicity. However, the Ti02 treated with 2% 3-methacryloxypropyltrimethoxysilane, showed good hydrophobicity immediately after heat treatment. Hence it is possible to achieve particles with varying levels of hydrophobicity and hydrophilicity, a balance being preferred to achieve the desired coating composition.
- a mortar and pestle can be used for crushing the lumps and the particles can be passed through a suitably sized sieve.
- the extent of hydrophobization varied with the particle being treated, the quantity of treating agent and the time and temperature of the treatment reaction. Higher temperatures were used for treatment with oiigomeric silicones (siires sy 231) as compared to the monomeric silanes.
- a plurality of particles of different particle sizes can be used for the coating composition with different hydrophobic and hydrophilic silicone treatments provided they are incorporated with the polymer in certain ratios which are discussed below.
- rutile and anatase grades of titanium dioxide are available such as R902, R902 plus, R 900, R931 etc that can be treated as per the process discussed above.
- Hydrophilic grades of nano silica are available such as Cabosil M5 from Cabot or Aerosil R200 (average particle size of 12 nm) from Evonik that can be either heat treated or treated by sol-gel process as discussed above.
- Example 5 Preparation of a solvent borne resin
- a solvent borne resin was prepared using hydrophobic monomers wherein 38 pbw of styrene, 40 pbw of n-BMA (n-butyl Methacrylate), 20 pbw of MPTMS (3- methacryloxypropyl trimethoxy silane were copolymerized in the presence of 2 pbw of Zonyl TM from DuPont (perfluorinated monomer), 66 pbw of o-xylene, 1.2 pbw of tbpb (tertiary butyl perbenzoate) initiator and 4 pbw of triethylorthoformate (moisture scavenger) at 135°C for 6 hours with drop wise monomer addition for 3 hours followed by digestion for 3 hours.
- hydrophobic monomers wherein 38 pbw of styrene, 40 pbw of n-BMA (n-butyl Methacrylate), 20 pbw of MPTMS
- the preparation of the resin of example 5 was repeated with tertiary butyl methacrylate as a substitute for styrene, wherein the said resin was prepared by reacting 20 pbw of tertiary butyl methacrylate, 18 pbw styrene, 40 pbw n-BMA (n-butyl Methacrylate), 20 pbw MPTMS (3-methacryloxypropyl trimethoxy silane) in the presence of 2 pbw Zonyl TM (perfluorinated monomer), 66 pbw o-xylene, 1.2 pbw tbpb (tertiary butyl perbenzoate) initiator and 4 pbw triethylorthoformate.
- the resin cast on a glass panel showed similar water repellency/ beading as that of a solvent borne resin of Example 5, wherein the similar repellency to water droplets was also observed on the coating.
- Another batch with 4 pbw Zonyl TM showed similar results.
- the Contact angle hysteresis values for water were more than 25° i.e. complete roll off of water was not observed.
- Example 7 Preparation of amine functional water borne/ water reducible resin in absence of a fluoro monomer with a high content of amine monomer
- a cationic water borne resin was prepared by reacting methyl methacrylate 60 pbw, butyl acrylate (25 pbw), N,N-dimethylaminopropylmethacrylamide (15 pbw), cellosolve (43 pbw), tert-butyl perbenzoate (1.2 pbw), water (100 pbw), acetic acid (6 pbw).
- the reaction was carried out at 130 °C for 6 hours with drop wise monomer addition for 3 hours followed by digestion for 3 hours. Subsequently water and acetic acid was added to disperse the resin into water at 50°C resulting into a 40% solids resin with an amine value of 99 mg KOH/g.
- this resin Since this resin has an amine backbone, the same is responsive to stimuli of change of temperature as well as humidity.
- Example 8 Preparation of cationic water borne/ water reducible resin by incorporation of a fluoro monomer and with high content of amine monomer
- a cationic water borne resin was prepared by reacting tertiary butyl methacrylate 60 pbw, Zonyl TAN (perfluorinated monomer, 25 pbw), DMAPMA ( ⁇ , ⁇ -dimethyl aminopropyl methacrylamide, 15 pbw), cellosolve (43 pbw), tert-butyl perbenzoate (1.2 pbw), water (100 pbw), glacial acetic acid (6 pbw).
- the reaction was carried out at 130 °C for 6 hours with drop wise monomer addition for 3 hours followed by digestion for 3 hours. Subsequently water and acetic acid was added to disperse the resin into water at 50°C resulting into a 40% solids resin with an amine value of 99 mg KOH/g.
- this resin has an amine backbone, the same is responsive to stimuli of change of temperature which is demonstrated in the example 27. In addition it is responsive to the stimulus of humidity.
- Example 9 Preparation of acid functional water borne/ water reducible resin in the absence of fluoro monomer but with a high content of acid monomer
- the anionic water reducible resin was prepared by the reaction of MMA (methyl methacrylate, 65 pbw), BA (butyl acrylate, 29.5 pbw), MAA (glacial methacrylic acid, 5.5 pbw), cellosolve (43 pbw), tert-butyl perbenzoate (1.2 pbw), water (100 pbw), triethylamine (4.5 pbw).
- the reaction was carried out at 130 °C for 6 hours with drop wise monomer addition for 3 hours followed by digestion for 3 hours. Subsequently water and triethylamine was added to disperse the resin into water at 50°C resulting into a 40% solids resin with an acid value of 36 mg KOH/g.
- Example 10 Preparation of acid functional water borne/ water reducible resin by incorporation of a fluoro monomer with a high content of acid monomer.
- an anionic water borne resin was prepared by reacting tertiary butyl methacrylate 64.5 pbw, Zonyl TAN (perfluorinated monomer, 30 pbw), MAA (glacial methacrylic acid, 5.5 pbw), cellosolve (43 pbw), tert-butyl perbenzoate (1.2 pbw), water (100 pbw), triethylamine (4.5 pbw). The reaction was carried out at 130 °C for 6 hours with drop wise monomer addition for 3 hours followed by digestion for 3 hours.
- this resin is devoid of amine backbone and is acid functional instead, the same is responsive to stimuli of change of pH and humidity.
- Example 11 Selection of type of resin in the aqueous coating composition of the present invention
- the surface structure is not conducive towards obtaining superhydrophobicity.
- the solvent borne polymers are less preferred over water borne coatings especially in decorative applications as environmental regulations are driving the use of low VOC (volatile organic content) coatings which contain minimum level of solvents.
- water can be used to clean the brushes used by the painters for application thereby being relatively easier to apply in addition to being environmentally friendly.
- Example 12 Evaluation of incorporation of only one kind of functionalized particle in making paint/ coating composition with water borne resin
- treated silica of Example 3 was used in high doses (50:50 particles: binder) in combination with either the resin of Example 8 or the resin of example 10 to prepare the paint/ coating composition. It was found by way of the present invention that paint/ coating composition obtained from the above experiments though showed thermoresponsive and pH responsive character none of the paint/ coating compositions obtained of the above experiments imparted complete water rolling property, even when the above particles are specifically present in combination with the resins of examples 8 and 10.
- Example 13 Evaluation of one. and two kinds of functionalized particles in making paint/ coating composition with water borne resin
- Blends of treated Ti02 micron sized treated with silres sy231) and treated Cabosil M5 fumed silica (nano sized treated with VTMQ) have been evaluated with different resins of experiments 7, 8, 9 and 10.
- Ratio of Micron to nano sized particles 4.5
- the coatings were made by simple mixing, diluted with water and applied by brush on fiber cement board panels coated with Asian Paints water based exterior primer (Asian paints exterior primer is based on a modified acrylic emulsion polymer with titanium dioxide along with fillers like calcium carbonate, clays, silicates with a PVC in the range 50 to 80%. It contains additives such as rheology modifiers, pigment dispersing and wetting agents, film and in can stabilizers.), and allowed to dry under ambient conditions for 7 days. It was observed that a combination of treated Ti02 and treated silica when mixed in water reducible resins 8 and 10 gave contact angle hysteresis ⁇ 10°.
- Charcoal powder when deposited on the coating gets cleaned with a stream of water giving the benefit of superhydrophobic cleaning.
- the coating subsequently gets completely wet offering the benefits of superhydrophilic cleaning.
- the balance of hydrophilicity and hydrophobicity of the polymer, pigments and extenders with a high content of particles/ binder may be responsible for this behavior.
- the coating was also oleophobic due to the presence of fluorine based monomer.
- the pigmented coating of resin 8 when placed under a continuous stream of water shows complete wettability. However, as the coating was dried overnight at ambient conditions it regained its superhydrophobicity (CAH ⁇ 10°).
- the coating showed a CAH ⁇ 10° and also a stimuli responsive behaviour.
- Example 15 Further evaluation of two kinds of functionalized particles in making paint/ coating composition with water borne resin .
- Ratio of Micron to nano sized particles 4
- Ratio of particles to polymer 1.094
- the coating showed a CAH ⁇ 10° and also a stimuli responsive behaviour.
- Example 16 Preparation of amine functional water borne/ water reducible resin using styrene and 2-ethyl hexyl acrylate in place of MMA and BA in absence of a fluoro monomer with a high content of amine monomer
- a water borne resin was prepared by reacting styrene 60 pbw, 2- ethyl hexyl acrylate (25 pbw), N,N-dimethylaminopropylmethacrylamide, (15 pbw), cellpsolve (43 pbw), tert- butyl perbenzoate (1.2 pbw), water (100 pbw), acetic acid (6 pbw).
- the reaction was carried out at 130 °C for 6 hours with drop wise monomer addition for 3 hours followed by digestion for 3 hours. Subsequently water and acetic acid was added to disperse the resin into water at 50°C resulting into a 40% solids resin with an amine value of 99 mg KOH/g and a pH of 6.
- Ratio of Micron to nano sized particles 4
- Ratio of particles to polymer 1.094
- the coating did not show rolling of water and showed a CAH>20°
- Example 17 Preparation of amine functional water borne/ water reducible resin using isobutyl Methacrylate, t-butyl Methacrylate and fluoro monomer with a high content of amine monomer
- a water borne resin was prepared by reacting t-butyl Methacrylate (30 pbw), isobutyl Methacrylate ( 30 pbw), N,N-dimethylaminopropylmethacrylamide, (15 pbw), zonyl TAN (25 pbw), cellosolve (43 pbw), tert-butyl perbenzoate (1.2 pbw), water (100 pbw), acetic acid (6 pbw).
- the reaction was carried out at 130 °C for 6 hours with drop wise monomer addition for 3 hours followed by digestion for 3 hours. Subsequently water and acetic acid was added to disperse the resin into water at 50°C resulting into a 40% solids resin with an amine value of 99 mg KOH/g and a pH of 6.
- Ratio of Micron to nano sized particles 4
- Ratio of particles to polymer 1.094
- the coating showed a CAH ⁇ 10° and also a stimuli responsive behaviour indicating the importance of selective monomers (in this example isobutyl Methacrylate, t-butyl methacrylate and perfluorinated monomer) in the composition.
- Example 18 Preparation of acid functional water borne/ water reducible resin in the presence of fluoro monomer but with a high content of acid monomer
- the anionic water reducible resin was prepared by the reaction of tert-butyl acrylate (67.4 pbw), zonyl TAN (25 pbw), MAA (glacial methacrylic acid, 7.6 pbw), cellosolve (43 pbw), tert-butyl perbenzoate (1.2 pbw), water (98.4 pbw), triethylamine (7.6 pbw).
- the reaction was carried out at 130 °C for 6 hours with drop wise monomer addition for 3 hours followed by digestion for 3 hours. Subsequently water and triethylamine was added to disperse the resin into water at 50°C resulting into a 40% solids resin with an acid value of 50 mg KOH/g.
- Example 19 Preparation of acid functional water borne/ water reducible resin in the absence of fluoro monomer but with a high content of acid monomer
- the anionic water reducible resin was prepared by the reaction of tert-butyl acrylate (58.5 pbw), 2-ethyl hexyl acrylate (35.5 pbw), MAA (glacial methacrylic acid, 6 pbw), cellosolve (43 pbw), tert-butyl perbenzoate (1.2 pbw), water (98.95 pbw), triethylamine (7.05 pbw).
- the reaction was carried out at 130 °C for 6 hours with drop wise monomer addition for 3 hours followed by digestion for 3 hours.
- Example 20 Preparation of acid functional solvent borne (unneutralized) resin in the presence of fluoro monomer but with a high content of acid monomer
- solvent borne (unneutralized) resin was prepared by the reaction of tert-butyl acrylate (53.3 pbw), 2-ethyl hexyl acrylate (17 pbw),- MAA (glacial methacrylic acid, 4.7 pbw), Zonyl TM (25 pbw), butyl cellosolve (43 pbw), tert-butyl perbenzoate (0.9 pbw).
- the reaction was carried out at 130 °C for 6 hours with drop wise monomer addition for 3 hours followed by digestion for 3 hours.
- the resin was discharged without neutralization at 70% solids in cellosolve for subsequent use for the preparation of a stimuli responsive coating with reversible wettability switching and CAH ⁇ 25°.
- Example 21 Further evaluation of two kinds of functionalized particles in making paint/ coating composition with water borne resin
- thermoresponsive aqueous coating/ paint composition was achieved due to the amine backbone in the resin exhibiting superhydrophobicity and superhydrophilicity with a low contact angle hysteresis of ⁇ 10 degrees having self cleaning behaviour as it helps the dirt get cleaned from its surface with the flow of rain water on the paint surface without leaving behind water spots, dirt lines or streaks only when the resin having a specific backbone of acrylic, fluorinated and amine/ acid is present in combination with both kinds of surface treated particles with a balance of hydrophobicity and hydrophilicity.
- experiment C the only difference was that .
- a pH and humidity responsive aqueous coating/ paint composition was achieved with other characteristics of the coating composition remaining the same.
- the coating can be brush applied on a cement/ concrete panel.
- the coating is recoatable and 2 coats can be applied on a waterborne primer.
- a self priming system can be used.
- Charcoal powder when deposited on the coating is cleaned with a stream of water giving the benefit of superhydrophobic cleaning.
- the coating subsequently gets completely wet offering the benefits of superhydrophilic cleaning.
- the balance of hydrophilicity and hydrophobicity of the polymer, pigments and extenders with a high ratio of particles/binder may be responsible for this behaviour.
- Example 22 Evaluation of stimuli responsive resin having the specific acrylic and amine/ acid backbone in combination with both functionalized Ti02 and nanosilica based pigments to achieve the desired contact angle hysteresis vis-a-vis the resin beyond the scope of the present invention
- Example 9 (lOg) devoid of the fluoro and tertiary butyl Methacrylate backbone in combination with the treated Ti02 of Example 1 (4 g), and silica of Example 3 (1 g) were used to prepare an aqueous paint composition.
- the resin of Example 9 (lOg) devoid of the fluoro and tertiary butyl methacrylate backbone in combination with the treated Ti02 of Example 2 (4 g), treated Silica of Example 3 (1 g) were used to prepare an aqueous paint composition.
- the aqueous coating composition under 22 D and E above failed to exhibit the desired superhydrophobicity (low contact angle hysteresis ⁇ 10 °) as compared to the aqueous coating compositions under Examples 21 A, B and C above, and hence it was thus found by way of the present invention that the backbone of the resin plays an equally important role to impart the desired superhydrophobicity and superhydrophilicity having self cleaning behaviour and low contact angle hysteresis of ⁇ 10 degrees when present in combination with two types of functionalized pigments as discussed above.
- superhydrophobic coatings can be prepared using externally treated combination of micron and nano sized particles in combination with a polymer; in this example an acrylic polymer with suitably selected monomers.
- Example 23 Evaluation of the ratio of functionalized particles of two different sizes present in combination with resin involving the selective acrylic fluorinated amine/acid backbone to attain very low contact angle hysteresis of ⁇ 10 degrees
- Ratio of particulate mixture (micron sized and nano sized particles) to polymer 0.9 to 2.5 (higher ratios being preferred for 2 coat application)"
- the CAH of ⁇ 25°, preferably ⁇ 20°, more preferably ⁇ 10° and more preferably ⁇ 5° is desired in one state and higher than 25° in the other switched state.
- the selective combination of hydrophobic and hydrophilic silicones with suitable acrylic, fiuorinated and hydrophilic backbone gives the desired durability and water resistance to the coating composition along with reversible hydrophilic-hydrophobic switching under variable environmental conditions including variations in at least one or more of temperature, pH, humidity or other external stimuli including light .
- Example 24 Preparation of acid functional water borne/ water reducible resin using tertiary butyl Methacrylate and fluoro monomer with a high content of acid monomer by dispersion polymerization
- the anionic water reducible resin was prepared by the reaction of t- BMA (t-butyl methacrylate, 42.4 pbw), Zonyl TM (50 pbw), MAA (glacial methacrylic acid, 7.6 pbw), butyl cellosolve (43 pbw), t-butyl perbenzoate (2.2 pbw), water (98.4 pbw), triethylamine (7.6 pbw).
- Example 25 Details of acrylic latex prepared using emulsion polymerization
- the emulsion film has a water contact angle of 74°, hexadecane contact angle of 2.9° and a surface energy of 36.9 ml ⁇ l/m.
- the blend of example 24 with the emulsion of example 25 shows the following surface energies. Thus a low surface energy was obtained even for a blend of the 2 polymers (emulsion and dispersion).
- Example 26 Evaluation of the ratio of dispersion to emulsion polymer involving the selective acrylic fluorinated amine/acid backbone in combination with the specific ratios of ⁇ micronized to nano sized particles to attain desired low contact angle hysteresis of ⁇ 10 degrees
- emulsion polymer has been used in 40/60 ratio
- Dispersion polymerization is known to a person skilled in the art wherein the polymer with acid or amine functionality is prepared in water compatible cosolvent such as cellosolve, carbitol or any other water compatible co solvent or other suitable solvent. After preparation of the polymer, amine such as liquor ammonia etc. or acid such as acetic acid etc. is added to neutralize the polymer (anionic or cationic) and water is added to disperse the polymer into water at a high speed (500 rpm using a stirrer). The dispersion polymers are typically free from surfactants.
- the low contact angle hysteresis ⁇ 10° was obtained above for 1 coat application in accordance with the invention, however for 2 coat applications higher ratios of total particles to binder and micron to. nanometer particles were required to achieve contact angle hysteresis ⁇ 10 degrees since the surface profile of the topcoat may change depending on the surface profile of the substrate/ undercoat.
- emulsion polymer having surfactants may still give low hysteresis provided a dispersion polymer is present in the system. Ratio of dispersion polymer to emulsion polymer (5/95 to 100% dispersion polymer is preferred).
- Emulsion polymers are not preferred in the composition of the present invention as the film formation mechanism coupled with the presence of surfactants increases the contact angle hysteresis values possibly due to a non-continuous film covering the pigment and extender particles.
- emulsion polymers Only emulsion polymers may be used, however, the CAH values will be higher, more than 20 degrees for emulsions of example 25 and 44 (shown in example 55 ) and further the time of switching from hydrophobicity to hydrophilicity will be decreased to under few minutes. Also if the emulsion has a high contents of external surfactants, water marks may appear on the paint. Polymerizable surfactants may be used to prepare emulsions (shown in example 44), however the CAH value will be higher for a given composition when compared with dispersion. Thus dispersion polymers are preferred.
- ком ⁇ онент to prepare a coating formulation can be present in the paint formulation and low hysteresis can be obtained.
- Other components include but not limited to thickeners, biocides, dispersing agents, defoamers, buffers etc. These components are known to those skilled in the art. This is shown in example number 34.
- the ratio of dispersion to emulsion polymers in the aqueous coating composition of the present invention also plays a role in determining the contact angle hysteresis.
- the adhesion of dirt particles to these surfaces is minimal because they touch only the tips of the structure leading to the dirt particles being only lightly deposited on the coating surface. The particles are easily washed away by water droplets, which roll off.
- Example 27 Characterization and study of the water-borne acrylic random copolymer dispersions in combination with the functionaiized pigments in accordance with the present invention
- a series of water-borne acrylic random copolymer dispersions comprising specialty monomers; tertiary butyl Methacrylate (TBMA) for hydrophobicity and perfluorinated monomer (Zonyl TAN/ Zonyl TM) for oleophobicity and improved dirt pickup resistance of coating were synthesized by conventional free radical polymerization as per examples 8 and 10.
- TBMA tertiary butyl Methacrylate
- Zonyl TAN/ Zonyl TM perfluorinated monomer
- copolymers can be acid or amine functional. These copolymers were processed with several functionaiized pigments to give stimuli responsive (pH/ temperature/ humidity) water-borne coatings as shown above.
- One of these coatings (cationic) was taken up for the study that was prepared by dispersing the treated pigments in the amino functional acrylic dispersions to get weight solids of about 40 % in water.
- the resulting coating composition was free flowing liquid that could be applied as such or with little dilution using the conventional brush, roller or airless spray application techniques.
- coatings were applied on glass panels of dimensions 6 x 4 square inches using a 100- ⁇ SHEEN draw-down applicator to form uniform films of about 50-jjm DFT.
- the coated panels were matured for 7 days and tested for contact angle measurements using a Digidrop Contact Angle Meter of GBX Surface Science Technologies make. The contact angle values were measured using the static sessile drop method after a time period of 60 seconds. An average of 5 readings taken at different parts of the coating is reported.
- the contact angle measurements were first carried out at ambient temperature of about 30°C. It was an interesting observation to note that the coating surface behaved superhydrophobic with contact angle hysteresis ⁇ ⁇ - 9 R ⁇ 10 degrees.
- a 5 ⁇ _ water droplet is pumped through a syringe needle, it touches the surface and becomes non- coaxial with the needle as soon as it begins to advance. The droplet then moves to the right and around to the back of the needle. As the water droplet is removed with the syringe, it remains stationary until it spontaneously dewets the surface and rises into the needle.
- the study of contact angle was also carried out using hexadecane as a probe.
- the minimum volume eluting from the syringe needle to form a droplet was 15 pL in the case of hexadecane due to its low surface tension.
- the contact angle value for hexadecane was found to be 98° at ambient temperature. The value clearly indicates that the coating is not just hydrophobic but also oleophobic in nature.
- Table 6 above shows the surface energies of the coating.
- the surface energy was calculated by the Owens-Wendt equation utilizing the contact angles of distilled water and hexadecane using Windrop++ software.
- the surface energy obtained for the coating is 5.3 mN/m.
- the switching behaviour from superhydrophobicity to superhydrophilicity is demonstrated from the following experiments.
- the coated glass panel was placed in a deep freezer at 0°C for 5 hours and then immediately subjected to contact angle measurement. It was possible to measure the contact angle with just a 5 pL water droplet with the angle reducing down to ⁇ 90°.
- the pigmented coating exhibits superhydrophilicity as a thin sheet of water covers the surface. This complete wetting of the surface with water enables cleaning of the surface thereby preventing dirt streak marks, which is associated with non-responsive superhydrophobic coatings.
- the switching behavior from superhydrophilicity to superhydrophobicity with the temperature variation was further substantiated by carrying out rheological studies of the liquid pigmented system.
- the rheological measurements were performed at low shear rates 0.001, 0.01, 0.1 and 1 s "1 .
- the viscosity values are plotted as a function of temperature in Figure 6.
- the rheograms show an initial decrease in viscosity with temperature followed by an anomalous increase between 20-50°C.
- the viscosity passes through a maxima at a low shear rate ( ⁇ 1 s "1 ) while at a higher shear rate (1 s "1 ) there is only a slight increase in viscosity before it again decreases.
- This interchain association results into the observed temperature independent viscosity behaviour because there are two opposing tendencies, which apparently seem to balance each other.
- the usual Arrhenius-like decrease in viscosity with increase in temperature is balanced by association driven increase in viscosity with increase in temperature.
- DSC Differential Scanning Calorimetry
- Figure 7 depicts an endotherm corresponding to the temperature region (30-55°C) where the coating is switching from hydrophilicity to hydrophobicity.
- the topography of the paint surface is further substantiated by AFM (Atomic Force Microscopy) studies.
- the AFM picture as shown in Figure 9 depicts high surface heterogeneity having a root mean square surface roughness of 210 nm.
- the AFM image of Figure 10 shows the thermoresponsive behaviour of the paint, wherein the images on the left are under hot condition and the images on the right are under cold condition.
- the stimuli responsive coating of the present invention can be thus used to make self- cleaning/ easy cleaning, anti-graffiti and stain resistant coatings wherein the said coatings are made by a simple, facile and economically viable process and can be applied by conventional techniques such as brush, roller and airless spray application.
- Example 28 Preparation Of acid functional water borne/ water reducible resin using tertiary butyl Methacrylate and fluoro monomer with a high content of acid monomer and a triacrylate crosslinker.
- the anionic water reducible resin was prepared by the reaction of t- BMA (t-butyl methacrylate, 42.4 pbw), Zonyl TM (perfluorinated monomer, 50 pbw), MAA (glacial methacrylic acid, 7.6 pbw), TMPTA (trimethylolpropane triacrylate, 0.5 pbw), butyl cellosolve (43 pbw), di-tert butyl peroxide (2.2 pbw), water (98.4 pbw), triethylamine (7.6 pbw).
- the reaction was carried out at 140 °C for 6 hours with drop wise monomer addition for 3 hours followed by digestion for 3 hours.
- Example 29 Preparation of acid functional water borne/ water reducible resin using tertiary butyl Methacrylate and fluoro monomer with a high content of acid monomer and a triacrylate crosslinker using an alternate initiator
- the anionic water reducible resin was prepared by the reaction of t- BMA (t-butyl methacrylate, 42.4 pbw), Zonyl TM (perfluorinated monomer, 50 pbw), MAA (glacial methacrylic acid, 7.6 pbw), TMPTA (trimethylolpropane triacrylate, 0.5 pbw), butyl cellosolve (43 pbw), t-butyl perbenzoate (2.2 pbw), water (98.4 pbw), triethylamine (7.6 pbw).
- Example 30 Preparation of acid functional water borne/ water reducible resin using tertiary butyl acrylate and fluoro monomer with a high content of acid monomer
- the anionic water reducible resin was prepared by the reaction of t- BA (t-butyl acrylate, 42.4 pbw), zonyl TM (50 pbw), MAA (glacial methacrylic acid, 7.6 pbw), butyl cellosolve (43 pbw), t-butyl perbenzoate (1 pbw), water (98.4 pbw), triethylamine (7.6 pbw).
- Example 31 Preparation of acid functional water borne/ water reducible resin using tertiary butyl methacrylate and fluoro monomer with a high content of acid monomer and high initiator content.
- anionic water reducible resin was prepared by the reaction of t- B A (t-butyl methacrylate, 42.4 pbw), MAA (glacial methacrylic acid, 7.6 pbw), Zonyl TM (50 pbw), butyl cellosolve (43 pbw), ditertiarybutyl peroxide (2.2 pbw), water (98.4 pbw), triethylamine (7.6 pbw).
- Example 32 Preparation of acid functional water borne/ water reducible resin using tertiary butyl methacrylate and fluoro monomer with a high content of acid monomer, multifunctional acrylate and a silane monomer
- anionic water reducible resin was prepared by the reaction of t- BMA (t-butyl methacrylate, 40.4 pbw), MAA (glacial methacrylic acid, 7.6 pbw), Zonyl TM (50 pbw), 3-methacryloxypropyltrimethoxysilane (0.5 pbw), Trimethylolpropanetriacrylate (0.5 pbw), butyl cellosolve (43 pbw), ditertiarybutyl peroxide (2.2 pbw), water (98.4 pbw), triethylamine (7.6 pbw).
- Example 33 Preparation of acid functional water borne/ water reducible resin using tertiary butyl methacrylate and fluoro monomer with a high content of acid monomer
- the anionic water reducible resin was prepared by the reaction of t- BA (t-butyl acrylate, 41.9 pbw), MAA (glacial methacrylic acid, 7.6 pbw), Zonyl TAN (50 pbw), Trimethylolpropanetriacrylate (0.5 pbw), butyl cellosolve (43 pbw), ditertiarybutyl peroxide (1 pbw), water (98,4 pbw), triethylamine (7.6 pbw).
- Example 34 Sample Paints prepared using the water-borne acrylic random copolymer dispersions in combination with the functionalized particles (pigments and extenders) in accordance with the present invention
- the water, dispersion polymer, dispersing agent, wetting agent, defoamer, glycol, biocides, dipentene, rheological additive are mixed on a high speed disperser for 15 minutes at 500 rpm, after which the pigments and extenders are added.
- the grinding stage is carried out at 2000 rpm for 20 minutes after which speed is reduced to 800 rpm and coalescent is added followed by the binder.
- the examples show the contact angle hysteresis values after 1 coat application on fiber cement boards after 7 days air drying.
- Emulsion 25 1.92 grams
- Emulsion 25 1.92 grams
- Aerosil R972 slurry (14.3%) 1 gram
- Aerosil R972 (14.3% slurry) 2.5 grams
- Emulsion 25 1.92 grams
- Emulsion 25 1.92 grams
- Emulsion 25 1.92 grams
- Aerosil R972 slurry (14.3%) 2.5 grams
- Emulsion 25 1.92 grams
- Aerosil R972 slurry (14.3%) 2.5 grams
- Aerosil R972 slurry (14.3%) 2.5 grams
- Emulsion 25 1.92 grams
- Aerosil R972 slurry (14.3%) 2.5 grams
- Emulsion 25 1.92 grams
- Emulsion 25 1.92 grams
- Aerosil R972 slurry (14.3%) 2.5 grams
- Emulsion 25 1.92 grams
- Aerosil R972 slurry (14.3%) 2.5 grams
- the examples show the contact angle hysteresis values after 1 coat application in the presence of conventional paint ingredients.
- a Sample PVC calculated for example d indicates the coating is 23.13% above the theoretical CPVC (critical Pigment Volume concentration).
- Tables 9 and 10 above show that it is possible to attain low CAH values provided certain ratios of micron and nano sized particles (commercially available treated clay and treated silica) are present along with conventional paint ingredients and specific polymers.
- Example 35 Sample Paints prepared using the water-borne acrylic random copolymer dispersions in combination with the functionalized particles in accordance with the present invention
- Example 36 Sample Paints prepared using the water-borne acrylic random copolymer dispersions in combination with the functionalized pigments in accordance with the present invention and their exterior exposure data
- Blends of dispersion and emulsion have been used in this example with a balance of hydrophobicity and hydrophilicity. Conventional paint ingredients have been used in this formula. These coatings exhibit good water repellency after application. Table 12
- the paints pass the water immersion test over a period of 7 days without blistering.
- the wet scrub resistance of these coatings was 60 cycles. Accelerated anti-algal chamber testing for 30 days shows the paints 1 and 2 to have a rating of 7.5/10. 2 coats of paint were brush applied over 1 coat of Asian Paints exterior primer.
- the water, dispersion polymer, surfactant, defoamer, glycol, biocides, dipentene, rheologicai additive are mixed on a high speed disperser for 15 minutes at 500 rpm, after which the pigments and extenders are added.
- the grinding stage is carried out at 2000 rpm for 20 minutes after which speed is reduced to 800 rpm and coalescent is added followed by the binder.
- MMA methyl methacrylate
- n-BMA n-Butyl methacrylate
- MAA Glacial methacrylic acid
- Zonyl TAN and Zonyl TM Perfluorinated monomer
- 2-EHA 2-ethyl hexyl acrylate Paints with 65, 70 and 75 PVC were prepared according to this invention and exposed to QUV B, 313 nm for 1100 hours.
- Example 38 Water-borne acrylic random copolymer dispersions prepared with a combination of different monomers
- a series of different polymers were prepared with different starting monomers. All the polymers have an acid value of 50 mg KOH/g and contain the perfluorinated monomer at a fixed content of 5% on monomer composition (except E which does not contain the fluorine based monomer). The resin E does not contain the perfluorinated monomer and is shown to compare the surface energies of the coatings with and without the perfluorinated monomer.
- MMA methyl methacrylate
- n-BMA n-Butyl methacrylate
- MAA Glacial methacrylic acid
- t- BMA Tertiary butyl methacrylate
- MA 2000 Polyethylene glycol methacrylate with molecular weight 2000
- Zonyl TM Perfluorinated monomer
- BA Butyl acrylate
- 2-EHA 2- ethyl hexyl acrylate
- the film cast from resin E which does not contain fluorinated monomer shows a surface energy of 31.9 mN/nrv while films of resins A to D which contain 5% fluorinated monomer on resin solids give a surface energy of ⁇ 25 mN/m and is preferred for exhibiting dirt pickup resistance.
- the water absorption of the resin films varied between 5 to 20% after 96 hours in water for the different resins.
- Example 39 Paints prepared using the water-borne acrylic random copolymer dispersions of example 38 in combination with the functionalized pigments in accordance with the present invention and their exterior exposure data with only hydrophobic organosilicone resin which is hydrophobic.
- paints were prepared using the resins 39A, B, C and D.
- the paint formulation is given in Table below.
- micron sized pigments and extenders have been used to prepare the paints.
- the Ti02 used has an average particle size of 0.32 microns.
- the water, surfactants, defoamer, glycol, biocides, rheological additive are mixed on a high speed disperser for 15 minutes at 600 rpm, after which the pigments and extenders are added.
- Insitu treatment of the pigments and extenders was carried out during paint making using a hydrophobic organosilicone in the formulation.
- the grinding stage is carried out at 2000 rpm for 20 minutes after which speed is reduced to 800 rpm and the binder is added followed by thickener and coalescent.
- Acrylic binder 18.679 21.893 0 19.33 18.679 21.023 21.06 19.06 18.916
- Asian Paints exterior primer followed by 2 coats of these paints at 25% volume dilution was applied on a vertical wall with a horizontal ledge.
- the wet scrub resistance of these coatings was 500 cycles.
- all the samples except sc6 show good properties in terms of DPUR, whiteness, hydrophobicity and good antimicrobial properties.
- This demonstrates the importance of other acrylic monomers such as t-butyl methacrylate, n- butyl methacrylate as part of the resin composition, apart from the perfluorinated monomer and a high acid value (50 mg KOH/g) in this example.
- Example 40 Water-borne acrylic random copolymer dispersions prepared with a combination of different monomers and varying acid values
- a series of different polymers were prepared with varying acid values from 25 to 50 mg KOH/g. 6 resins made with different hydrophobic monomers were made. All the resins had a pH of 8 to 9.
- MMA methyl methacrylate
- n-BMA n-Butyl methacrylate
- MAA Glacial methacrylic acid
- t- BMA Tertiary butyl methacrylate
- Zonyl TM Perfluorinated monomer
- BA Butyl acrylate
- 2- EUA 2-ethylhexyl acrylate
- the above resins with a range of acid values can be used in the present invention in combination with hydrophobic and hydrophilic organosilicones and the particulate mixture to achieve the desired wettability switching.
- Monomer water solubility of common monomers and octanol water coefficient values indicates that a combination of monomers is preferred with a balance of hydrophobicity and hydrophilicity to prepare polymers suited for the current invention.
- Table 20 The values of octanol water coefficients for the monomers are taken from "Journal of Biomedical Materials Research” Volume 15, 787-793 (1981), S. Fujisawa and E. Masuhara (log Kow values for acrylic acid, methacrylic acid and t-butyl acrylate are from literature)
- Example 41 Paints prepared using the water-borne acrylic random copolymer dispersions of example 40 in combination with the functionalized pigments in accordance with the present invention and their exterior exposure data
- organosilicone 0 4 9 4 4 4 4 0 0 4 9
- the Styrene acrylic based paints showed the maximum algal growth. Hence it is preferred to use a styrene free polymer. All the paints had a water contact angle of paint > 130° and the contact angle hysteresis for all the paints ⁇ 10 degrees. (C2 is styrene based as is revealed from Table 19 above). Both the contact angle and CAH values were maintained even after QUV exposure of 2000 hours and the Atlas exposure. After continuous showering of 15 days and 24 hours drying period the contact angle of the paint when checked is maintained. The ⁇ values of the coatings are ⁇ 2 even after 2000 hours indicating the coatings in accordance with this invention have excellent durability as well as retain the contact angle hysteresis values of ⁇ 10 degrees.
- ⁇ Colour difference
- YI Yellowness index
- WI whiteness index
- CR Contrast ratio
- the styrene based resin system C2 shows heavy chalking after Atlas, QUV exposure and the yellowness index is above 3 while for the styrene free systems, the yellowness index is below 3 and the extent of chalking was negligible.
- Example 42 Study of different contents of hydrophobic and hydrophilic organosilicones in paint formulation of example 41
- Example 43 Paints prepared with different micron sized particles using the water-borne acrylic random copolymer dispersions with a low content of short chain fluorinated monomer in combination with the functionalized pigments and extenders in accordance with the present invention
- MMA methyl methacrylate
- n-BMA n-Butyl methacrylate
- MAA Glacial methacrylic acid
- t- BMA Tertiary butyl methacrylate
- BA Butyl acrylate
- 2-EHA 2-ethylhexyl acrylate
- Bisomer amine D700 form Cognis
- Paint 43-A shows dirt streak marks after a 12 month exterior exposure while the others do not show any dirt streaks, lines or water spots. This shows the importance of the particle size of the fillers and extenders added. Quartz used in 43A has a d50 particles size of 40 microns (size range of 1 to 160 microns). Thus it is preferred to have a coating with plurality of particles with particles having a size less than d50 of 40 microns to avoid dirt streak lines.
- Example 44 Emulsions/ latex polymers prepared using the preferred monomer backbone in accordance with the present invention and their paint data
- Sample latex with the preferred monomers was prepared as shown in the following table.
- a combination of polymerizable surfactants (ADEKA SR10, Ether sulfate with 100% solids and ER40, nonionic, 60% solids) was used to prepare the latex. It was found that using the above latex the paints showed a CAH of >20 degrees (refer example 55) even though the paint formulas were same as that used above and the monomer combination of the latex was the same. Therefore the latex polymer of example 44 for a given coating formulation alone does not reduce the value of CAH below 20 degrees and it is preferred to have a dispersion polymer in the formulation either alone or in combination with the latex polymer.
- Example 45 Paints prepared with respirable crystalline silica free extenders using the water-borne acrylic random copolymer dispersions of example 43 (2) in combination with the functionalized pigments in accordance with the present invention and their exterior exposure data
- the paint 1 given in Table 29 (1) when exposed outdoors for a period of 6 months on a vertical surface shows excellent resistance to dirt streaks and water marks (left side of figure: paint according to current invention, right side of figure: commercial self cleaning paint).
- Example 46 Microbiology data of the paints prepared using the water-borne acrylic random copolymer dispersions in combination with the functionalized pigments in accordance with the present invention i i£r Various encapsulated and free biocides were evaluated with paints according to the invention such as Rocima 344, Rocima 350, Rozone 2000 alone and in combination with different chemistries. Paints in accordance with the present invention, show excellent anti algal properties in accelerated algal chamber testing, showing a rating of at least 8 to 9 out of 10 after 30 days testing. Further the coatings also show excellent anti fungal and anti bacterial properties after accelerated testing.
- Example 47 Photocata lytic activity of the paints 4 and 5 of example 45 prepared using the water-borne acrylic random copolymer dispersions in combination with the functionalized pigments in accordance with the present invention
- the paints 4 and 5 were subject to the methylene blue discoloration test. They were brush applied on a fiber cement board panel (6 inch x 3 inch). The coating was dried under ambient conditions for 7 days.
- the photoactivity of the paints was determined . by determining the photocatalytic decolorization of a methylene blue solution by UV-vis measurements. The peak of methylene blue was monitored at 664 nm.
- Table 30 showing the concentration of methylene blue in solution after 4 h and 8 h of UV exposure
- the coating was brush applied over Asian Paints exterior primer applied on a fibre cement board panel (6 inch x 3 inch).
- the coating was dried under ambient conditions for 7 days at room temperature (25 to 30 °C) and a relative humidity of 50 to 60%.
- the photoactivity of the paints was determined by determining the photocatalytic decolorization of a methylene blue solution by UV-vis measurements. The peak of methylene blue was monitored at 664 nm.
- Antifungal agent 0.2 0.2 0.2 0.2 0.2 0.2 0.2 0.2 0.2 0.2 0.2 0.2 0.2 0.2 0.2 0.2 0.2
- nTi ' 02 0.5% nTi ' 02 is lower for both the cases indicating enhanced durability with a low dosage of nano Titania.
- the DE value is higher with silicones which may be due to the enhancement of photocatalytic activity caused by the presence of organosilicones.
- Example 49 Enhancement of photocatalytic activity with oragnosilicones (both hydrophilic and hydrophobic) : 2 sets of paints were prepared each with 0%, 0.5% and 1% n Ti02 (p 25 from Evonik). One set contains 2 organosilicones (scl, 2 and 3) whereas the other is without organosilicones (sc4, sc5, sc6) as shown in Table 31.
- the control sample shows that methylene blue is itself undergoing some degradation under UV (365 nm) i.e. (about 23% in 8 hours).
- Both paints Scl and sc4 with 0% nano Ti0 2 show comparable degradation under UV ( ⁇ 35% in 8 hours), with sc4 showing a marginally higher value due to the presence of organosilicones. This may be due to the photocata lytic activity conferred by rutile Ti0 2 , DuPont R902 plus.
- increasing nano Titania is enhancing the activity
- paints containing a combination of organosilicones is enhancing the degradation of methylene blue and hence the photocatalytic activity.
- Example 51 Photocatalytic activity of the coating under diffused sunlight
- Example 52 Rust resistance of the paints prepared according to the current invention
- lit accordance with present invention were applied which exhibit contact angles ⁇ 90 degrees and show hydrophilicity when water is poured on their surface.
- Coating 1, 2 and 3 show good water repellency which indicates that the water containing rust would roll off from the surface.
- the coatings according to the present invention have a reduced tendency of spreading of rust marks thereby leading to coatings which can maintain their aesthetic appeal for a longer time.
- Increased contents of nano Titania are leading to increased hydrophilicity and hence the coatings are showing increased spreading of the rust stains.
- the 2 non stimuli responsive hydrophilic paints taken for comparison (paint 4 and 5 in Table 36) showed inferior performance.
- a disadvantage of only hydrophilic surfaces is that corrosion/ rust stains tend to spread over large areas leading to loss of the aesthetic appeal of the surface. Hence it is desired to have a reversible switchable surface in accordance with the present invention.
- Example 54 Surface energy measurements of coating according to the invention after 7 days drying under ambient conditions (30 °C and a relative humidity of 50-60%). Table 38
- **Binder used is example 43 (1).
- Example 55 Contact angle hysteresis measurements of coating according to the present invention for the paints of example 54
- the contact angle hysteresis is measured as the difference between advancing angle al and rl. Advancing angle for 10, 20 and 30 pL is measured (al, a2 and a3 respectively) and receding angle is measured for 20 and 10 pL (r2 and rl respectively). All measurements were carried out at 30 deg C. For the hydrophobic and hydrophilic organosilicones, their clear coats are applied on aerated cement blocks and the average value of CAH is taken as shown in Table 39.
- Example 56 Paints prepared with different contents of wetting and dispersing agents showing the effect on accelerated storage stability (30 days at 55 C) in accordance with the present invention with varying contents of glycol
- said aqueous coating composition that is oleophobic and has a water contact angle hysteresis of ⁇ 25° under certain environmental conditions with variations in at least one or more of temperature, humidity, pH, light which is reversibly switchable to a water contact angle hysteresis of >25° under said environmental conditions offering benefits of both superhydrophobic and superhydrophilic cleaning, good organic and inorganic dirt pickup resistance, resistance to dirt streaks, dirt lines, water spotting, water marks, good cleanability with water and forced air or wind, reduced chalking, good resistance to organic dirt and deposits such as bird droppings and oily stains, and reduced tendency for spreading of water containing rust with the additional benefits of better binding, durability .
Abstract
Stimuli responsive self-cleaning aqueous coating compositions for masonry and other substrates involving coatings that are durable, storage stable, ambient curing and recoatable comprising of smart polymers, which said coatings exhibits reversible super-hydrophobic and super-hydrophilic behaviour in response to external stimuli based variable environmental conditions such as variations in at least one temperature, pH, humidity and light; also exhibiting low contact angle hysteresis of < 25 degrees which reversibly switches to a contact angle hysteresis of > 25 degrees with external stimuli. Said aqueous coating composition comprising a polymeric dispersion of either acrylic, fluorinated and amine backbone or acrylic, fluorinated and acid backbone in combination with hydrophobic and hydrophilic silicones and at least partially surface treated particles also including particulate mixture of micro and nano particles having balance of hydrophobicity and hydrophilicity, advantageously facilitates the attainment of contact angle hysteresis of < 25 degrees by the said self cleaning coating composition.
Description
TITLE: STIMULI RESPONSIVE SELF CLEANING COATING
FIELD OF THE INVENTION
The present invention particularly relates to stimuli responsive self-cleaning aqueous coating compositions for masonry and other substrates involving coatings that are durable, storage stable, ambient curing and recoatable comprising of smart polymers, and more particularly, relates to said coatings exhibiting reversible super-hydrophobic and super-hydrophi!ic behaviour in response to external stimuli based variable environmental conditions such as variations in at least one temperature, pH, humidity and light; also exhibiting low contact angle hysteresis of < 25 degrees which reversibly switches to a contact angle hysteresis of 25 degrees with external stimuli. More specifically said aqueous coating composition Comprising a polymeric dispersion of either acrylic, fluorinated and amine backbone or acrylic, fluorinated and acid backbone in combination with hydrophobic and hydrophilic silicones and at least partially surface treated particles also including particulate mixture of micro and nano particles having balance of hydrophobicity and hydrophilicity, advantageously facilitates the attainment of contact angle hysteresis of < 25 degrees by the said self cleaning coating composition. Advantageously, said attainment of such low contact angle hysteresis (< 25 degrees) and its subsequent reversible wettability switching by the aqueous coating composition/ stimuli responsive self-cleaning coatings enables the flow of rain water or forced air/ wind to clean the dirt from said coating surface wherein said coating can be formed by simple mixing of the components and applied by simple techniques such as brush, spray and dip coating application. More advantageously, the coating exhibits good dirt pickup resistance and resistance to dirt lines, streaks and water spots and also exhibits good anti microbial resistance to microorganisms, particularly algae. Additionally the coating displays enhanced photocatalytic activity under diffused sunlight with reduced chalking and shows good resistance to organic dirt and deposits such as bird droppings and oily stains. The- coating also has a reduced; tendency for spreading of water containing rust.
BACKGROUND ART
The surface of exterior coatings on exposure to outdoor environments is contaminated by organic and inorganic dust, clay particles, smoke, exhausts gas, rust, bird droppings and discoloured by ultraviolet rays.
The wetting of surfaces with water results in the retention of water droplets on the surface and their evaporation, with the solids suspended or dissolved in the water remaining as unsightly residues (water marks, smears or spots) on the surface.
The wetting of a surface with water is frequently also a trigger for its degradation or for infestation with microorganisms and the growth of algae, fungi, lichen, mosses, bacteria etc. Therefore, it is particularly required for such outdoor coatings to show physical properties such as self-cleaning, weather resistance and anti microbial properties with long-term reliability.
Tremendous research efforts are being directed towards self-cleaning and stimuli responsive coatings. The field of self-cleaning coatings is divided into two types:
-hydrophilic
-and hydrophobic
Wettability of the surface can be measured by the contact angle which depends on the surface material and the surface texture. Alternatively, the wettability of a surface can be represented by the roll-off angle or Contact angle hysteresis. A hydrophobic surface in general exists if the roll-off angle is less than 25°.
Definition of terms:
Contact angle: The contact angle Θ is the : angle at which a liquid/vapor interface meets the solid surface wherein Fig 1 reveals contact angle measurement showing a hydrophobic and hydrophilic drop*
Hydrophobic: When the contact angle is >90° the surface is said to be hydrophobic.
Superhydrophobic: When the contact angle is >150° the surface is said to be superhydrophobic.
Hydrophilic: When the contact angle is <90° the surface is said to be hydrophilic.
Superhydrophilic: When the contact angle is <30° the surface is said to be superhydrophilic
Oleophobic: When the contact angle is >90° with oil/ oil like substance, the surface is said to be Oleophobic.
Contact angle hysteresis (CAH): Measure of the ease with which a drop of water rolls off a surface. It is the difference between the advancing angle and the receding angle.
Figure 2 reveals contact angle measurement showing the advancing and the receding angle wherein the Advancing angle: The contact angle which is a result of increasing volume of water on a surface is called the advancing angle and wherein the Receding angle: When
water with a syringe is removed from a surface the contact angle is called the receding angle.
It is known that the wettability of a surface by hydrophilic liquids may be reduced by a hydrophobic coating of the surface. Furthermore, it has proven favorable to structure hydrophobic surfaces. The structuring reduces the adhesion of the surface for polar liquids such as water and leads to reduced adhesion of solid deposits such as dirt particles on the surface. Given appropriate structuring, the dirt particles are flushed from the surface by moving water. The particulate solids may also be removed very easily by means of forced air or wind.
U.S. 2002/0048679 describes surfaces having a smooth, extremely hydrophobic polymer (for example, polytetrafluoroethylene) film and surfaces having a smooth extremely hydrophilic polymer film as examples of surfaces on which water and dirt can run off without forming droplets. U.S. 2002/0048679 further describes how a long-term hydrophobic coating may be formed by applying certain silane derivatives underneath a hydrophobic coating on a surface. Other self-cleaning surfaces are described in U.S. Patent Application numbers US 2002/0150723, US 2002/0150724, US 2002/0150725, US 2002/0150726, US 2003/0013795 and US 2003/0147932.
Oil and water repellent compositions based on fluoropolymers are described by Linert, et al, in WO 199700230, describe a composition comprising a fluoroaliphatic groups, carboxyl containing groups, oxyalkene groups and optionally silyl groups which provide repellency to oil- and water-based stains for porous substrates.
Ueda et al, in US20070197717, describe a masonry treating agent comprising fluoromonomer, a monomer having at least one acid group, and a non-fluorrnated monomer having a hydrophobic group. A high content of fluorine containing monomer (35 to 90%) is being used.
Random fluorinated copolymers prepared by radical copolymerisation of monomers in solution in a water-miscible organic solvent using peroxides or azo compounds as initiators have been described (see, for example, EP 542598, EP 1106630 and US 2004026053), together with their hydrophobic and oleophobic properties on various substrates.
US 7781027 teaches durable, weatherable and scratch -resistant hydrophobic coatings comprising a fluorinated component and an adhesion promoter compound. The adhesion promoter compound is said to include an alkoxy group, a furfuryl-containing ring structure, and a reactive group.
US 4859754 teaches a water and oil repellent polymer having desoiling properties composed of a polyfiuorinated group-containing copolymer obtained by copolymerizing high levels (65-95%) of a polyfiuorinated group-containing monomer and an amphipathic monomer (5-35%) having a hydrophilic moiety and a lipophilic moiety.
A further development of these surfaces consists in structuring the surfaces in the im to nm range wherein U.S. Pat. No. 5,599,489 discloses a process in which a surface can be rendered repellent by bombardment with particles of an appropriate size, followed by perfluorination.
US3,354,022 and WO 96/04123 describe other processes for reducing the wettability of articles by topological alterations in the surfaces wherein artificial elevations or depressions with a height of from about 5 to 1000 m and with a separation of from about 5 to 500 pm are applied to materials which are hydrophobic or are hydrophobized after the structuring process. Unfortunately, such coatings have a disadvantage in that they possess a low abrasion resistance and only a moderate self-cleaning effect.
US 20110111659 teaches a method of providing oil repellency, water repellency, and dynamic water repellency to a substrate by coating with a polymer composition comprising hydrophobized nanoparticles and one or more monomers of Ci. to. Ci8, straight, branched, or cyclic alkyl (meth)acrylate> one or more monomers selected from the group consisting of N- methylol (meth)acrylamide and a second polymer comprising high quantities of fluorinated monomer.
EP2210921 teaches a hydrophobic composite coating which comprises a plurality of carbon nanotubes dispersed in a hydrophobic polymer resulting in a coating which has a water contact angle of least about 120°.
Super-hydrophobicity is frequently also referred to as the "lotus effect" because it was first observed on the leaves of the lotus plant. Super-hydrophobicity of a surface results in particularly easy dripping or rolling off of water from the surface, which supports fast drying of the surface and hampers the adhesion of dirt to the surface.
In this context, two states are known, namely the Wenzel state and the Cassie-Baxter state. In the Wenzel state, the drop of fluid completely covers the surface. As roughness increases, the hydrophobicity of a hydrophobic surface rises. Vice versa, the contact angle of a hydrophilic surface decreases with increasing roughness. In this way, a hydrophobic
surface can be turned into a super-hydrophobic surface, and a hydrophilic surface can be turned into a super-hydrophilic surface by increasing the surface roughness. The Cassie- Baxter state furthermore considers that air bubbles are trapped between the drop and the rough surface.
The leaves of the lotus plant have elevations made from a wax, and these elevations lower the contact area with water. WO 00/58410 describes these structures and claims the formation of the same by spray-application of hydrophobic alcohols, such as 10- nonacosanol, or of alkanediols, such as 5, 10-nonacosanediol. The separations of the elevations in the structures are in the range from 0.1 to 200 pm and the heights of the elevations are from 0.1 to 100 Mm.
US 7544411 teaches a self-cleaning paint coating that exhibits the "Lotus Effect". The paint coating comprises a paint base coat and a topcoat. The topcoat includes structure-forming particles and is at least partially hydrophobic. The structure-forming particles have an average particle diameter of less than 100 nanometers (nm) and form elevations in the topcoat surface that are interspaced at a distance of less than 50 nm. The topcoat of the paint coating is formed from the agent which contains a liquid medium, a hydrophobating agent dissolved in the liquid medium, and structure-forming particles suspended in the medium.
US7196043 targets a composition for producing a self-cleaning coating on a surface, the composition comprising: an aqueous mixture comprising water, metaJ oxide nanoparticles present at between 0.001 percent to 10 percent by weight of the mixture and having a particle size- of less than 300 nanometers and a fluorinated water-soluble hydrophobic surface modifier of between 0.001 percent and 10 percent by weight of the mixture wherein the surface modifier can produce an unstructured surface having a surface energy below 30 dynes per centimeter, which fluorinated water-soluble hydrophobic surface includes fluorinated acrylic polymers. CH-PS-26 82 58 describes water-repellent surfaces which exhibit a contact angle with water of more than 120°. They are obtained by applying powders, such as china clay, talc, clay or silica gel, on a substrate, the powder being first hydrophobized by organic silicon compounds. The application is performed together with curable resins or from solutions in organic solvents.
US3227675 and US4297145 teaches the hydrophobizaton of fillers such as kaolin, clay, chalk, siliceous chalk or diatomaceous earth with' silane for use in vuicanisable rubber moulding compositions. US4715986, US6605351, EP0156270A2 and German Offenlegungsschrift No. 2107082 teaches particles modified by hydrophilic and hydrophobic groups wherein the hydrophilic and hydrophobic groups are distributed anisotropically on the surface of the modified particles and the use of such particles as surface active products.
US 4061503 teaches a composition comprising titanium dioxide particles containing on their surfaces a silane, in an amount sufficient to improve the dispersibility of said particles in a resin or plastic medium.
US8187707 teaches hydrophobic film or coating, comprising primary particles covalently bonded with secondary particles, adhering to the surface of the primary particles and having an average diameter that is smaller than the average diameter of the primary particles, and a hydrophobic layer covering at least partly the surface of the secondary particles and adhering to that surface.
US8202614 discloses an additive particle comprising a carrier microparticle, a plurality of nanoparticles adhered to and substantially covering a surface of the microparticle, and a hydrophobic surface treatment material in contact with exposed surfaces of the carrier microparticle and/or the nanoparticles sufficient to impart a superhydrophobic contact angle to the additive particle of greater than 150°; wherein the hydrophobic surface treatment material comprises a perfluorinated alkyl silane.
US8147607, US7964244 and US20050118433 relates to a hydrophobic coating composition for forming a coating on a substrate, consisting of hydrophobic particles, a solvent and water. The hydrophobic particles comprise one or more metal oxides that are treated with an organosilane, alkylsilane, fluorinated silane, and/or disilazane.
US6683126 provides a coating composition for producing difficult-to-wet surfaces, comprising at least one finely divided powder whose particles have a hydrophobic surface and a porous structure characterized by a BEET surface area of at least 1 m2 /g, and at least one film-forming binder characterized by a surface tension <50 mN/m, the weight ratio of powder to binder being at least 1 :4.
On the other hand a hydrophilic surface may also produce good dirt shedding. The hydrophilic coating resists dirt pickup since water can wet the surface and flow off more easily allowing dirt to be easily washed off by rainwater. US20060058490 claims a hydrophilic coating (contact angle <30°) formed using a hydrophilic silane and mixture of hydrophilic particles. They mention that if an organic binder is used, it may undergo degradation if photo catalytic particles are present in their system. Solgel network formation leads to a coating film with a hydrophilic surface where dirt streak marks can be avoided.
US20040082494 teaches hydrophobic-hydrophilic block copolymers to increase the water affinity of low energy surfaces using high contents of hydrophilic monomers (0 to 95%).
Waterborne fluoropolymers for paint use by Asakawa et al. in Journal of Fluorine Chemistry, Volume 104, Issue 1, June 2000, Pages 47-51 describes the synthesis of fluoropolymer emulsion having a hydroxy functionality, which could be cross-linked with water dispersible polyisocyanate resulting in a hydrophilic film-surface modifier which can prevent the dirt line on an outer wall.
Under UV irradiation Ti02 (Titanium dioxide) is photocatalytically active and can produce super-wetting effects as a result of water hydrolysis effects. US6048910 teaches a coating composition comprising of an emulsion of a silicone resin, typically a water-insoluble, silanol group-bearing silicone resin dispersed in water, and contains photocatalyst particles, typically titanium oxide which forms a hydrophilic film having a water-drop contact angle of no more than 60° when irradiated with UV-containing light. Films obtained by applying this coating composition to the surface of substrates have excellent weathering resistance and antifouling properties due to a self-cleaning effect arising from the photocatalytic action of the particles.
The crystalline forms of titanium dioxide exhibit photocatalytic activity, of which, only anatase and rutile have potential commercial applicability as photocatalysts. Rutile titanium dioxide has band gap energy of 3.05 eV, which corresponds to approximately 420 nm wavelength of light, and anatase titanium dioxide has a band gap of 3.2 eV (approximately 385 nm). Within the field of photocatalysis, anatase titanium dioxide has established a reputation as generally superior to rutile titanium dioxide.
When a titanium dioxide crystal absorbs a photon of sufficient energy (from natural radiation, sun or from an artificial radiation exposure, lamp), an electron is promoted to the conduction band, and a positive hole (h+) is generated in the valence band. This "electron- hole pair" generates highly oxidizing hydroxyl and superoxide radicals at the crystal surface that are capable of oxidizing practically all organic matter to C02 and H20.
Photocata lytic oxides are known to have antifouling, antimicrobial, soil-degrading, deodorizing, air-cleaning, antifouling, antimicrobial, water-cleaning, superhydrophilicizing and/or antifogging effects. Due to photocata lysis the organic dirt even when deposited on the surface of the coating, are unstable and hence easily removed, permitting the surface to be kept clean.
US6337129 teaches an antifouling coating composition with both a hydrophobic portion, and a hydrophilic portion induced by a photocatalyst, both portions being present in a microscopically dispersed and exposed state on the surface. The content of the photocata lytic oxide in the surface layer is also not particularly limited. For example, the content of the photocatalytic oxide in the surface layer is preferably about 10 to 80% by weight, more preferably about 20 to 50% by weight. Thus high contents of photocatalysts are being used.
US7521039 describe photocatalytically active rutile titanium dioxide which demonstrates enhanced activity in the visible light spectrum. It is produced by neutralizing the rutile seed to a high pH prior to washing and thermally treating the seed. US7955430 teaches a coating material comprising a binding agent and at least one filler including particles having a size and/or surface roughness of about 100 microns or less, and a photocatalytically active agent about 2 to about 15 weight percent of the coating material. The binding agent may be at least partially decomposed by a photocatalytic action, and a microstructured, self-cleaning surface may be formed. The photocatalytically active agent used is an oxide of titanium, zinc, iron, manganese, molybdenum and/or tungsten, which comprises at least one additive selected from C, N, S and/or from a transition metal oxide and/or transition metal halide. The selected additive(s) may enable the stimulation of the catalytic property even with wave lengths e.g. in the visible range of the solar spectrum. The desired self-cleaning properties may advantageously be achieved if the coating material is formulated using an excess of binding agent.
In WO00/06633, coating materials for plastics based on inorganic binding agents and fillers are described. The coating materials set forth in the known publication may additionally contain photocatalytically active agents for self-cleaning by means of decomposition of organic soiling and superhydrophilics, while the binding agents used in the manufacture of the known materials are stable to the photocata lytic effect of the photocatalytically active agents.
US20100311572 discloses a coating material comprising at least one binder and at least one photocatalytically active particle comprising a core composed of at least one first substance having a diameter of 0.1 to 1 pm and at least one envelope at least partly surrounding the core and composed of at least one second substance having an average layer thickness of 0.1 to 10 nm. The active particles are generally present in the coating material in an amount ranging from 0.1% to 10% by weight. Useful metals or semimetals whose oxides are present in the core of the photocatalytically active particles are generally selected from the group consisting of elements of groups I to XV of the periodic table (in accordance with IUPAC), lanthanides, actinides and mixtures thereof, more preferably from the group consisting of V, Ti, Zr, Ce, Mo, Bi, Zn, Mn, Si, Ba, Au, Ag, Pd, Pt, u, Rh, La and mixtures thereof.
Very particularly preferred metal or semimetal oxides present in the at least one envelope of the particles are Si02, ZnO, Ce02, Ti02 SnO or mixtures thereof.
It is thus possible to adjust the activity of the core-envelope particles such that soils adhering to the coating material of the present invention are catalytically degraded, while the binder present in the coating of the present invention is essentially not catalytically degraded.
JP 11 181339 A discloses a hydrophilic coating comprising photocatalytically active titanium' dioxide particles. The titanium dioxide particles have a particle diameter of 1 to 100 nm, comprise particles of Sn02 and are silica and/or silicone coated. The coating material prevents soiling of a surface coated with this material. A surface thus treated further has self-cleaning properties when rained upon. JP 2006-233343 discloses a liquid photocatalyst composition for coating textiles which comprise photocatalyst particles having a preset particle diameter and a coating. The size of
these photocatalyst particles is 0.5 to 10 pm. In a preferred embodiment, the core of the photocatalyst particle consists of titanium dioxide and the envelope consists of silica.
JP 11228873 A discloses a paint composition comprising titanium dioxide particles coated with porous silica and having photocatalytic properties. The composition further comprises titanium dioxide pigment and an organic resin binder and does not teach any particle size of said photocatalytically active particles.
US20110129204 discloses a photocatalytic coating composition comprising a hydroxyapatite-coated photocatalyst, a silica-based binder, an acrylic emulsion, a thickener and water.
US 20110313095 relates to a mixture, comprising at least one thermoplastic polymer as component (A) and at least one photocatalytically active particle, comprising a non-porous core comprising at least one metal oxide or semimetal oxide with a diameter of from 0.1 nm to l.mu.m, and, at least to some extent surrounding the core, at least one porous outer layer comprising at least one further metal oxide or further semimetal oxide with an average layer thickness of from 0.1 to 10 nm, as component (B), to a process for the production of this mixture, via mixing of components (A) and (B), to the use of the mixture as photocatalytically active surface, to moldings, comprising this mixture, and to the use of this mixture for the production of moldings.
EP1955767 Al discloses a mixture comprising photocatalysts and organic polymers, such as polyacetals, polyethylene, polypropylene', polystyrene, polyvinyl chloride, nylon-type polymers, polycarbonates, etc. The photocatalysts used preferably comprise titanium dioxide particles, which have a coating composed of non-porous silicon dioxide.
JP2000-017096 discloses foamed foils composed of thermoplastic resins, which have titanium dioxide as antibacterial coating. Silanes have been applied as surface modifier on the surface of the titanium dioxide.
JP2000-204194 discloses a process for the production of polymer compositions which comprise titanium dioxide as photocatalyst. The titanium dioxide has, for example, a coating of silicon dioxide. The ratio of polymer to titanium dioxide is from 97: 3 to 20:80.
JP 09/225321 A discloses a photocatalytically active body composed of titanium dioxide in the anatase form. The titanium dioxide used in that composition is present between two porous layers of another inorganic compound, such as silicon dioxide. JP 2005-097608 A discloses a polyoiefin film which comprises photocatalyst particles, where the photocatalyst particles are mixed crystalline oxides comprising titanium dioxide and silicon dioxide. Said JP patent does not disclose any core-outer layer photocatalysts or any use in thermoplastic polymers. KJA Raj et al. in Indian Journal of Chemistry, July 2010, 49A(07), 867 have prepared a series of titania samples with different loadings of silica (0.5-10 wt.%) by wet impregnation method. High dispersion of silica in the Titania matrix and the bonding of Ti-O-Si in silica- titania samples are found to inhibit the transformation of amorphous to anatase Titania. Evaluation of photocata lytic activity of the samples shows greater activity for samples modified with silica with an optimal loading of 7.5 weight % silica calcined at 500 °C. Conversion of 90 % and 35 % of 4-chlorophenol for UV-visible and visible light respectively is observed.
US20110312065 A teaches a substrate or coating that includes a protease which is capable of enzymatically degrading of one or more components of the biological stain to facilitate biological stain removal from the substrate or said coating.
The early applications of titanium dioxide in photocatalysts used nearly exclusively ultraviolet light sources as is required by the band gap energies of anatase titania. Ultraviolet (UV) light refers to light of a wavelength below about 400 nm. By contrast, light from the visible spectrum refers to light of a wavelength from about 400 nm to about 800 nm.
Accordingly, when conventional photocata lytic products are applied to interior products, their performance is greatly reduced because UV light constitutes only a relatively small fraction of sunlight and in part because indoor lighting is typically very low in UV intensity. Thus, there is a need to develop a photocatalytic coating composition that is suitably applicable to interior products which work under diffused sunlight as well as exterior products. If a larger proportion of the electromagnetic spectrum could be used, then a more efficient catalytic performance could be achieved and a larger number of potential applications could be realized.
Conventionally high contents (>1% of formulation) of nano Titania are used in photocatalytic coatings required for interior paints. There is also a need to develop an economical formulation where a low content of nano Titania can be used to achieve enhanced photocatalytic activity even in diffused sunlight.
Most photocatalytic paints comprise silicone type mineral binders and not binders based on organic products, so as to prevent the binder itself from being photocatalytically degraded. There is also a need to develop an economical photocatalytic coating composition that can minimize degradation of the organic binder.
With conventional coating materials, resistance to weathering is achieved by subjecting coating components to which a photocatalytic action is attributed, such as titanium dioxide pigments, to a surface treatment in order to reduce the photocatalytic activity. In this way, highly stabilized pigments with excellent protective effect for coating materials are obtained with conventional coatings. Historically, when rutile titanium dioxide has been considered applicable as a photocatalyst, it has been in a state in which the rutile crystals are "metallized," that is, containing small islands of metals such as platinum, rhodium, gold and silver on the rutile crystals. Unfortunately, such catalysts are prohibitively costly and may be leached out in practical use. Thus, there is a need to develop an economical formulation - where even rutile titanium dioxide photocatalyst can have enhanced photocatalytic activity and be activated by visible light.
Conventionally nano sized Titania (<100 nm, predominantly anatase) are used' in photocatalytic coatings. There is also a need to develop an economical formulation where a variety of particle sizes of anatase or rutile Ti02 from 1 nm to 1 micron can be used to achieve enhanced photocatalytic activity even in diffused sunlight.
US20030147932A1 claims an anti microbial coating with surface roughness. Amine functional monomer is used in combination with fluorinated monomers in US6521730, US5965659, US4147851 and US20110111659.
WO2007044784 teaches a self-decontaminating surface coating resistant to spores which when cured has a partially hydrophobic surface. The coating comprises a polymeric coating resin which, when cured, is hydrophilic. The coating also comprises a biocide, a germinating
agent, and a hydrophobic micro/nano particulate material. The coating is suitable for application to ceramics, metals, and polymer substrates.
Polymers that have anti-microbial properties are disclosed by the following patent applications: DE10024270, DE10022406, PCT/EP 00/06501, DE10014726, and DE 10008177. The antimicrobial properties are attributable to the contact of bacteria with'the surface. European patent application EP0862858 discloses that copolymers of tert- butylaminoethyl methacrylate, a methacrylate with a secondary amino function have microbicidal properties.
Peter Forbes dislcoses self cleaning materials in Scientific American 299, 88 - 95 (2008) wherein superhydrophilic titania has a contact angle of 0 degrees, hydrophilic standard window glass has a contact angle of 30 degrees, hydrophobic Teflon 100 degrees and superhydrophobic lotus leaves 160 degrees. The convergence of opposites is mentioned where researchers are exploring ways to get the same structure to switch from being superhydrophobic to superhydrophilic and vice versa. In the year 2000, titania pioneers Fujishima, Watanabe and Hashimoto in L Jiang, R. Wang, B. Yang, T. Li, D. A. Tryk, A. Fujishima, K. Hashimoto, D. Zhu, Pure Appl. Chem. 2000, 72, 73, used small amounts of titania to prolong the life of lotus effect surfaces. In 2003, Rubner and Cohen used a LBL technique to stack up alternating layers of positively charged poly(allylamine hydrochloride) and negatively charged silica particles. This led to a transparent superhydrophilic coating that demonstrated anti fog behavior through a phenomenon called nanowicking. In earlier work they used silica particles to mimic the lotus's rough hydrophobic surface. Smart beetles in the Namib Desert harvest water from thick morning fogs. It squats with its head down and back up, facing the foggy wind. Water condenses on its back and trickles down its mouth. In 2001, Andrew R. Parker [Nature 414, 33-34 (1 November 2001)], found that the back of the beetle has a bumpy, waxy, superhydrophobic surface. The tops of the bumps are free of wax and are hydrophilic. These hydrophilic spots capture water from the fog, forming droplets that quickly grow large enough for gravity and the surrounding superhydrophobic area to dislodge them. In 2006, Rubner and Cohen's team [Nano Lett, 2006, 6 (6), pp 1213-1217] created superhydrophilic spots of silica on superhydrophobic multilayers. Thus textured surfaces can either be superhydrophobic or superhydrophilic depending on the surface chemistry of the elevated regions. In 2006, a team led by Kilwon Cho in H. S. Lim, J. T. Han, D. Kwak, M. Jin, K. Cho, J. Am. Chem. Soc. 2006, 128, 14458 achieved complete switchability by adding a compound based on the molecule azobenzene
to the siliconized surface of a silica-polyelectrolyte multilayer. The surface is superhydrophobic, but under UV light the azobenzene compound changes configuration and changes to superhydrophilic.
Combinations of self-cleaning as well as stimuli responsive coatings with hydrophilicity and hydrophobicity as evident from the above discussions are thus reported in literature. To design such responsive surfaces, various methods such as self-assembled monolayers, layer-by-layer assembly, surface-initiated atom-transfer radical polymerization and electrochemical deposition have been proposed by X. Feng, L. Feng, M. Jin, J. Zhai, L. Jiang, D. Zhu, J. Am. Chem. Soc. 2004, 126, 62; H. S. Lim, J. T. Han, D. Kwak, M. Jin, K. Cho, J. Am. Chem. Soc. 2006, 128, 14458; X. Yu, Z. Wang, Y. Jiang, F. Shi, X. Zhang, Adv. Mater. .2005, 17, 1289; J. Lahann, S. Mitragotri, T. Tran, H. Kaido, J. Sundaram, I. S. Choi, S. Hoffer, G. A. Somorjai, R. Langer, Science 2003, 17, 371.
Most of these responsive materials designed by these methods are still limited to the laboratory as they involve use of complex polymers and application procedures.
Polymer systems that undergo a conformational change or phase transition in response to external stimuli such as temperature, pH, light and electric fields have opened up a number of opportunities for the development of new materials. Several environmentally responsive polymers are also described in WO2012075033.
Acid functional polymers are pH responsive with the polymer becoming hydrophilic with pH>7 and hydrophobic with pH<7. Polymers with sufficient hydrophilicity in their backbone can exhibit response to humidity with higher humidity conditions leading to water absorption and hence the surfaces behaving hydrophilic and vice versa. Of these, the most commonly utilised stimulus is temperature and polyacrylamide based copolymers are well known to create temperature-stimulus-sensitive surfaces as taught by Cheng, X.; Canavan, Η.Έ.; Stein, M. J.; Hull, J. R.; Kweskin, S. J.; Wagner, M. S.; Somorjai, G. A.; Castner, D. G.; Ratner, B. D. Langmuir 2005, 21, 7833; Sun, T.; Wang, G.; Feng, L; Liu, B.; Ma, Y.; Jiang, L; Zhu, D. Angew. Chem., Int. Ed. 2004, 43, 357; Xia, F.; Feng, L; Wang, S.; Sun, T.; Song, W.; Jiang, W.; Jiang, L. AdV. Mater. 2006, 18, 432.
Responses based on temperature stimulus are largely dependent on lower critical solution temperature (LCST). For example, poly-n-isopropylacrylamide (PNiPAm) exhibits a LCST of 32 °C in water. Below the LCST, PNiPAm expands and becomes hydrophilic (swollen coil conformation) while above the LCST it shrinks becoming hydrophobic (globular conformation) as disclosed in Benee, L. S.; Snowden, M. J., Chowdhry B. Z. Langmuir,
2002, 18, 6025. Copolymerization with hydrophobic monomers are known to decrease the LCST whereas with hydrophilic monomers increases the LCST.
Aqueous polymeric dispersions are taught in US5173523, using a two stage monomer addition process followed by stripping of the co-solvent. US6277953 teaches preparation of an aqueous polymeric dispersion which is substantially free of an emulsifier, having an acid value in the range of from about 10 to about 40 with a particle size of not more than about 300 nm and the aqueous dispersion has less than about 2 weight percent organic solvent, at least about 30 weight percent solids. US4616058, US5817370 also teach the preparation of polymeric dispersions. US5319019 teaches a process for making acrylic polymer ammonium salt/organic solvent/water mixture, the organic solvent and water being in amounts effective for forming an azeotrope; resulting in a polymer having less than 2 weight percent of organic solvent.
Responsive colloidal systems: Reversible aggregation and fabrication of superhydrophobic surfaces by Mikhail Motornov, Journal of Colloid and Interface Science 310 (2007) 481-488, reported a method of fabricating stimuli-responsive core-shell nanoparticles using block copolymers of poly(styrene-/?-2-vinylpyridine-£>-ethylene oxide) and poly(styrene-£»-4- vinylpyridine) covalently bound to silica nanoparticles with two different diameters: colloidal silica 200 nm in diameter and fumed silica 15 nm in diameter.
The construction of a rough surface functionalized with carboxylic acid groups is capable of switching between superhydrophilic and superhydrophobic in response to pH. Wang and coworkers Wang, J.; Hu, J.; Wen, Y.; Song, Y. Jiang, L, Chem. Mater. 2006, 18, 4984- 4986 produced a' pH responsive surface from self assembled layers of polystyrene- po!y(methyl methacrylate)/poly (aery lie acid) core-shell beads. The beads form the rough surface that is needed to obtain the extreme contact angles. Sodium dodecyl- benzenesulfonate (SDBS) was added as an emulsifier but is also part of the key to switching the behavior of the surface. The S03 groups of the SDBS hydrogen bonds with the COOH groups on the bead, creating a hydrophobic surface state. When the acid groups are deprotonated the COO- and the S03- groups repel each other and provide a hydrophilic surface state.
N Verplanck in Nanoscale Res Lett (2007) 2:577-596 teaches the use of self-organized Ti02 nanotube layers grown on Ti by electrochemical anodization. The as-prepared Ti02 nanotubes displayed a superhydrophilic wetting behavior. When modified with organic molecules, such as octadecylsilane or octadecylphosphonic acid layers, the surfaces showed
a superhydrophobic behavior, wherein by combining said Ti02 layer with an irreversible UV induced decomposition of the organic monolayers can be used to adjust the surface wetting properties to any desired degree from super-hydrophobic to superhydrophilic. US7695814 and US7923106 are directed to a responsive coated substrate comprising at least one silicone-based, substantially hydrophobic polymer and at least one substantially hydrophilic polymer comprising polyethylenimine wherein said responsive coating substrate is in a first state; and methods of coating the same.
US6919398 teaches a hydrophobic coating with a contact angle of greater than 130° after 3 minutes equilibrium consisting of a hydrophobic polymer and a bimodal particle size distribution of particles more than 5 microns particle size and less than 3 microns. They also teach about their formulation containing an insufficient amount of hydrophilic components. It is stated in DE2150736 that it is known to use vinyl chloride copolymers, chloro rubber, copolymers of acrylic esters, optionally with styrene and vinyl toluene, and siloxane resins as binders in modern pigment-containing facade paints; the publication suggests the use of a mixture of (a) siloxane resins and (b) copolymers or terpolymers of methylmethacrylate with acrylic acid esters, carboxylic acids or carboxylic acid esters, vinyl toluene or styrene. It is stated that such paint can penetrate into the outer layers of the surface to be painted, i.e. act to impregnate it and to be water-repellent. The kind of siloxane resins is not stated.
A paint described in DE2352242, particularly for protecting facades such as concrete facades, which paint contains pigment and as a binder copolymerisates of methacrylic acid esters has in the binder, in order to increase the water-repellent properties, a content of lower molecular weight methyl, phenyl or methylphenyl siloxanes in an amount of 10-50% by weight of the copolymerisate, and preferably also a catalyst for the siloxanes, e.g. an organotin compound. The siloxanes are stated to polymerise to form higher molecular weight compounds, which improves the water-repellency.
From DE2307422 there is known a water-repellent coating composition consisting of an inert carrier liquid, a resin-like binder and one or more metal or metalloid oxides the surface of which has been rendered hydrophobic with an organic silicon compound. A coating formed by such a composition must be expected to be surface-hydrophobic and hence give a good water-repellency. US5584921 discloses coatings for building materials, in the form of an aqueous dispersion comprising: (a) 10 to 60 wt % of at least one polysiloxane (an aqueous emulsion of a
hydroxyl-functional methyl silicone resin); (b) 5 to 65 wt % of at least one colloidal silica in the form of silica sol; (c) 5 to 80 wt % of at least one pigment or unreactive filler; and (d) 0 to 30 wt % of at least one further varnish or paint additive.
US6013724 teaches a raindrop fouling -resistant paint film comprising a tetramethoxysilane partial hydrolyzed condensate having a silanol (SiOH)-methoxysilyl (SiOMe) group molar ratio of not more than (SiOH)/(SiOMe)=l/10, a dimer through octamer content of 0-30 weight %, and a weight average molecular weight of 1500-5000 as available upon hydrolysis at a hydrolysis degree of less than 100% and condensation of tetramethoxysilane and at least one film-forming resin and/or at least one inorganic binder having alkoxysilyl as a reactive curing group as selected from the group consisting of an alkoxysilane compound, a partial hydrolyzed condensate of said alkoxysilane compound, and a siloxane polymer.
Coatings containing silicates have been shown to provide superior stain resistance. Such coatings are described, for example in EP0942052, JP2002294154, and JP01172389. Organosilicates have also been used with fluoropolymer resins to produce desirable coating compositions as described in JP02003775, JP2003020450, U.S. Pat. No. 6,635,341 and EP1035184. An organosilicate works by a mechanism of stratification first, and then hydrolysis with rain water in the surface increasing the hydrophilicity. The book "Silicone resins and their combinations" Wernfried Heilen, Vincentz Network, 2005 teaches the use of silicone containing binders to enhance durability of architectural paints. The binder in silicone resin emulsion paints comprises 50 % mineral-based silicone resin and 50 .% polymer emulsion. On mineral building surfaces and in coatings, silicone resin binders build up stable three-dimensional silicone resin networks which are chemically bound to the mineral substrate. The water repellency arises from the organic group on the silicone resin while the inorganic portion of the silicone resin bonds with the filler and the pigment together, creating permanently water-repellent capillaries and pores.
There are several published articles which address the elimination of water marks. In one example, US5759980 teaches a composition, which comprises a surfactant package consisting of a silicone-based surfactant and a polymer which is capable of bonding to a surface to make a hydrophilic film which eliminates the problem of water marks. However, this hydrophilic coating may tend to be removed from the surface by a single water rinse. German publication DE-A2161591 also describes a composition for cleaning cars wherein the surface is again made hydrophilic by using amino functional polymers. This coating also
tends to be rinsed off from a single rinse. There are number of waxes and other products available in the market for attempting to retain this spot free finish. However, certain wax containing hydrophobic surfaces result in water marks when the water dries giving an unaesthetic appearance.
Therefore, there is a need to develop a process and coating for protecting various surfaces from the appearance of water marks, corrosion streaks, and dirt streaks while maintaining the water repellency even after several water rinses. US20060110542 discloses a composition for forming a detachable and renewable protective coating produced by making a highly concentrated dispersion of hydrophobically modified silica particles in the presence of a disilazane derivative under high shear conditions.
Examples of commercially available materials which attempt to produce this "Lotus" cleaning effect are products sold under the trade name of MINCOR available from BASF, and TEGTOP available from Degussa. As stated in US20090018249 these products, when tested for their ability to protect various surfaces from the appearance of water marks, corrosion, and dirt repellency and while maintaining the water repellency, were deemed unsuitable. The coating is initially super hydrophobic and may remain so for long periods indoors; however, when exposed to outdoor UV light, rubbed even slightly, or in general exposed to weather, the coating loses super hydrophobicity and becomes less hydrophobic within days or even hydrophilic and hence less useful for the object of the present invention. Examination under the microscope after a week of exposure on a panel in a UV cabinet reveals that a coating made from fumed silica and at least one film forming binder as taught in US6683126 disintegrates.
US20030013795 teaches a self-regenerating, self-cleaning hydrophobic surface formed when particles are secured on a carrier, resulting in a long lasting self cleaning action. WO2008071957 relates to novel block copolymers for use in coating surfaces to provide self-cleaning surfaces and capture/release materials wherein the copolymers comprises a first hydrophobic block and a second hydrophilic block comprising a non-fluorinated acrylate or methacrylate monomer. Although some of the above-mentioned surfaces may have excellent self-cleaning properties, the attachment or colonisation of microorganisms can impair these properties.
For instance, micro-organisms (fungi, algae, bacteria) may colonise a self-cleaning surface which may impair, or may entirely remove, the self-cleaning properties of the surface. Further, organic dirt and deposits such as bird droppings and oily stains further mar the surface of these self cleaning coatings. Coatings containing photocata lytic particles use high contents of nano Titania or are doped with expensive materials to enhance activity under visible light which is not a feasible proposition.
As apparent from the above, most of the responsive materials discussed above and methods to reach to the said materials are either still limited to the laboratory as they involve use of complex polymers and application procedures or are not sufficiently durable, permanent or mechanically robust. Additionally, there is often a need for high contents of organofluorine compounds or fluoropolymers which are very expensive. With the ever- increasing practical needs of such smart surfaces, it is of particular importance and a long- felt need in the art to explore achievement of new water-borne pigmented coating exhibiting reversible hydrophilic-hydrophobic character with variation in temperature/ pH/ humidity/ light which would also possess the desired contact angle hysteresis of < 25 degrees which switches reversibly to > 25 degrees and would be obtained of a simple process with lower production cost to be thus economically and industrially facile thereby being attractive to the end users.
A further requirement of new generation smart surfaces in addition to the abovesaid, is the ability to dispose off the dirt deposited on the surface by a combination of hydrophobic and hydrophilic cleaning, to eventually not only prevent the deposition of the dirt but also prevent the infestation and growth of unwanted organisms, such as microorganisms, algae, lichen, and mosses. There is, therefore, a need for ambient curing, storage stable and recoatable water-based coating composition, the composition which even when applied on a surface restores a balance between hydrophobicity and hydrophilicity such that it prevents the appearance of water marks and prevents attachment of dirt and the like, and above all would maintain its most important attribute of reversible switching between hydrophobicity and hydrophilicity even after long exposures to the environment, which in addition would also exhibit good resistance to bird droppings and oily stains and enhanced photocatalytic activity even under diffused sunlight with negligible chalking.
Hence there is a longfelt need for such a substrate comprising substantially hydrophobic coating as abovesaid which when exposed to at least one external stimulus such as UV, pH, humidity or temperature would cause the substrate to switch from the first substantially
hydrophobic state to a second substantially hydrophilic state, with a provision to produce such smart coatings by facile, economical and simple techniques for manufacturing on a large scale and applied by conventional techniques such as brush and spray application that would thus exhibit superhydrophobicity under certain environmental conditions of high temperature/ low pH/ low humidity/ low intensity of light and superhydrophilicity at low temperature/ high pH/ high humidity/ high intensity of light.
OBJECTS OF THE INVENTION
It is thus a primary object of the present invention to provide for a stimuli responsive self- cleaning aqueous paint/ coating composition involving coatings that are durable, recoatable and storage stable and comprises of smart polymers in combination with at least partially surface treated particles including a particulate mixture that would be responsive to external stimuli related to certain environmental conditions such as variations in temperature, pH, light and humidity to thus exhibit super-hydrophobic and super-hydrophilic behaviour, and processes for manufacturing the same.
Another object of the present invention is to provide for a stimuli responsive self-cleaning aqueous paint/ coating compositions and self-cleanable surfaces which would be oleophobic and exhibit the desired reversible hydrophilic-hydrophobic switching to provide for a water contact angle hysteresis of <25° under certain environmental conditions with variations in at least one or more of temperature, humidity, pH, light that would be switchable to a water contact angle hysteresis of >25° under said environmental conditions offering benefits of both superhydrophobic and superhydrophilic cleaning, water marks, good organic and inorganic dirt pickup resistance, resistance to dirt streaks, dirt lines, water spotting, good cleanability with water and forced air or wind, reduced chalking, good resistance to organic dirt and deposits such as bird droppings and oily Stains, and reduced tendency for spreading of water containing rust.
It is another object of the present invention to provide for stimuli responsive self-cleaning aqueous coating composition involving polymers that would be recoatable with low coating surface energy preferably in the range of < 40 mN/m, more preferably < 20 mN/m and most preferably < 10 mN/m.
It is yet another object of the present invention to provide for said stimuli responsive self- cleaning coating composition with low contact angle hysteresis (<25 degrees, preferably <10 degrees and more preferably<5 degrees) that would enable the flow of rain water or
forced air to thereby clean the dirt from the coating surface and would also display good resistance to water containing rust i.e. corrosion marks, bird droppings.
It is yet another object of the present invention to provide for said stimuli responsive self- cleaning coating composition having high durability and negligible chalking of organic binders involved in the composition with photocata lytic particles and yet maintaining the stimuli responsive behaviour.
It is another object of the present invention to provide enhanced photocata lytic activity of the said composition even in the presence of very low contents of photocatalytically active particles in the said particulate mixture that would be photocatalytically active even in diffused sunlight thus facilitating use in both interior and exterior.
It is yet another object of the present invention to provide for said stimuli responsive self- cleaning coating composition with enhanced photocatalytical activity based on the presence of either nano anatase titania or even micronized rutile grades of Ti02.
Still another object of the present invention is to provide a super hydrophobic coating, which would be so hydrophobic that water and even muddy water will bounce off the surface of nominally horizontal coated plates, and which would also be preferably styrene free to give a coating with reduced chalking behavior and high durability.
It is yet another object of the present invention to provide for said coating composition involving low contents of fluorine monomer, thus offering economic benefits and yet possessing said stimuli responsive self-cleaning coating composition with enhanced photocatalytical activity together with reduced chalking behavior, high durability and oleophobicity along with good dirt pickup resistance.
Another object of the present invention is to provide said coatings that are practical in use, that do not degrade rapidly in sunlight and have enough resistance to abrasion to survive for a practical length of time, preferably for several months or even several years depending upon environmental conditions to which the coating is exposed, which would not lose said hydrophobic-hydrophilic switching properties even over a period of time.
It is another object of the present invention to provide for said self-cleaning coating composition that cures by evaporation of the water and does not require any special treatment, such as heating or exposing to IR or UV light to cure. It is yet another object of the present invention to provide a hydrophobic self-cleaning composition which upon application to a surface forms a uniform coating by drying and evaporation of water forming a coating or film at ambient temperature within 5 to 20 minutes. It is another object of the present invention to provide for said self-cleaning hydrophobic coating which is easy to apply, in the form of a liquid or paste which can be diluted with water or suitable solvent.
It is another object of the present invention to provide for a process to reach to said stimuli responsive self-cleaning paint/ coating composition that would be based on simple mixing of components applicable through simple techniques such as brush, roller, spray applications or dip coating, which process would be simple, industrially and economically facile to thereby lower the production cost to be thus attractive to the end users. SUMMARY OF THE INVENTION
It is thus the basic aspect of the present invention to provide for an aqueous coating composition exhibiting reversible hydrophilic-hydrophobic switching with the variations in temperature, humidity, pH and or light comprising:
a) One or more oligomeric or polymeric dispersion/binder comprising acrylic, fluorinated and hydrophilic (acid/amine/ionic/non-ionic) backbone; and
b) One or more hydrophobic organosilicone or silane (monomeric, oligomeric or polymeric) c) One or more hydrophilic organosilicone or silane (monomeric, oligomeric or polymeric) and
d) a particulate mixture comprising a plurality of particles at least partially surface treated (with a balance of hydrophobicity and hydrophilicity) with a particle size less than or equal to 100 microns, preferably less than or equal to 325 mesh (44 microns)
e) Wetting and dispersing agents,
f) At least one fungicide, algaecide and in-can preservative
g) Optionally contains one or more nano particles exhibiting photocatalytic properties, less than 1% and preferably less than 0.5% of total formulation especially for coatings containing particles equal to and over 325 mesh to selectively provide a balance of
hydrophobicity and hydrophilicity such as to provide the desired reversible hydrophilic- hydrophobic switching.
Preferbaly, said aqueous coating composition is oleophobic and has a water contact angle hysteresis of < 25° under certain environmental conditions with variations in at least one or more of temperature, humidity, pH, light which is reversibly switchable to a water contact angle hysteresis of >25° under said environmental conditions offering benefits of both superhydrophobic and superhydrophilic cleaning, good organic and inorganic dirt pickup resistance, resistance to dirt streaks, dirt lines, water spotting, water marks, good cleanability with water and forced air or wind, reduced chalking, good resistance to organic dirt and deposits such as bird droppings and oily stains, and reduced tendency for spreading of water containing rust.
Said coating has excellent resistance to microbial growth, particularly algae and also exhibits enhanced photocatalytic activity for even rutile grades of Titania, even under diffused sunlight.
It is thus unexpectedly found by way of the present invention that when one or more oligomeric or polymeric dispersion/binder comprising acrylic, fluorinated and hydrophilic (acid/amine/ionic/non-ionic) backbone is selectively present in combination with at least partially surface treated particles, , hydrophobic and hydrophilic silanes, an aqueous coating composition exhibiting reversible hydrophilic-hydrophobic switching under variable environmental conditions including variations in at least one or more of temperature, pH, humidity or other external stimuli including light can be thus achieved with low contact angle hysteresis of < 25 degrees that is reversibly switchable to a contact angle hysteresis of >25°. The method of preparation of said aqueous coating composition is simple and industrially facile which coating composition in being adapted for both brush or spray type applications is also thus attractive to the consumers.
Surprisingly, said silicones both hydrophobic and hydrophilic when combined with the abovesaid ingredients including the particles with a balance of hydrophobicity and hydrophilicity, leads to a surface similar to desert beetle with the protrusions which arise from a plurality of particles being either hydrophobic or hydrophilic. Said silicones in the composition while advantageously facilitates to retain the stimuli responsive behavior and reversible wettability switching upon long exposures to environmental conditions, also provides for better weather durability and improved water resistance of the dried film. The effect of the silicone crosslinking further enables the coating to maintain a high contact angle conducive for self cleaning even after weathering of the coating over a period of time.
The coating composition of the present invention thus also exhibits good resistance to organic dirt and deposits such as bird droppings and oily stains even with lovy contents of fluoromonomers (<5%), wherein enhancement in photocata lytic activity are also exhibited even under diffused sunlight with low contents of nano anatase titania or even micronized rutile grades of Ti02 with negligible degradation of the organic binder, subject to the selectively high particle to binder ratio with better dispersion of the photocata lytic particles due to the involvement of hydrophobic and hydrophilic silicones in a selective polymeric dispersion. According to another preferred aspect of the present invention said aqueous coating composition reversibly switches from superhydrophobic (contact angle hysteresis <25° and water droplet rolling effect) to superhydrophilic (contact angle hysteresis >25° and complete wetting) for at least 2000 hours of accelerated exterior exposure (Atlas and QUV testing), preferably the contact angle hysteresis values are <20°, more preferably <10° and most preferably <5° in one state and higher in the switched state for a deposited 0.02 mL water droplet, wherein preferably the contact angle exhibited in one state is >100° with water, more preferably >120°, even more preferably superhydrophobic having contact angle >150° and contact angle <90° in the switched state for a deposited 0.02 mL droplet; and wherein said switching takes place within a varying period of time from a few seconds to several minutes dependent on a particular stimuli.
Preferably, said aqueous coating composition has low surface energy of <40 mN/m and preferably <20 mN/m and most preferably <10 mN/m even with the second coat and hence recoatable to enhance film ' build-up to attain desired opacity/ translucency/ hiding/ whiteness.
According to a preferred aspect of the present invention there is provided said aqueous coating composition comprising at least one or more wetting and dispersing agents, encapsulated or free antimicrobial agents selected from fungicide, algaecide, in-can preservative and optionally involving, one or more nanoparticles exhibiting photocata lytic activity.
Preferably, said aqueous coating composition especially favours photocatalytic activity even under diffused sunlight even with cost effective grades of micronized particles such as Titania while maintaining its durability together also exhibiting antimicrobial properties towards microorganisms such as algae, fungi and bacteria.
According to yet another preferred aspect of the present invention there is provided said aqueous coating composition wherein said polymeric dispersion comprises dispersion polymers involving primary or secondary dispersion in water or in a water/ co-solvent mixture with or without emulsion/latex polymers.
Preferably said polymeric dispersion comprises dispersion polymers with or without emulsion/ latex polymers that is preferably styrene free and is a free radical polymerization or controlled radical polymerization product of hydrophobic monomers C1-C8 straight, branched or cyclic esters of methacrylic acid/ acrylic acid selected from methyl methacrylate, ethyl methacrylate, butyl Methacrylate, isobutyl ethacrylate, tertiary butyl methacrylate, isobornyl methacrylate, 2-ethylhexyl methacrylate and various isomers and their corresponding acrylates also involving >C8 esters of methacrylic acid/ acrylic acid selected from lauryl acrylate, stearyl acrylate. More preferably, said polymeric dispersion comprises dispersion polymers with or without emulsion/ latex polymers and has monomers selected such that the resulting polymer has an average octanol-water partition coefficient (log Kow) greater than 0.6, preferably greater than 1.5 and more preferably greater than 2. According to another preferred aspect of the present invention there is provided said aqueous coating composition wherein said polymeric dispersion including dispersion polymers with or without emulsion/ latex polymers in being styrene free involves styrene or alkylstyrene group as part of the polymer or as a part of additives including opacifiers in less than 15% of the' film forming composition favouring reduced chalking of said composition on UV exposure.
Preferably in said aqueous coating composition said polymeric dispersion comprising dispersion polymers and emulsion/ latex polymers is in the ratio range of dispersion to emulsion polymers of 95:5 to 5:95.
More preferably, in said aqueous coating composition said polymeric dispersion comprising dispersion polymers and emulsion/ latex polymers is preferably in the ratio range of 30:70.
According to yet another preferred aspect of the present invention there is provided said aqueous coating composition wherein said polymeric dispersion comprising dispersion polymers includes primary or secondary dispersion in water or in a water/ co-solvent
mixture involves co-solvent that is VOC exempt under Green Seal standard GS-11 with resulting coating VOC's in the range of 1 to 100 grams/liter.
In said aqueous coating composition said polymeric dispersion comprising dispersion polymers with or without emulsion/ latex polymers includes polymers selected from natural polymer, a neoprene polymeri a nitrile polymer, a vinyl acrylic polymer, acrylic polymer; styrene acrylic polymer, styrene butadiene polymer, copolymer of vinyl acetate and butyl acrylate or veova monomers or ethylene/ propylene/ butylene, butadiene, isoprene involving crosslinkers selected from diacrylates, triacrylates, multifunctional acrylates, silane functional monomers, acetoacetate functional monomers, monomers with aziridine, hydrazine, carbodiimide or allylic functionality.
According to yet another aspect of the present invention there is provided said aqueous coating composition wherein said polymeric dispersion comprising dispersion polymers with or without emulsion/ latex polymers includes polymers of fluorinated monomers comprising 2 to 20 carbon atoms and polymeric fluorine content in the range of 0.1 to 50% on monomer solids and preferably in the range of less than 5% on monomer solids resulting in economic benefits and a surface energy of <50 mN/m , preferably <30 mN/m and more preferably <20 mN/m
Preferably, in said aqueous coating composition said polymeric dispersion comprising fluorinated backbone comprises fluorinated monomers including esters of acrylic or methacrylic acid with a linear or branched perfluoroalkyl functional group preferably involving 1-6 fluorinated carbons selected from trifluoroethyl 'meth(acrylate), pentafluoropropyl meth (acrylate), heptafluorobutyl meth(acrylate).
' More preferably, said aqueous coating composition comprises said ionic or ionizable or non- ionic hydrophilic monomers selected from acrylic acid, methacrylic acid and their aklali metal or quaternary ammonium ion salts; monoolefinic sulphonic acid compounds and their alkali metal salts including 2-acrylamido-2-methylpropanesulphonic acid; acrylates or methacrylates of aminoalcohols, acrylamides selected from ΙΜ,Ν- dimethylaminopropylacrylamide or N, N-Dimethylaminoethylmethacrylate, polyalkylene oxide group containing monomers or a polymeric chain prepared by polymerizing a polyalkylene glycol (meth)acrylate, hydroxyl alkyl (ethyl, propyl or butyl) (meth)acrylates, wherein the hydrophilic component comprises at least one oxygen, sulfur, phosphorus and/or nitrogen atom.
According to another preferred aspect of the present invention there is provided said aqueous coating composition wherein said acrylic, fluorinated and amine backbone or acrylic, fluorinated and acid backbone of the primary or secondary polymeric dispersion comprises sufficient content of acid or amine monomer including ionic or non-ionic external surfactants adapted for acid or amine value in the range 5 to 100 mg KOH/g.
According to yet another preferred aspect of the present invention there is provided said aqueous coating composition wherein said particles comprising either micron sized particles or including particulate mixture of both micron sized and nano sized particles (mixture of micron sized and nano sized particles) and polymeric dispersion are present in selective ratios of 0.9 to 2.5 wherein higher ratios closer to 2 is preferred for a two coat application.
Said aqueous coating composition including said particles comprises either micron sized particles or includes particulate mixture of both micron sized and nano sized particles, and includes particulate pigments wherein the pigment volume concentration (PVC) is in the range of about 40 to 90 % and preferably in the range of 60 to 80% for the coating and preferably the coating PVC is lower than the practical critical pigment volume concentration (CPVC).
Preferably, in said aqueous coating composition said particles comprising either micron sized particles or including particulate mixture of both micron sized and nano sized particles comprises microhized and nanosized particles present in selective ratios of 3.5 to 8 with ratios closer to 8 being preferred for a two coat application.
More preferably, in said aqueous coating composition said at least partially surface treated particles involve organic and/or inorganic particles having size ranging from 1 nm-100 microns includes surface treated nanometer sized organic and/or inorganic particles have size ranging from about 1 nm to 300 nm, and wherein said particles preferably has mean particle size less than 325 mesh (44 microns) and even more preferably less than 40 microns.
In another preferred aspect of the present invention there is provided said aqueous coating composition wherein the mass ratio of said hydrophobic organosilicone to hydrophilic organosilicone is in the range 10/90 to 60/40.
Preferably, in said aqueous coating composition said hydrophilic organosilicone/silane having a contact angle hysteresis of >10 degrees based on the average of readings for 10 and 20 pL of water when applied on an aerated cement block is present in the range of 10% to 90% by weight of dispersion solids in said composition.
More preferably, in said aqueous coating composition said hydrophobic organosilicone/silane having a contact angle hysteresis of <10 degrees based on the average of readings for 10 and 20 μΙ_ of water when applied on an aerated cement block is present in the range of 5% to 80% by weight of dispersion solids in said composition.
Preferably, in said aqueous coating composition said hydrophobic and hydrophilic organosilicone content present as a part of the continuous phase in said composition is 10- 90 % of the weight of the dispersion/binder. In yet another aspect of the present invention there is provided said aqueous coating composition wherein said wetting agent and dispersing agent is balanced by having said dispersing agent upto 1% and preferably in the range of 0.1 to 0.5% and the wetting agent upto 2% and preferably in the range of 0.2 to 1% by weight in said composition favouring accelerated storage stability for atleast 30 days at 55°C and a composition that passes at least 5 cycles of freeze thaw stability test (-15°C to 30°C).
Aqueous coating composition as claimed in anyone of the preceding claims including photocatalytically active agents comprising one or more nano particles exhibiting photocatalytic properties in amounts less than 1% and preferably less than 0.5% of total formulation especially for coatings containing particles more than or equal to 325 mesh, preferably comprise nano titania of the anatase or rutile grade in the range of 0.1 to 1% of the formulation with a particle size of 1 to 300 nm and more preferably comprise titania in anatase form adapted for enhancement of photocatalytic activity of said composition (while - maintaining its durability) against organic pollutants of atleast >5%, more preferably >20% and most preferably >80% of the activity of the control. Enhanced photocatalytic activity of coating with good durability is also demonstrated by even micron sized particles (>300 nm) such as Titania (anatase or rutile).
Said aqueous coating composition including photocatalytically active agents comprising one or more nano particles exhibiting photocatalytic properties in amounts less than 1% and preferably less than 0.5% of total formulation especially for coatings containing particles
more than or equal to 325 mesh, preferably comprise nano titania of the anatase or rutile grade in the range of 0.1 to 1% of the formulation with a particle size of 1 to 300 nm and more preferably comprise titania in anatase form adapted for enhancement of photocata lytic activity of said composition while maintaining its durability by even micron sized particles (>300 nm) of Titania (anatase or rutile) against organic pollutants of atleast >5%, more preferably >20% and most preferably >80% of the activity of the control.
Preferably in said aqueous coating composition said at least partially surface treated particles are selected from substantially spherical particles, nodular particles, platy particles, cubical paraticles, various irregularly shaped particles, and mixtures thereof. According to yet another preferred aspect of the present invention there is provided said aqueous coating composition wherein said at least partially surface treated particles comprise at least one material selected from the group of silicates, doped silicates, minerals, metal oxides, silicas and metal powders and are selected from the group of aluminum oxides (alumina), titanium oxide, zirconium oxide, silver, nickel, nickel oxide, iron oxide, and alloys, polystyrene particles, (meth)acrylate particles, PTFE particles, silica particles, polyolefin particles, polycarbonate particles, polysiloxane particles, silicone particles, polyester particles, polyamide particles, polyurethane particles, ethylenically unsaturated polymer particles, polyanhydride particles, biodegradable particles, polycaprolactone, nanofibers, nanotubes, nanowires, or combinations thereof that are preferably insoluble in organic solvents and water; and preferably comprise particles other than respirable crystalline silica wherein said silica is amorphous.
According to another preferred aspect of the present invention there is provided said aqueous coating composition wherein said at least partially surface treated particles comprise particles that are surface treated with compounds selected from silanes (alkylsilanes, perfluoroalkylsilanes, and alkyldisilazanes), silicones and fluorinated compounds or fluorinated acrylics or other organic/ inorganic treatments to achieve a balance between hydrophobicity and hydrophilicity. Preferably, in said aqueous coating composition said at least partially surface treated particles comprise preferably externally surface treated particles with selective hydrophobic and hydrophilic balance and are obtained of sol-gel treatment to prevent any colour change or graying of white particles present in said coating composition, especially where fluorosilanes or fluorosilicones are used for treatment or is alternatively in-situ treated while preparation of the coating with said mixture of hydrophobic and hydrophilic silicones/silanes.
More preferably, said aqueous coating composition is flowable comprising at least one organic diluent or solvent, wherein the solvent is selected from the group comprising of water, an alcohol, an ether, a ketone, an ester, a glycol, a glycol ether, an alkylene carbonate, a C5 - QL8 aliphatic hydrocarbon, a C6 - Ci8 aromatic hydrocarbon, various
polar/non-polar solvents and mixtures thereof.
In said aqueous coating composition said polymeric dispersion comprises thermoplastic or thermosetting polymers including a single polymer or a blend of more than one polymer or self crosslinking polymers selected from acrylics, polyesters, polyurethanes, polycarbonates, polyolefins, alkyds, epoxies, polyamides, fluoropolymers, silicone polymers and their hybrids, wherein said fluoropolymers include polyperfluoropolyethers, and a polymer having one or more monomer repeat units selected from the group consisting of ethylene, propylene, styrene, tetrafluoroethylene, vinylidene fluoride, hexafluoropropylene, perfluoro(methyl vinyl ether), perfluoro(ethyl vinyl ether), and perfluoro(propyl vinyl ether) and/ or includes a colloid, a latex, or a suspension of a fluoropolymer selected from a group comprising of polytetrafluoroethylene; perfluoroalkoxy polymer resin; copolymer of tetrafluoroethylene and hexafluoropropylene; copolymers of hexafluoropropylene and vinylidene fluoride; terpolymers of tetrafluoroethylene, vinylidene fluoride, and hexafluoropropylene.
Preferably, in said aqueous coating composition said polymeric dispersion comprising polymers have a Tg of -30 to 100 deg C or preferably Tg in the range of 0 to 50°C. More preferably, said aqueous coating composition wherein said polymeric dispersion comprising polymers have surface energy of <50 ml\l/m and preferably less than 30 mN/m and more preferably <20 mN/m.
Said aqueous coating composition comprise components that are multifunctional or having individual functions selected from thickeners, biocides, wetting and dispersing agents, fluorosurfactants, defoamers, opacifying polymers, cosolvents, coalescents, plasticizers, pigments, special effect pigments, extenders, colorants, freeze thaw stabilizers, buffers, fire retardants, uv absorbers, organic fiber material, and inorganic fiber materials, flow and levelling agents, adhesion promoters, oil and water repellents and other components known to be provided in paint formulations.
According to another preferred aspect of the present invention said aqueous coating composition is ambient curing including air curing, infra-red curing and thermal curing. According to yet another preferred aspect of the present invention there is provided said aqueous coating composition wherein a dry film of said coating has a dry film thickness of 1
to 200 microns and preferably 40 to 120 microns and withstands at least 30 to 1000 cycles and preferably 100 to 3000 cycles in a wet scrub resistance test with a scrub brush and a 0.45 kg load. Said aqueous coating composition is adapted for applications comprising self cleaning, easy cleaning, anti-graffiti, stain resistant, soil-degrading, deodorizing, air-cleaning, antifouling, antimicrobial, water-cleaning, superhydrophobicizing, superhydrophilicizing and/or antifogging effects. According to another aspect of the present invention there is provided a process for the preparation of said aqueous coating composition comprising the steps of:
a) providing at least partially surface treated organic or inorganic particles to selectively provide a balance of hydrophobicity and hydrophilicity that is either micron sized or includes a particulate mixture of micron sized and nanometer sized particles;
b) providing for one or more oligomeric or polymeric dispersion/binder comprising acrylic, fluorinated and hydrophilic (acid/amine/ionic/non-ionic) backbone;
c) providing for one or more hydrophobic organosilicone or silane (monomeric, oligomeric or polymeric) and one or more hydrophilic organosilicone or silane (monomeric, oligomeric or polymeric) ;
d) mixing said particles with said polymeric dispersion and silane; to obtain said aqueous coating composition therefrom having desired reversible hydrophilic-hydrophobic switching adapted for coating various types of substrates by brush, spray or roller type applications and also dipping, padding, doctor blade, wipe, wet-on-wet procedure or spin coating type applications.
According to yet another preferred aspect of the process for the preparation of said aqueous coating composition said at least partially surface treated organic or inorganic particles is provided by subjecting the particles to sol-gel treatment with compounds selected from silicones, fluorinated compounds to prevent color change of the particles.
According to another preferred aspect of the process for the preparation said step (a) of providing treated organic or inorganic particles that is either micron sized has a mean particle size of less than or equal to 100 microns, preferably less than or equal to 325 mesh (44 microns) or providing a particulate mixture of micron and nanometer sized particles involves providing micro and nanosized particles in selective ratios of 3.5 to 8, and are externally or in-situ treated particles with a balance of hydrophobicity and hydrophilicity;
wherein said step (d) comprising mixing said particles preferably in a slurry form in a water miscible co-solvent to the polymer dispersion including organosilicone or silane is mixed in selective ratios of 0.9 to 2.5. According to yet another preferred aspect of the said process of preparation of aqueous coating composition that is adapted for substrates selected from masonary, concrete, cementitious, plaster, baked clay tiles, celiulosic, wood, one or more polymer, dry or damp surfaces, brick, tile, stone, grout, mortar, composite materials, gypsum board, , porous and non porous surfaces, interior surfaces or surfaces exposed to weathering on at least one surface of the substrate, which surface is preferably pretreated for better adhesion of the aqueous coating composition with various types of surface treatments.
According to another aspect of the present invention there is provided a commercial or industrial material, a structure or building or a substrate comprising said aqueous coating reversible switchable composition exhibiting reversible hydrophilic-hydrophobic switching under variable environmental conditions including variations in at least one or more of temperature, pH, humidity or other external stimuli including light.
According to yet another aspect of the present invention there is provided a method of producing reversible switchable coated commercial or industrial material, structure or building or substrate comprising
selectively applying the aqueous coating of said reversible switchable composition to atleast a portion of commercial or industrial material, a structure or building or a substrate requiring said" reversible coating characteristic such as to exhibit reversible hydrophilic- hydrophobic switching under variable environmental conditions including variations in at least one or more of temperature, pH, humidity or other external stimuli including light.
BRIEF DESCRIPTION OF FIGURES:
Fig 1: reveals contact angle measurement showing a hydrophobic and hydrophilic drop;
Fig 2: reveals contact angle measurement showing the advancing and the receding angle
Fig 3: reveals the image of untreated Ti02;
Fig 4: reveals the image of treated "Π02;
Fig 5: reveals Consecutive images of 5 pL incrementing volumes of water droplet from the syringe of the contact angle meter, with 45 pL forming a droplet having a contact angle of 143°;
Fig 6: reveals rheogram (temperature sweep at constant shear rate) for the water based pigmented coating system;
Fig 7: reveals differential scanning calorimetry thermogram for the dried pigmented coating; Fig 8: reveals scanning electron micrograph of the paint surface showing 2 levels of topography;
Fig 9: reveals an AFM picture of the paint composition;
Fig 10: reveals an AFM picture showing the thermoresponsive behaviour of the paint (left: hot condition, right: cold condition)
Fig 11: reveals a photograph of the paint exposed outdoors for a period of 6 months showing its resistance to dirt streaks and water marks; (left side: paint according to present invention, right side: commercially available self cleaning paint).
DETAILED DESCRIPTION OF THE INVENTION As discussed hereinbefore, the present invention provides for stimuli responsive self- cleaning water-borne pigmented aqueous coating composition responsive to at least one or more variations of temperature, pH, humidity or other external stimuli including light under variable environmental conditions that exhibits reversible switchable super hydrophilic- super hydrophobic characteristics. The said coating in comprising one or more oligomeric or polymeric dispersion/binder comprising acrylic, fluorinated and hydrophilic (acid/amipe/ionic/non-ionic) backbone selectively present in combination with at least ; partially surface treated particles, hydrophobic and hydrophilic silanes is thus adapted for a stimuli responsive self-cleaning aqueous paint/ coating compositions and self-cleanable surfaces, which is oleophobic and exhibit the desired reversible hydrophilic-hydrophobic switching to provide for a water contact angle hysteresis of <25° under certain environmental conditions with variations in at least one or more of temperature, humidity, pH, light that is switchable to a water contact angle hysteresis of >25° under said environmental conditions offering benefits of both superhydrophobic and superhydrophilic cleaning, good organic and inorganic dirt pickup resistance, resistance to dirt streaks, dirt lines, water spotting, water marks, good cleanability with water and forced air or wind,
reduced chalking, good resistance to organic dirt and deposits such as bird droppings and oily stains, and reduced tendency for spreading of water containing rust.
Said particles comprising at least partially surface treated particles are functionalized pigments or extenders present in selective sizes and ratios involving particulate mixture in selective ratios of micro and nano sized particles in said polymeric dispersion. More specifically, said pigments are surface treated/ functionalized either in-situ or prior to incorporation in the coatings and are present in said polymeric dispersion under a selective binder to pigment/ filler ratio such as to favour the desired contact angle hysteresis of < 25 degrees.
Advantageously, said attainment of low contact angle hysteresis (<25 degrees) and subsequent reversible wettability switching by the aqueous coating composition/ stimuli responsive self-cleaning coatings of the present invention enables the flow of rain water or forced air to clean the dirt from said coating/ paint surface wherein said coating can be formed by simple mixing of the components and applied by simple techniques such as brush and spray application.
The present invention in providing for at least partially surface treated particles including particles based on a mixture of micro and nanosized particles with a balance of hydrophobicity and hydrophilicity, a combination of silicones both hydrophobic and hydrophilic in combination with a suitable dispersion polymer leads to a surface similar to the desert beetle, with the protrusions which arise from a plurality of particles being either hydrophobic or hydrophilic.
Within the scope of the present invention, the hydrophobic or hydrophilic polymer coating shall be understood as a polymer coating which can change reversibly from a hydrophobic state into a hydrophilic state. The surface is therefore "switchable" and reversibly changed from one state to the other as a result of the influence of an external parameter. This external stimulus includes temperature change or humidity change or change in the intensity of UV light or change in the pH value.
Preferably, at a pH value of less than approximately 7.0, the polymer coating is hydrophobic and at a pH value of greater than approximately 7.0 it is hydrophilic. The coating, however, changes from a hydrophobic state into a hydrophilic state and vice versa at another suitable pH value, which preferably ranges between 3.0 and 10.0.
Preferably, the coating is hydrophobic at a temperature greater than ambient temperature (approximately 30° C) and is hydrophilic at a temperature of less than approximately 30° C. The coating, however, also changes from a hydrophobic state to a hydrophilic state at another suitable temperature, which preferably ranges between 5° C and 60° C.
Preferably, at a humidity of less than approximatel 50%, the polymer coating is hydrophobic and at a humidity value of greater than approximately 50% it is hydrophilic. The coating, however, also changes from a hydrophobic state into a hydrophilic state and vice versa at another suitable humidity value, which preferably ranges between 5 to 100%.
Preferably for coatings containing particles with photocatalytic activity, under high intensity light, the polymer coating is hydrophilic and under low intensity light the polymer coating is hydrophobic. The coating, however, can also change from a hydrophobic state into a hydrophilic state and vice versa under a range of light between 200 to 800 nm.
As a result of this the stimuli responsive switchability of the coating of the present invention even when applied on a substrate, thus produce a masonary coating which displays benefits of both hydrophobic and hydrophilic cleaning, washing off the lightly deposited surface dirt at the beginning of the rains or with wind and on a rainy day, enables thorough cleaning of the surface. In addition to the abovesaid, said coating also exhibits enhanced photocatalytic activity even under diffused sunlight with negligible degradation of the organic polymer due to the presence of a combination of hydrophilic and hydrophobic organosilicones and titania, thereby showing improved resistance to organic dirt and deposits such as bird droppings and oily stains allowing them to degrade with UV exposure and finally get cleaned from the surface with rain or wind. In the present invention, under normal conditions the ; perfluorinated groups and silicone groups cover the surface of the coating. At the time of the rains, however, the hydrophilic moiety contained in the copolymer and the silicone appears on the surface of the coating to provide wettability and thereby to improve the desoiling properties. This is believed to be the reason why the water repellency and desoiling properties can be obtained simultaneously that is further assisted by the selectively high particle to polymer ratio.
The applied switchable hydrophobic or hydrophilic coating comprises at least one oligomer or polymer and a plurality of treated particles (with a balance of hydrophobicity and hydrophilicity) which is suited to reversibly switch the coating from a hydrophobic state to a hydrophilic state as a result of the influence of an external parameters.
Particles treated with a balance of hydrophobicity and hydrophilicity imply that the surface of the particles are functionalized with a combination of hydrophobic and hydrophilic groups. The formulation of the present invention is thus economical in involving cost effective photocatalysts wherein even micron sized rutile or anatase titanium dioxide photocatalyst in the selective polymeric dispersion involving silicones exhibits enhanced photocatalytic activity to be activated by visible light and even diffused sunlight. The deposited dirt on the rough surface of the smart paint of the present invention is able to get cleaned by this combination of hydrophobic and hydrophilic cleaning. Thus the coating, not only prevents the deposition of dirt from the water but also prevents the infestation and growth of unwanted organisms, such as microorganisms, algae, lichen, and mosses. The silicone containing components (both hydrophobic and hydrophilic) are present in a concentration of 10 to 90% of the organic binder while maintaining the stimuli responsive behavior and also maintaining a clean surface that is free from dirt lines, streaks, water spots and algae, wherein said coating composition of the present invention displays enhanced photocatalytic activity under diffused sunlight with reduced chalking and also exhibits good resistance to organic dirt and deposits such as bird droppings and oily stains. Further said coating also has a reduced tendency for spreading of water containing rust.
Preferably, the coated substrate loses the hydrophobic property over the course of approximately one second to approximately one day, preferably over trie course of approximately one minute to approximately 10 minutes. The switching may happen within a varying period of time from a few seconds to several minutes which are stimuli dependent.
Polymer
By binders are meant, polymers, oligomers and low molecular mass substances which form a solid film on a surface. The binders serve, for example, to fix the particles on the surface of the substrate to be coated. The binder should be stable when exposed outdoors to strong UV light, rain, wind, etc. for a minimum time of one month to several years. Preferred binders used in the invention have a surface energy <50 mN/m, preferably <30 mN/m and in particular <20 mN/m. The binders generally comprise thermoplastic polymers or organic
prepolymers which undergo crosslinking by a thermal, oxidative or photochemical curing process.
The fluoropolymer could be any polymer having fluorine functionality. Examples of such polymers include, but are not limited to fluoroolefins, vinylidene difluoride-based polymers, fluoroethylene vinyl ethers. The fluoropolymer may be a thermoplastic or thermoset polymer. Fluoropolymers are available under the trade name Kynar and lumiflon and may be blended with the preferred binder. Further, to the polyfluorinated copolymer, other polymer blends may be incorporated. Further self crosslinking polymers can also be used. The polymers can be acrylics, polyesters, polyurethanes, alkyds, epoxies, urethane acrylates, homopolymers and copolymers of ethylenically unsaturated monomers, and acrylic acid/maleic anhydride copolymers known in the art.
Also suitable as co-binders are natural waxes such as beeswax, carnauba wax, wool wax, candelilla wax, and also synthetic waxes such as montanic acid waxes, montanic ester waxes, amide waxes, and also waxlike polymers of ethylene and of propylene (polyethylene wax, polypropylene wax).
As the polymerization initiating source, various polymerization initiators such as an organic peroxide, an azo compound and a persulfate may be employed. As an emulsifier for emulsion polymerization, almost all emulsifiers including anionic, cationic and non-ionic emulsifiers may be used. Catalyst concentration can be about 0.1 to 2 percent based on the weight of total monomers.
Conventional chain transfer agents, such as dodecyl mercaptan, and crosslinking agents, such as ethylene dimethacrylate, can be used in amounts of 0.1 to 2 percent by weight of the monomers to control the molecular weight of the polymer. The number average molecular weight of the binder polymers may vary over a wide range and is generally in the range from 1000 to 1 million g/mol and preferably in the range from 2500 to 0.5 million g/mol, in particular 10,000 to 0.5 million, g/mol (determined by GPC). The polymer has a Tg of -30 to 100 deg C. Alternatively a blend of polymers having different Tg's can be used. Most preferably the Tg is in the range of 0 to 50°C.
The aqueous polymer dispersions have a solid content of 25% to 70% by weight, preferably 40% to 65% by weight.
The polymer can be made by Free radical polymerization or controlled radical polymerization (CRP). CRP / living polymerization can be used to achieve a desired molecular weight and a narrow weight distribution or polydispersity. CRP provides a maximum degree of control for the synthesis of polymers with predictable and well-defined structures. Atom transfer radical polymerization (ATRP), nitroxide-mediated polymerization (NMP), reversible addition fragmentation transfer polymerization (RAFT), activated regenerated by electron transfer (ARGET) are some of the techniques of CRP.
To obtain the copolymer of the present invention, various systems and conditions for the polymerization reaction may be employed, and any of various polymerization systems such as bulk, solution, suspension or emulsion polymerization may be used.
Examples of preferred hydrophobic monomers are C2 -C24 olefins, such as ethylene, propylene, n-butene, isobutene, n-hexene, n-octene, ispoctene, n-decene, isotridecene, C5 - C8 cycloolefins such as cyclopentene, cyclopentadiene, cyclooctene, vinyl aromatic monomers, such as styrene and alpha-methylstyrene, and also fluoroolefins and fluorochloroolefins such as vinylidene fluoride, chlorotrifluoroethylene, tetrafluoroethylene, vinyl esters of linear or branched alkane carboxylic acids having 2 to 36 carbon atoms, e.g., vinyl acetate, vinyl propionate, vinyl n-butyrate, vinyl isobutyrate, vinyl hexanoate, vinyl octanoate, vinyl laurate and vinyl stearate, and also esters of acrylic acid and of methacrylic acid with linear or branched C.sub.2 -C. sub.36 alkanols, e.g., ethyl (meth)acrylate, n-butyl (meth)acrylate, tert-butyl (meth)acrylate, n-hexyl (meth)acrylate, 2-ethylhexyl (meth)acrylate, 2-propylheptyl (meth)acrylate, lauryl (meth>acrylate and stearyl (meth)acrylate and also vinyl ethers and allyl ethers of C.sub.2 -C.sub.36 alkanols, such as n-butyl vinyl ether and octadecyl vinyl ether, fluorinated monomers and also monomers containing poiysiloxane groups.
Typical binder polymers of this type are polyethylene, polypropylene, polyisobutene, polychlorotrifluoroethylene, polytetrafluoroethylene, polyvinyl acetate, polyethyl methacrylate, poly-n-butyl methacrylate, polyisobutyl methacrylate, poly-tert-butyl methacrylate, polyhexyl methacrylate, poly(2-ethylhexyl methacrylate), polyethyl acrylate, poly-n-butyl acrylate, polyisobutyl acrylate, poly-tert-butyl acrylate, poly(2-ethylhexyl acrylate), and copolymers of maleic acid with at least one hydrophobic monomer selected from C.sub.3 -C.sub.6 olefins, C.sub.l -C. sub.36 alkyl vinyl ethers, the vinyl esters of aliphatic C.sub.l -C. sub.36 carboxylic acids.
Further suitable binders are poly-C.sub.l -C.sub.4 -alkylene oxides, such as polyoxymethylene, polypropylene oxide and polybutylene oxide, polytetrahydrofuran and also polycaprolactone, polycarbonates, polyvinylbutyral, polyvinylformal, and also linear or branched polydialkylsiloxanes such as polydimethylsiloxane (silicones).
The polymer contains fluorinated monomers (comprising from 2 to 20 carbon atoms). Further the polymer contains fluorine content in the range of 0.1 to 50% on monomer solids and preferably in the range of less than 5% on monomer solids thus offering economic benefits. The fluorinated monomers can be esters of acrylic or methacrylic acid with a linear or branched perfluoroalkyl functional group.
Preferably the fluoromonomer or fluoromonomers contain up to six fluorinated carbons such as trifluoroethyl meth(acrylate), pentafluoropropyl meth(acrylate), heptafluorobutyl meth(acrylate) among others.
In the present invention, the hydrophilic moiety, a non-ionic group such as a polyoxyethylene chain, a polyoxypropylene chain, a polyoxybutylene chain, an anionic group such as a sulfonic acid group or a carboxylic acid group, or a cationic group such as an ammonium salt or an amine salt. Usually, an amphipathic monomer having a hydrophilic- lipophilic balance (HUB) of hydrophilic moiety/lipophilic moiety being from 5 to 15 is selected.
The polymer wherein the ionic or ionizable hydrophilic monomer or monomers are selected from: acrylic acid, methacrylic acjd, maleic acid and their alkali metal or quaternary ammonium ions salts; monoolefinic sulphonic acid compounds and their alkali metal salts including 2-acrylamido-2-methylpropanesulphonic acid; acrylates or methacrylates of aminoalcohols, acrylamides such as Ν,Ν-dimethylaminopropylacrylamide or N,N- Dimethylaminoethylmethacrylate, polyalkylene oxide group containing monomers or a polymeric chain prepared by polymerizing a polyalkylene glycol (meth)acrylate, hydroxyl alkyl (ethyl, propyl or butyl) (meth)acrylates. Preferably the hydrophilic component comprises at least one oxygen, sulfur, phosphorus and/or nitrogen atom.
A monomer containing at least one acid group selected from the group comprising of a carboxylic acid group, a phosphoric acid group, a phosphonic acid group, a phosphinic acid group, a sulfuric acid group, a sulfonic acid group and a sulfinic acid group.
The polymeric dispersion comprises acid or amine monomer (in the range 5 to 100 .mg KOH/g acid or amine value) and may contain external surfactants;
The amount of hydrophilic monomer is in the range of 2 to 20%. If the hydrophilic monomer content is high (>20%), the coating will have very poor water resistance, exhibit yellowing (where hydrophilic monomer is amine) and hence poor durability. Further, it will not be of any commercial importance as a coating.
In the present invention, in addition to the above-mentioned components, one or more additional monomers such as a monomer having a cross-linking group or a chelating group to the substrate, a high Tg monomer for improving the film-forming property, or a low Tg monomer for improving the flexibility of the coating film, may be incorporated without any particular restriction.
The polymer (dispersion or latex) is preferably styrene free with the preferred hydrophobic monomers being CI to C8 straight, branched or cyclic esters of methacrylic acid/ acrylic acid such as methyl methacrylate, ethyl methacrylate, butyl Methacrylate, isobutyl Methacrylate, tertiary butyl methacrylate, isobornyl methacrylate, 2-ethylhexyl methacrylate and various isomers and their corresponding acrylates. Styrene or alkylstyrene may be used as part of the polymer or through additives such as opacifiers such that it is under 15% of the film forming composition to reduce chalking tendency on UV exposure. Other long chain monomers, >C8 esters of methacrylic acid/ acrylic acid such as lauryl acrylate, stearyl acrylate etc. can also be used.
The polymer is selected from the group consisting of a natural polymer; a neoprene polymer; a nitrile polymer; a vinyl acrylic polymer; a acrylic polymer; a styrene acrylic polymer; a styrene butadiene polymer; a copolymer of vinyl acetate and butyl acrylate or veova monomers or ethylene/ propylene/ butylenes, butadiene, isoprene etc. Crosslinkers such as diacrylates, triacrylates, multifunctional acrylates, silane functional monomers, acetoacetate functional- monomers, monomers with aziridine, hydrazine, carbodiimide, allylic functionality, divinyl benzene, divinylsulfone, thallyl phosphate, zinc diacrylate, zinc dimethacrylate, diallyphthalate, diallylacrylamide, polyethylene glycol dimethacrylate, polypropylene glycol dimethacrylate, pentaerythritol tetramethacrylate, pentaerythritol trimethacrylate, pentaerythritol dimethacrylate, trimethylolpropane -tri methacrylate, dipentaerythritol hexamethacrylate, di pentaerythritol pentamethacrylate, glycerol trimethacrylate or a mixture thereof can also be used.
The monomers are selected such that the resulting polymer has an octanol-water partition coefficient (log Kow) greater than 0.6, preferably greater than 1.5 and more preferably
greater than 2 (calculated as a weighted average of the monomers used to prepare the polymer). The octanol/water partition coefficient is defined as the ratio of a chemical's concentration in the octanol phase to its concentration in the aqueous phase of a two-phase octanol/water system and can be calculated by methods known in the art such as UV/visible absorbance, use of separatory funnels, high performance liquid chromatography etc.
An aqueous coating composition wherein said polymeric dispersion comprises dispersion polymers with or without emulsion/latex polymers wherein the ratio of dispersion to emulsion polymers is preferably in the range of 95: 5 to 5:95. Neutralizing agents
Various amines can be used as the neutralizing agents for the anionic polymers such as liquor ammonia, morpholine, triethyl amine, vantex T, AMP-95, Advantex etc. Various acids can be used such as acetic acid etc. for the neutralization of the cationic polymers.
Particles type, sizes and contents
As used herein, the term "particle" is intended to include any discrete particle, primary particle, aggregate and/or aggregated collection of primary particles, agglomerate and/or agglomerated' collection of aggregates, colloidally dispersed particles, loose assemblies of particulate materials, and combinations thereof.
The particles that may be used include organic or inorganic particles currently used and known to one skilled in the art. The particles preferably have an average particle diameter of between 1 nm to 44 microns, preferably being of inorganic nature. Depending on the size and material of the particles used, it is possible to obtain translucent or opaque self- cleaning surfaces.
Surface treated micron sized particles involve organic or inorganic particles lower than 44 microns and wherein said surface treated nanometer sized organic or inorganic particles in the size range of about 1 nm to 300 nm.
Only micron sized particles can be used or a combination of micron and nano sized particles can be used to prepare the aqueous coating composition. Only nano sized particles may be used, however they increase the cost of the coating. The surface of particles should preferably have a high oil absorption value or an irregular structure. Examples of such materials include clays from Hoffmann (Silitin and Aktisil grades), high oil absorption value
titanium dioxide from DuPont e.g. Ti02 R931, calcium carbonate grades from Omya and Jorcal grades form Jordan carbonate Co. Several such materials are available such as extenders prepared using the ground process which leads to rougher surfaces as compared to the precipitation process. Cristobalite and respirable crystalline silica are not desired due to their carcinogenicity. Preferred fillers include gypsum, magnesium silicate, magnesium or calcium carbonate, pyrophyllite, mica, pumice, silica, diatomite, barium sulphate, alumina or mixtures thereof.
The filler also serves to improve the film durability, abrasion resistance, and lower cost. The filler may be present in the aqueous composition in a range of from about 2.0 percent by weight to about 50.0 percent by weight, preferably from about 5.0 percent by weight to about 40.0 percent by weight based on the total weight of the composition.
The combination of said micronized to nanosized particles is in selective ratios of 3.5 to 8 with ratios closer to 8 being preferred for a two coat application.
The particulate mixture (micron and nano meter sized) and polymeric dispersion is in selective ratios of 0.9 to 2.5 wherein higher ratios closer to 2 being preferred for a two coat application.
The micron sized and nano sized particles may be of the same chemistry or different chemistries. The nano sized particle maybe for example silicon dioxide, calcium carbonate or Teflon and the micron sized particle maybe silicon dioxide, calcium carbonate or titanium oxide. Nanoparticles that can be used to make the coatings of this invention are generally from the class of fumed silica's, titania's, zinc oxides, aluminium oxides, zirconium dioxide and tin dioxide. The average particle size may be from about 1 nm to 300 nm.
Among the inorganic substances there may be mentioned by way of example: metal oxides, mixed oxides, silicates, sulfates, phosphates, borates, metal sulfides, oxosulfides, selenides and sulfoselenides, metal nitrides and oxide-nitrides and metal powders. Among the organic structure-forming particles there may be mentioned by way of example carbon blacks and nanoscale organic polymeric particles, and among these fluorine-containing polymers. Examples of finely divided organic powders with a hydrophobic surface are finely divided polymer powders, e.g., polytetrafluoroethylene powders or C2-C4 polyolefin powders, examples being the polyethylene powders and polypropylene powders.
The structure-forming particles used are preferably those which comprise at least one material selected from silicates, doped silicates, minerals, metal oxides, silicas, and polymers. The structure-forming particles are particularly preferably naturally occurring, precipitated or pyrogenically prepared metal oxides. Pyrogenic silicas are commercially obtainable with an average primary particle size in the range from about 5 to 40 nm. In some embodiments of the invention, the silicon dioxide utilized is a colloidal silicon dioxide. Such products are commercially available from a number of sources, including Cabot Corporation, (under the trade name CAB-O-SIL) and Degussa, Inc., (under the trade name AEROSIL).
A plurality of particles of different particle sizes can be used for the coating composition with different hydrophobic and hydrophilic silicone treatments provided they are incorporated with the polymer in certain ratios which are discussed below.
Hydrophobic Titanium dioxide e.g. R101, R102, R103, R104 and R105 grade (0.31 microns average particle size) is available from companies such as DuPont, which are recommended for use in plastics products. These grades being hydrophobic are difficult to disperse into waterborne systems. Even if dispersed with high quantity of surfactants, they lead to poor hydrophobicity (low water contact angles because of high quantity of surfactants). Other hydrophobic micron sized particles are available from Hoffman Minerals under the trade name Aktisil PF777 (d50 particle size = 2.2 microns). These are a mixture of silica and kaolinite. Other Aktisil grades are also available with different silane treatments Aktisil EM (epoxysilane), AM (aminosilane) etc. Micronized PTFE is available from Micro Powders, Inc. Some examples are Fluo 300 (mean particle size 5 to 6 microns), Fluo 625F-H (13 to 21 microns), Fluo HT-LS (2 to 3 microns).
Several hydrophilic Rutile grades of titanium dioxide are available R902, R902 plus, R 900, R931 etc that can be treated. The photocatalytically active agent comprises titanium dioxide in a form selected from the group consisting of amorphous form, partially crystalline form, and anatase form.
The photocatalyst may be selected from titanium dioxide (anatase and rutile form), zinc oxide, zirconium oxide, tin oxide, vanadium trioxide, ferric oxide, dibismuth trioxide, tungsten trioxide, and-strontium titanate and combinations thereof. Titanium dioxide is preferred because of its high photocatalytic activity. It is known that the photocatalytic
activity of the photocatalysts changes in the order: titanium dioxide>zinc oxide>zirconium oxide>tin oxide>vanadium oxide.
A suitable source of light is from sunlight, diffused sunlight, a room lamp, a fluorescent lamp, a mercury lamp, an incandescent lamp, a xenon lamp, a high pressure sodium lamp, a metal halide lamp, a BLB lamp and light sources known to the person skilled in the art. In one preferred embodiment, the photocatalytically active particles present in the mixture according to the invention develop their activity on irradiation by sunlight. The preferred photocatalysts are p25 (21 nm, BET surface area 50 m2/g/ mixed anatase and rutile) from Evonik, VP Aeroperl P25/ 20 (20 pm) and LPX 21530 (30 nm) from BYK. Alternately rutile grade nanosized or micron sized Ti02 may also be used alone or in combination with nano Titania. Hydrophobic nanometer sized silica available from Evonik under the trade name Aerosil R972 (primary, particle size 16 nm), Aeroxide LEI (primary particle size 20 nm), LE2 (primary particle size 7 nm) and LE3 from Evonik. can be used.
Alternate hydrophilic grades are available such as Cabosil M5 (200 nm - 300 nm aggregates with a primary particle size of 14 nm in diameter) from Cabot or Aerosil R200 (average particle size of 12 nm) from Evonik that can be either heat treated or treated by sol-gel process.
Particles and treatment (external or internal)
It is important that the particle be treated to exhibit a balance of hydrophobicity and hydrophilicity. The hydrophilic or hydrophobic behavior of a solid is determined essentially by the groups at its surface. For example, particles which have hydroxyl groups at their surface are usually hydrophilic. However, if there are hydrocarbon groups, such as, alkyl groups, and especially longer chain alkyl groups, at the surface of the particle, then the particles have hydrophobic properties.
It has long been known that it is possible to influence the hydrophilic or hydrophobic character of inorganic or organic particles. This can be accomplished by adsorption of substances on the surface of a particle, especially by chemical reactions with reactive groups, which are at the surface of the particle.
This balance of hydrophobicity and hydrophilicity may be obtained by using a mixture of hydrophobic and hydrophilic particles which are available commercially, using particles treated with hydrophobic and hydrophilic surface modifiers (organosilanes etc.) by the sol- gel, heat treatment process or other process known in the art, using particles treated with hydrophobic and hydrophilic organosilicones during the paint making process.
It is possible that the nano sized particles are treated with hydrophobic surface groups and the micron sized particles are treated with hydrophilic surface groups or vice versa. Preferably both the particles are treated with functional groups such that they exhibit a balance of hydrophobicity and hydrophilicity.
Alternately, if the modifying agent is used in an amount less than equivalent, so that not all of the reactive groups at the surface of the particles can be modified, it may be assumed that the distribution of the modified groups on the surface follows statistical laws so that the modified particle behaves equally hydrophilic or hydrophobic at any position on its surface.
The solids with modified surface properties known from the state of the art, have hydrophobic or hydrophilic groups, which are located at the surface of the particles in a uniform random distribution. Therefore, it is only possible to hydrophobize a hydrophilic particle more or less pronouncedly. This is shown for by example dispersing treated Ti02 in water and the particles partly float on water and partly disperse in water.
During different types of treatment the surface of the particle may be modified by physisorption, chemisorption or covalent bonding with organic molecules with a variety bf hydrophobic and hydrophilic functional groups.
The hydrophobic properties are a result of a suitable hydrophobizing treatment, e.g., treatment with at least one compound from the group of the silsesquioxanes, perfluoroacrylic resins, organosilanes, alkylsilanes, fluorinated silanes, disilazanes, waxes, paraffins, fatty esters, fluorinated and/or functionalized alkanes.
Suitable organosilanes include, but are not limited to: alkylchlorosilanes; alkoxysilanes, e.g., ethyltrimethoxysilane, ethyltriethoxysilane, n-propyltrimethoxysilane, n- propyltriethoxysilane, i-propyltrimethoxysilane, i-propyltriethoxysilane, butyltrimethoxysilane, butyltriethoxysilane, hexyltrimethoxysilane, octyltrimethoxysilane, 3-
mercaptopropyltrimethoxysilane, n-octyltriethoxysilane, phenyltriethoxysilane, polytriethoxysilane; trialkoxyarylsilanes; isooctyltrimethoxy-silane; N-(3- triethoxysilylpropyl) methoxyethoxyethoxy ethyl carbamate; N-(3-triethoxysilylpropyl) methoxyethoxyethoxyethyl carbamate; polydialkylsiloxanes including, e.g., polydimethylsiloxane; arylsilanes including, e.g., substituted and unsubstituted arylsilanes; alkylsilanes including, e.g., substituted and unsubstituted alkyl silanes including, e. g., methoxy and hydroxy substituted alkyl silanes; and combinations thereof. Some suitable alkylchlorosilanes include, for example, methyltrichlorosilane, dimethyldichlorosilane, trimethylchlorosilane, octylmethyldichlorosilane, octyltrichlorosilane, octadecylmethyldichlorosilane and octadecyltrichlorosilane. Other suitable materials include, for example, methylmethoxysilanes such as methyltrimethoxysilane, dimethyldimethoxysilane and trimethylmethoxysilane; methylethoxysilanes such as methyltriethoxysilane, dimethyldiethoxysilane and trimethylethoxysilane; methylacetoxysilanes such as methyltriacetoxysilane, dimethyldiacetoxysilane and trimethylacetoxysilane; vinylsilanes such as vinyltrichlorosilane, vinylmethyldichlorosilane, vinyldimethylchlorosilane, vinyltrimethoxysilane, vinylmethyldimethoxysilane, vinyldimethylmethoxysilane, vinyltriethoxysilane, vinylmethyldiethoxysilane and vinyldimethylethoxysilane. Suitable disilazanes include for example, but are not limited to: hexamethyldisilazane, divinyltetramethyldisilazane and bis(3,3-trifluoropropyl)tetramethyldisilazane.
Cyclosilazanes are also suitable, and include, for example, octamethylcyclotetrasilazane. Suitable fluorinated silanes include the fluorinated alkyl-, alkoxy-, aryl- and/or alkylaryl- silanes, and fully perfluorinated alkyl-, alkoxy^, aryl- and/or alkylaryl-si lanes. Examples of fluoroalkyl silanes include, but are not limited to: those marketed by Degussa under the trade name of Dynasylan F8261, F8815 etc. An example of a suitable fluorinated alkoxy- silane is perfluorooctyl trimethoxysilane.
Alternately oligomeric silicones may also be used for the treatment such as DC 3074 and DC 3037 from Dow Corning, Silres SY 231 from Wacker etc. Oligomers have the benefit of having a very low vapour pressure whereas monomeric corresponding compounds have a comparatively high vapour pressure.
Hydrophilic organosilanes that may be used include tetraalkoxysilane or silanes having hydrophilic functional groups other than hydroxy groups after hydrolysis. It is preferable to use a silane having one or two hydrophilic functional groups. Particular examples thereof
include a glycidoxyalkyl group, aminoalkyl group, isocyanatoalkyl group, etc. Additionally, the hydrophilic functional group may include a thiol group, amine oxide group, sulfoxide group, phosphate group, sulfate group or salts thereof; or hydrophilic groups of polyoxyethyelne or polyoxypropylene.
More particularly, non-limiting examples of the hydrophilic silane include IM-aminoethyl gamma-aminopropyl trimethoxysilane, N-aminoethyl gamma-aminopropyl triethyoxysilane, gamma-aminopropyl trimethoxysilane, gamma-aminopropyl triethoxysilane, gamma- glycidoxypropyl trimethoxysilane, gamma-glycidoxypropyl triethoxysilane, gamma- isocyanopropyl triethoxysilane, gamma-isocyanopropyl trimethoxysilane or mixtures thereof.
The organosilane is used to treat the particles to prepare particles with a balance of hydrophobicity and hydrophilicity. They can be used suitably in an amount of 1-50 wt % based on 100 wt % of the particle content. This content varies with the chemistry and surface area of the particles to be treated. Nano sized particles may require higher quantities of silanes while micron sized particles may require lower quantities. Where fluorosilanes are used the slurry or sol-gel treatment may be preferred as compared to the heat treatment to avoid discoloration/ graying of the white particles such as titanium dioxide.
Further though external surface treatment of the particles can be performed, insitu surface treatment can also be performed. For hydrophobic surface treatment, organosilicones/ silanes such as Dow Corning 3074, Silres SY 231 from Wacker, Protectosil SC concentrate, Dynasylan F8261, Tegophobe 1500, 1401, 1505 etc from Evonik Degussa can be used to impart hydrophobicity to the pigments and extenders and subsequently to the paint film. Further one or more of the hydrophobic organosilicones/silanes can be used which are described above. For hydrophilic surface treatment, organosilicones such as Dow Corning QP8 5314, Tegophobe 1650, Wacker BS 45 etc. can be used. Further one or more of the hydrophilic organosilicones/silanes can be used which are described above. These silicones are added during the paint making process to functionalize the particles in-situ.
The hydrophobic agent may be present in the aqueous composition in a range of from about 5 to 80% by weight of polymeric dispersion. The hydrophilic agent may be present in the aqueous composition in a range of from about 10 to about 90% percent by weight of the polymeric dispersion. It is preferred to have a combination of both types of silicones
(hydrophilic and hydrophobic) to get a balance of hydrophobicity and hydrophilicity and attain a coating that exhibits good resistance to dirt lines, streaks and water spots.
The hydrophobic organosilicone/silane has a contact angle hysteresis of < 10 degrees (average of 2 readings for 10 and 20 pL of water) when applied on an aerated cement block. The hydrophilic organosilicone/silane has a contact angle hysteresis of > 10 degrees when applied on an aerated cement block (average of 2 readings for 10 and 20 pL of water). When a continuous stream of water is poured on a hydrophilic organosilicone coated aerated cement block it completely wets the surface. On the other hand the aerated cement block coated with the hydrophobic organosilicone is completely water repellent even under a continuous stream of water. One or more organosiloxanes are present as part of said continuous phase, the total organosiloxane being present in an amount of 10 to 90% of the weight of binder. The mass ratio of the hydrophobic silicone to the hydrophilic silicone is in the range 10/90 to 60/40.
PVC
The ratio of the various components may vary rather much and in many cases they may be characterized by various key figures. One of these is the pigment-volume concentration (PVC), which represents the percentage of the total volume of particles (pigments, extenders etc.) relative to the volume of non-volatile binder plus volume of particles.
If the concentration of particles in the paint is low even in the dry paint film each particle will be entirely enclosed by binder. This will also be the case with increasing particle concentration up to a certain limit at which the amount of binder'is just sufficient to enclose all of the particles. The point where the amount of binder is exactly sufficient for such enclosure is called the critical pigment-volume concentration, CPVC. At values above CPVC the dry paint film may contain voids and become porous and generally permeability, strength and gloss may hereby be influenced unfavourably; in case of outdoor paints the durability may become unsatisfactory.
Prior art uses organic silicon compounds in combination with the binder to render paints watertight or hydrophobic. However, paints with only hydrophobicity or superhydrophobicity lead to inefficient and uneven cleaning of dirt form the surface of coatings with rain water. This leads to dirt lines and dirt patches which eventually leads to microbial growth. Hence it is important according to the current invention, to use a combination of hydrophobic and hydrophilic silicones in a certain ratio with a specific binder, particular PVC and specific
particle size which lead to surfaces with very high durability and good aesthetic appearance benefitting from combined superhydrophobic and superhydrophilic cleaning effects.
The composition has a pigment volume concentration (PVC) in a range of from about 40 to 90 % and preferably in the range of 60 to 80%. Said concentration is under the practical CPVC as tested by the curling test or other suitable tests known in the art, wherein the benefit of operating below the practical CPVC (practical pigment volume concentration) could be derived by careful formulation aided by the use of dispersion polymers as compared to latex polymers in combination with functionalized particles that eventually generates coatings also having high durability and good antimicrobial properties at reduce dosages of biocides.
Solid contents The aqueous composition has a solid content in a range of from about 25 to about 65 percent, preferably in a range of from about 35 to about 55 percent. Solids content greater than about 65 percent may be less desirable in that there would be a difficulty in application and a loss of storage stability.
pH
The aqueous composition has a pH in a range of from about 5 to about 10, preferably in a range of from about 6 to about 9.0.
VOC
The water-based compositions are preferably free of volatile organic compounds. The water- based compositions have a calculated volatile organic content of < 100 g/liter and more preferably < 50 g/liter as per Green Seal standard GS-i l. The term volatile organic chemical as described herein is defined as an organic compound having a boiling point less than 280 °C at ambient pressure. Solvents "
In order to disperse treated particles into water, the particles must be wetted by one or more non-aqueous liquids such as ketones, glycol ethers, glycol ether acetates, alcohols, aliphatic hydrocarbon solvents, polydimethylsiloxane, cyclic polydimethylsiloxane, aromatic hydrocarbon solvents, tetrahydrofuran, acetic acid, acetates, and glycols. The preferred solvents are acetone and isopropyl alcohol. A non-VOC material may be selected or a VOC exempt material may be selected to produce non-VOC hydrophobic particle dispersion in
water. Example of non VOC solvents are Ecosoft PB form Dow, dimethyl carbonate, tertiary butyl acetate, Optifilm enhancer 300 from Eastman, Triethylene glycol etc.
The solvent is present at a level of less than 50 percent by weight based on the total weight of the composition, and preferably in an effective amount of between 0.001 to 50 percent by weight, more preferably between 1 to 5 percent by weight based on the weight of the total composition.
Suitable examples of less volatile organic solvents are those with lower vapor pressures, for example those having a low vapor pressure which include, but are not limited to: dipropylene glycol n-propyl ether, dipropylene glycol t-butyl ether, dipropylene glycol n- butyl ether, tripropylene glycol methyl ether, tripropylene glycol n-butyl ether, diethylene glycol propyl ether, diethylene glycol butyl ether, dipropylene glycol methyl ether acetate, diethylene glycol ethyl ether acetate, and diethylene glycol butyl ether acetate.
The composition may also comprises one or more organic solvents which are capable of dissolving the dispersion polymer. Suitable solvents are, for example, monohydric alcohols having 1 to 4 C atoms and mono-, di- and trialkylene glycol monoalkyl ethers, wherein the alkylene group usually contains 2 or 3 C atoms and the alkyl group 1 to 4 C atoms. The aqueous composition may contain at least one co-solvent. When present in the composition, the co-solvent is a means to improve film coalescence at low temperatures of below 10°C, and improve sprayability and substrate wetting. Useful cosolvents include Dowanol DPnB, PnP and PPH from Dow Chemical, Texanol from Eastman Chemical, Glycol Ether PB, EB, and DPM from Ashland Chemical. Examples of preferred cosolvents include Dowanol DPnB from Dow Chemical and Texanol from Eastman Chemical. The co-solvent may be present in the aqueous composition in a range of from about 0.20 percent by weight to about 4.0 percent by weight, preferably from about 0.40 percent by weight to about 2.0 percent by weight based on the total weight of the composition. Alternatively, co-solvent content less than about 0.20 percent by weight may be less desirable in that it would be insufficient for insuring good film formation at low temperatures.
UV Protection
The aqueous composition may contain at least one UV absorbing component. The UV absorbing component is used as a means to provide weather protection for the film forming polymer and antimicrobials used in the product by protecting them against degradation caused by UV radiation. Useful UV absorbers include conventional zinc oxide, nano zinc
oxide, titanium dioxide etc. The nanometer sized zinc oxide particles are broad spectrum UV absorbers (UV-A and UV-B) which is not the case for micro fine Ti02 and organic UV absorbers. It also has anti-bacterial properties and is mildew resistant. The UV absorbing component may be present in the aqueous composition in a range of from about 0.1 percent by weight to about 50 percent by weight of the dry composition. Other UV absorbing components useful include Tinuvin 292 1130, and 5151 from Ciba Specialty Chemical, These UV absorbing components may be present in the aqueous composition in a range of from about 0.10 percent by weight to about 5.0 percent by weight of the composition.
Preservatives (Algaecide, Fungicide, Incan preservative)
It is essential that the formulation contains at least one or more biocides which can be in liquid or solid form. Suitable types of incan preservatives, antifungal agents and anti algal agents and their contents can be decided by carrying out tests to determine a level required for long term protection depending upon the degree of exposure. Some biocides which can perform a multifunctional role of antifungal and antialgal can also be used. Antibacterial film properties can be conferred by suitable antibacterial agents such as silver etc. Further, encapsulated biocides can also be used.
The aqueous composition contains at least one algaecide. Useful algaecides include Terbutrin, Rocima 344, Rocima 350, Rozone 2000, etc. The algaecide (active content) is present in the aqueous composition in a range of from about 0.05 percent by weight to about 3.0 percent by weight, preferably from about 0.10 percent by weight to about 2.0 percent by weight based on the total weight of the composition.
The aqueous composition contains at least one fungicide. Useful fungicides include Zinc and Sodium Omadine from Arch Chemical, Rozone 2000, Rocima 200, Rocima 63, and Skane M- 8 from Rohm & Haas, Mergal S-90, Polyphase 678, and Mergal S-89 from Troy Chemical, and Fungitrol 720 from ISP Corp. The fungicide (active content) is present in the aqueous composition in a range of from about 0.05 percent by weight to about 3.0 percent by weight, preferably from about 0.10 percent by weight to about 2.0 percent by weight based on the total weight of the composition.
A biocide, such as DANTOGARD, TROYSAN 395, Nipacide CFX, Thor MBS etc. can be used as a preservative in the product. The preservative provides a useful shelf life to the product. The biocide preservative is added in an effective amount to preserve the composition
product and ranges from 0.01 to 1.0 percent by weight, and more preferably in a range of from 0.05 to 0.9 percent by weight.
In situations where multiple types or species of organisms are likely to be found, two or more biocides may be required in other to inhibit the growth of all of them. Preferably, the biocides have low water solubility (less than about 50 parts per million), excellent resistance against hydrolysis, be resistant to degradation by UV where exposed to sunlight (unless sufficient UV absorbers are used) and should be free from formaldehyde.
Wetting and dispersing agents
Surfactants may also be used to help wet the surface in order to form a uniform coating. Surfactants can be nonionic, cationic, amphoteric, or anionic in nature.
The aqueous composition contains at least one dispersant. The dispersant is a means to disperse the pigments and prevent agglomeration on product aging. Useful dispersants include BYK 190, 191, 194 from BYK Chemie, AMP-95, Orotan 850, Orotan 731 etc from Dow Chemical Company. The dispersant is present in the aqueous composition in a range of from about 0.10 percent by weight to about 1.0 percent by weight, preferably from about 0.20 percent by weight to about 0.5 percent by weight based on the total weight of the composition. Dispersant content greater than about 1.0 percent by weight may be less desirable in that it could adversely affect viscosity stability and water resistance of the dried film. Alternatively, a dispersant content less than about 0.10 percent by weight may be less desirable in that the product would show poor viscosity stability on aging, pigment agglomeration.
The aqueous composition contains at least one surfactant. The surfactant is a means to lower surface tension for improved surface wetting, pigment dispersion and polymer stability. Useful surfactants include Triton X-100 and Triton X-405 from Dow Chemical, Surfynol series from Air Products, Cresmer series from Croda. The surfactant is present in the composition in a range of from about 0.10 percent by weight to about 2.0 percent by weight, preferably from about 0.20 percent by weight to about 1.0 percent by weight based on the total weight of the composition. Surfactant content greater than about 2.0 percent by weight may be less desirable in that it would hamper the dried film's water resistance and durability. Alternatively, a surfactant content less than about 0.10 percent by weight may be less desirable in that it would not sufficiently lower the surface tension for adequate surface wetting and aiding in pigment dispersion.
Other suitable surfactants include non-ionic surfactants having an HLB value of from between 9-13, branched ethoxylated alcohols, linear ethoxylated alcohols, and silicone surfactants. These surfactants are sold under the trade names of Tomah, Triton, Surfonic, Igepal, Alfonic, Rhodasurf, Synperonic etc.
It is preferred to have certain ratios of the non-ionic wetting agent as compared to the ionic dispersing agent in the composition as shown in example number 56 to achieve a good accelerated storage stability (studied at 55°C for 30 days). In addition, other polymer and surfactant materials known in the art may also be suitably •employed in the inventive treatment compositions provided that they do not negatively impact the performance of the coatings.
Further the composition passes at least 5 cycles of freeze thaw stability (-15 deg C to 30 deg C).
Fluorosurfactants
The aqueous composition may contain at least one fluorosurfactant. These are available from DuPont and 3M. It is preferred that the fluorosurfactant contain less than 6 carbon atoms and can be either ionic or non-ionic.
Rheological agents
The aqueous composition may contain at least one rheological agent. Useful thickeners include Rheolate 278, Rheolate 244, Rheolate 300 from Elementis Specialties, Acrysol DR-1, Acrysol DR-72, Acrysol DR-73, Acrysol ASE-60, Acrysol TT-615, Acrysol TT-935, Acrysol RM- 5, Acrysol RM-6000, Acrysol RM-5000, Acrysol RM-2020NPR, Acrysol; RM-8W, Acrysol STC- 275, Acrysol RM-825 from Dow, Coapur 6050 form Coatex. Inorganic thickeners such as bentone Lt, Laponite SD, laponite RD can also be used. The thickener may be present in the aqueous composition in a range of from about 0.10 percent by weight to about 3.0 percent by weight, preferably from about 0.20 percent by weight to about 1.0 percent by weight based on the total weight of the composition. The aqueous composition has a viscosity in a range of from about 100 to 1000 grams on the stormer viscometer. A viscosity greater than about 1000 grams may be less desirable in that it would not be easy to handle. Alternatively, a viscosity less than about 100 grams may be less desirable in that it would run down on vertical surfaces providing reduced coat
weight and would tend to show separation in the container on standing before it could be applied.
Suspending agents may optionally be included in the inventive treatment compositions to improve the suspension and/or dispersion properties of the inventive compositions. Polymer type suspending agents include anionic, cationic and nonionic polymers. Examples include, but are not limited to: vinyl polymers such as, cross linked acrylic acid polymers, cellulose derivatives and modified cellulose polymers such as, methyl cellulose, ethyl cellulose, hydroxyethyl cellulose, hydroxypropyl methyl cellulose, nitro cellulose, sodium cellulose sulfate, sodium carboxymethyl cellulose, crystalline cellulose, cellulose powder, polyvinylpyrrolidone, polyvinyl alcohol, guar gum, hydroxypropyl guar gum, xanthan gum, arabia gum, tragacanth, galactan, carob gum, karaya gum, carrageen, pectin, agar, starch (rice, corn, potato, wheat), microbiological polymers such as, dextran, succinoglucan, pulleran, starch-based polymers such as, carboxymethyl starch, methylhydroxypropyl starch, alginic acid-based polymers such as, sodium alginate, alginic acid propylene glycol esters, acrylate polymers such as, sodium polyacrylate, polyethylacrylate, polyacrylamide, and polyethyleneimine.
Defoamer
The aqueous composition may contain at least one defoamer. Useful defoamers include mineral oil, silicone defoamers, etc. Examples of preferred defoamers include BYK -012, BYK-020, BYK-022, Dapro-4164, Dapro 7005, Dapro-7010. The defoamer may be present in the composition in a range of from about 0.05 percent by weight to about 1.0 percent by weight, preferably from about 0.1 percent by weight to about 0.50 percent by weight based on the total weight of the composition.
Freeze thaw stabilizer
The aqueous composition may contain at least one freeze/thaw stabilizer. Useful freeze/thaw stabilizers include ethylene glycol, propylene glycol and triethylene glycol. The freeze/thaw stabilizer may be present in the aqueous composition in a range of from about 0.50 percent by weight to about 4.0 percent by weight, preferably from about 1.0 percent by weight to about 3.0 percent by weight based on the total weight of the composition. Freeze/thaw stabilizer content greater than about 4.0 percent by weight may be less desirable in that it would make the composition too water sensitive and the drying time too long and increase the volatile organic content of the aqueous composition.
Colorants
Organic or inorganic colorants can be used. Some examples of colorants suitable for use include naphthol, pyrazalones, quinacridones, phthalocyanines, pigments including titanium nickel yellow, limonites, haematites, magnetites, micaceous oxides of iron, iron ferrites and Prussian blue. Further effect pigments may also be used to create special effect finishes with wettability switching properties e.g. Sumica Pearl (Bright gold) available from Sudarshan Chemical Industries Ltd.
Coating thickness
Although there is no limitation in thickness of the coating layer having self-cleaning property formed on the substrate surface, the coating can have a dry film thickness of 1 to 200 microns and preferably from 40 to 120 microns. If the thickness is less than 1 micron, the film hardness is poor. If the thickness is greater than 200 microns, the coating layer has poor flexibility, resulting in generation of cracking in the coating film. Method of making formulation
The aqueous composition can be formed using conventional blending and mixing techniques. Preferably, water is added first, then the t ickeners, surfactants, dispersants, and defoamer, followed by the particles, silicones, then the polymer and other additives using mechanical agitation.
Application
In the method of the present invention, the water-based composition can be either spray, brushed, or paint roller applied on the construction site to the construction surfaces, or supplied as an article to the construction site by pre-application to construction panels, such as oriented strand board, plywood, gypsum based sheeting, and other sheeting materials. It can also be applied by dip coating and spin coating.
If desired, the wet-coated substrate formed in the method of the present invention may be dried using conventional techniques including ambient environmental drying, forced air drying, and forced air/ thermal drying. Preferably, the composition is dried under ambient conditions.
The coating compositions may be processed both at room temperature and at temperatures above or below room temperature, for example, at temperatures in the range from 0 °C to 55 °C.
Substrates
When applied to a surface, the coating has good adhesion to masonry surfaces, painted and many other surfaces such as concrete, cementitious, plaster, cellulosic, wood, one or more polymer, dry or wet surfaces, surfaces exposed to weathering on at least one surface of the substrate. The adhesion of the coating to the substrate may be produced by chemical bonding, or else by physical forces.
Contact angle and surface energy
The contact angle can be measured using a goniometer and the CAH can be measured using the dynamic sessile drop method. 20 pL drop of water is taken, 10 μΙ_ of diiodomethane and 15 pL of hexadecane is taken for surface energy measurements by the Owens-Wendt equation.
The contact angle of water on the coated surface made according to the present invention may be difficult to measure with conventional means because the water droplet bounces or runs off the surface when applied. As illustrated in Example 27 the surface energy for one embodiment of the coating is < 10 mN/m. The self-cleaning surfaces of the invention have a roll-off angle of less than 20 degrees, preferably less than 10 degree, even more preferably less than < 5 degrees in one state. The water contact angles are preferably above 100 degrees, more preferably over 120 degrees, even more preferably superhydrophobic i.e. difficult to measure by conventional techniques.
The coating has a surface energy of <40 mN/m, preferably <20 mN/m and most preferably < 10 mN/m and is recoatable favouring application of a second coat to enhance film build- up to attain desired opacity/ hiding/ whiteness.
Durability
A coating formed from the coating composition of the present invention, has good durability when exposed outdoors to extreme weather for a time period of a few months to several years. Stability is determined by observing that the unwettability, by measuring the contact angle of a droplet on the surface, has not diminished over the course time being exposed to the environment in addition to the colour difference after exposure to Atlas and QUV testing after 2000 hours as illustrated in example 41.
Test methods
The following test methods were used to quantify the properties of the inventive water- based compositions.
Accelerated storage stability
To assess the stability of the paint on storage at elevated temperature conditions (55 +- 1 °C) for 30 days. The initial stormer viscosity of the paint was checked at 30 °C and the paint was kept in a 250 ml lacquered tin container in a forced air circulated oven maintained at 55 + 1 °C with an open vent. The container was removed after 30 days and allowed to reach ambient temperature. The contents were mixed well and the stormer viscosity was determined at 30 °C.
Wet Scrubs testing
To assess the wet scrub resistance of the paint under accelerated conditions to determine the resistance of paints to erosion caused by scrubbing in presence of scrub medium using paint washability apparatus (Erichsen or equivalent). A pig bristle brush (DIN specification) [Hog hair brush], (Brush holder, brush, packing mat.) Total weight: 430 +/- 20 gms.
A film of WFT (wet film thickness) 6 mil was applied on a Leneta P-121 ION dull black plastic panels (165 x 432 x 0.25 mm). The panels are air dried in horizontal position for seven days in a dust free open room.
Scrub medium consisting of water, thickener, Liquor ammonia, surfactant, Silica 400 mesh and preservative.
The panel was placed and clamped in the tray' of washability apparatus. The brush was soaked with 1% solution of SLS. The brush was removed and shaken vigorously to remove any excess SLS solution. The brush was mounted in the holder. The scrub medium was stirred with a stainless steel spatula and lOg was spead evenly on the panel.
The brush was placed at one end of the path and attached to the guide cables. The panel was wet with 5 ml of distilled water in the path of the brush.
After each 400 cycles before failure; 10 g of stirred scrub medium and 5 ml of water was added on the panel. The number of cycles was recorded to remove the paint film fully in one continuous line across the 1/2 inch wide of the shim. The panel was washed under tap water and allowed to stand vertically for 2 hrs. The panel was inspected for signs of integrity failure (colour change, gloss change & rate of loss of film).
Anti algal chamber testing
To assess the anti-algal performance of the paints under accelerated conditions over a period of 30 days. Algae inoculum preparation: Two species of algae, Chlorella and Oscillatoria, were cultured separately. Aerated Cement Blocks (ACBs) of similar surface profile were used for a set of paints under study. The ACBs were sanded with emery paper {Ho. 180). Two coats of paint were applied on the ACBs with the help of a brush, on the cut surface barring 1 cm area on the top portion of the side. Also paint was applied on the bottom side of the ACBs. During first coat, 3 ± 0.1 gm was deposited by wet wt. and during 2nd coat, 1.2 ± 0.1 gm was deposited by wet wt. on the top portion. Overnight curing was allowed between two coats and 7 days curing was allowed after 2nd coat. ACBs were prepared in triplicate for every sample. The cured painted panels were leached under running tap water for 3 hours, keeping the panel in inverted position [painted surface down] in a tray. The bottom tank of the Algae Test Chamber was filled with tap water. Mixed algal inoculum was charged and algae culture broth was added. Performance rating was given as 10 for No growth and 0 for complete growth.
Freeze thaw stability
To determine the freeze thaw stability of the water based paint to determine the extent to which water borne paints retain their original properties free of detrimental changes in consistency, resist coagulation or the formation of the lumps and deterioration of dried film properties such as appearance, gloss and film integrity, when subjected to freezing & subsequent thawing. The stormer viscosity of the paint was measured at 30 °C and the paint was kept in a 250 ml lacquered tin container in a deep freezer maintained at - 15 °C (+-2 °C) for 18 hrs and then removed and allowed to come to thermal equilibrium undisturbed at 30 +- 2 °C or room temperature for 6 hrs. This constitutes one cycle. The test was repeated for 5 cycles. Before stirring, the specimen condition in the can was examined for settling, gelation, coagulation, lumpiness. The paint specimens were stirred and the consistency on stormer viscometer at 30 + 1 °C was determined. The paint was applied to a cement concrete block of 6"x6" by brushing. The performance of the test specimen with the control was compared in terms of dried film properties, such as gloss and film integrity. The paint will be considered to have passed this test if no significant difference in performance/ behaviour is observed between control and test samples.
The present invention is illustrated hereunder in greater details in reference to the non- limiting exemplary illustrations and figures.
Example 1: Surface treatment/ Functionalization of Ti02 with Dynasylan F8261 by heat treatment/ sol-gel treatment
In a typical experiment 100 g of Ti02 R902 (0.32 microns average particle size) from DuPont was treated with 10 g of Dynasylan F8261 (tridecaflurooctyl triethoxysilane) from Degussa, Evonik at 80 °C for 30 minutes after which it was subjected to the process of heat treatment (upto 150°C) for 2 hours. Subsequently, soxhlet extraction was carried out with boiling acetone for 3 hrs to measure the permanency of the treatment and to test whether the particles still remain hydrophobic following the soxhlet wash.
The thus treated Ti02 was both hydrophobic as well as oleophobic as it floats on water and a bed of the Ti02 powder spread on a glass panel is able to repel both water and oil. Heating the pigment with Dynasylan F8261 caused greying of Ti02 which was not desired as Ti02 is added to paint and coatings for its whiteness and hence the greying is undesirable.
Therefore, a sol-gel process under ambient conditions was followed for surface treatment and modification of Ti02 with Dynasylan F8261 to prevent the undesired greying of Ti02 using isopropanol as the solvent and liquor ammonia as the catalyst. The resulting Ti02 was white in colour by visual observation and also showed both hydrophobicity and oleophobicity.
Example 2: Surface treatment/ Functionalization of Ti02 with VTMO by heat treatment/ sol- gel treatment
Further in another typical experiment, the functionalization of Ti02 R902 pigment (20 g) was carried out with VTMO (vinyl trimethoxy silane, 10 g) in 20 g of isopropanol and in 1 g of 25% solution of liquor ammonia. However, to avoid greying of Ti02 the functionalization was carried out at room temperature (for 4 hours) using a sol-gel process instead of employing high temperatures as discussed above. The results reveal that the treated Ti02 pigment showed good hydrophobicity (floating on water) which may be used in the cdating composition.
It was found by way of the present invention that even when heat treatment process was followed (upto 150 °C, 2 hours) for functionalization of Ti02 pigment (100 grams) with VTMO (10 grams), no discoloration of the Ti02 pigments was observed when functionalized with VTMO as was observed with the fluorosilane shown in example 1.
Example 3: Surface treatment/ Functionalization of amorphous nanosilica with VTMO and Dynasylan F8261 by sol-gel treatment
Cabosil M5 amorphous nanosilica (200 nm - 300 nm aggregates with a primary particle size of 14 nm in diameter) (5g) was treated with VTMO (2.5 g) in isopropanol (1.8 g) and 25% liquor ammonia solution (0.7g) using a sol-gel method at room temperature (30°C) for 5 hours and 30 minutes.
The results revealed that the same process of functionalization with Dynasylan F8261 instead of VTMO resulted in both hydrophobic and oleophobic nanosilica as compared to only the hydrophobic nanosilica obtained by treatment with VTMO by following the same said method.
Example 4: Selection of the treatment process of the pigments present in the aqueous coating composition of the present invention, and selection of the material for functionalization to achieve a balance of hydrophobicity and hydrophilicity.
It was therefore found by way of the present invention that discolouration or greying of Ti02 pigments was observed only when heat treatment (upto 150°C) was applied while functiorializing with Dynasylan F8261, whereas the same Ti02 pigments did not change colour when heat treatment was given while functionalizing with VTMO.
However functionalization with Dynasylan F8261 is preferred over functionalization with VTMO as VTMO imparts only hydrophobicity whereas Dynasylan F8261 imparts both hydrophobicity and oleophobicity.
Table 1 below reveals the surface treatments carried out with monomeric and oligomeric silicones for Titanium dioxide and other fillers. These were carried out by heat treatment/ sol gel method. Further they also represent some of the particles and their sizes that may be used in the present invention.
Table 1
Cabosil M5, 14 nm 2% 3-methacryloxypropyltrimethoxysilane
Also, Fig 3 and 4 clearly shows the image of untreated and treated Ti02 with silicone Siires SY231, wherein it is clearly seen in Fig 4 that a fine 27 nm layer of silicone is bonded to the pigment. All of the above treated pigments and extenders as per Table 1 were found to be either hydrophilic (disperses in water) or hydrophobic (floating on water) or partly floating and partly dispersing in water.
As an example, the Ti02 treated with 10% 3-glycidoxypropyltrimethoxysilane showed a lower degree of hydrophobicity (the treated materials are getting dispersed in water and not floating on it) as compared to treatment with Siires SY231 and may be considered as a hydrophilic treatment of silicone. Other such hydrophilic silanes are those with the amine or isocyanate group.
Further, it was noticed that the treatment of nano particles such as nanosilica even up to higher temperatures 290-300°C was showing partial settling in water (vigorous shaking of a test tube containing water) indicating complete hydrophobization was not taking place. This was also observed for the micron sized particles treated with a lower dosage (2%) of VTMO (vinyltrimethoxy silane) and Siires Sy231. This is advantageous since we want a balance of hydrophobicity and hydrophilicity in the polymer as well as the particles to get a surface that is free of dirt streak marks, dirt lines and water spots. Hence, particles showing a balance of hydrophobicity and hydrophilicity are preferred. Further, treatment of Ti02 with 0.5% of Siires Sy231 did not result in hydrophobic particles.
In the case of nano silica treated with 25% 3-methacryloxypropyltrimethoxysilane, the treated silica was not having hydrophobic character (treated for 5h 30 minutes at upto 80°C. However, when checked after a period of 1 month, the particles showed good hydrophobicity (floating on water). Treatment of nanosilica with 2% 3- methacryloxypropyltrimethoxysilane did not lead to hydrophobicity. However, the Ti02 treated with 2% 3-methacryloxypropyltrimethoxysilane, showed good hydrophobicity immediately after heat treatment. Hence it is possible to achieve particles with varying levels of hydrophobicity and hydrophilicity, a balance being preferred to achieve the desired coating composition.
In the case of formation of any lumps during treatment, a mortar and pestle can be used for crushing the lumps and the particles can be passed through a suitably sized sieve. Also, the extent of hydrophobization varied with the particle being treated, the quantity of treating
agent and the time and temperature of the treatment reaction. Higher temperatures were used for treatment with oiigomeric silicones (siires sy 231) as compared to the monomeric silanes. A plurality of particles of different particle sizes can be used for the coating composition with different hydrophobic and hydrophilic silicone treatments provided they are incorporated with the polymer in certain ratios which are discussed below.
Several rutile and anatase grades of titanium dioxide are available such as R902, R902 plus, R 900, R931 etc that can be treated as per the process discussed above.
Hydrophilic grades of nano silica are available such as Cabosil M5 from Cabot or Aerosil R200 (average particle size of 12 nm) from Evonik that can be either heat treated or treated by sol-gel process as discussed above.
Further though external surface treatment of the particles can be used in order to ensure uniform treatment of the particles, in-situ treatment can also be used with, a combination of hydrophobic and hydrophilic organosilicones/ silanes which are shown in latter examples. Example 5: Preparation of a solvent borne resin
In a typical experiment, a solvent borne resin was prepared using hydrophobic monomers wherein 38 pbw of styrene, 40 pbw of n-BMA (n-butyl Methacrylate), 20 pbw of MPTMS (3- methacryloxypropyl trimethoxy silane were copolymerized in the presence of 2 pbw of Zonyl TM from DuPont (perfluorinated monomer), 66 pbw of o-xylene, 1.2 pbw of tbpb (tertiary butyl perbenzoate) initiator and 4 pbw of triethylorthoformate (moisture scavenger) at 135°C for 6 hours with drop wise monomer addition for 3 hours followed by digestion for 3 hours. Nitrogen gas was passed during the reaction. The resin cast on a glass panel showed good water repellency/beading. However, the Contact angle hysteresis values for water were more than 25 ° i.e. complete roll off of water was not observed.
Example 6: Preparation of a modified solvent borne resin
In another typical experiment for the preparation of a hydrophobic solvent borne resin, the preparation of the resin of example 5 was repeated with tertiary butyl methacrylate as a
substitute for styrene, wherein the said resin was prepared by reacting 20 pbw of tertiary butyl methacrylate, 18 pbw styrene, 40 pbw n-BMA (n-butyl Methacrylate), 20 pbw MPTMS (3-methacryloxypropyl trimethoxy silane) in the presence of 2 pbw Zonyl TM (perfluorinated monomer), 66 pbw o-xylene, 1.2 pbw tbpb (tertiary butyl perbenzoate) initiator and 4 pbw triethylorthoformate.
The resin cast on a glass panel showed similar water repellency/ beading as that of a solvent borne resin of Example 5, wherein the similar repellency to water droplets was also observed on the coating. Another batch with 4 pbw Zonyl TM showed similar results. The Contact angle hysteresis values for water were more than 25° i.e. complete roll off of water was not observed.
Example 7: Preparation of amine functional water borne/ water reducible resin in absence of a fluoro monomer with a high content of amine monomer
In a typical experiment a cationic water borne resin was prepared by reacting methyl methacrylate 60 pbw, butyl acrylate (25 pbw), N,N-dimethylaminopropylmethacrylamide (15 pbw), cellosolve (43 pbw), tert-butyl perbenzoate (1.2 pbw), water (100 pbw), acetic acid (6 pbw). The reaction was carried out at 130 °C for 6 hours with drop wise monomer addition for 3 hours followed by digestion for 3 hours. Subsequently water and acetic acid was added to disperse the resin into water at 50°C resulting into a 40% solids resin with an amine value of 99 mg KOH/g.
Since this resin has an amine backbone, the same is responsive to stimuli of change of temperature as well as humidity.
Example 8: Preparation of cationic water borne/ water reducible resin by incorporation of a fluoro monomer and with high content of amine monomer
Since fluoro monomer imparts hydrophobicity and oleophobicity, and does not undergo hydrolysis and condensation reaction with water like MPTMS (3- methacryloxypropyltrimethoxysilane), in a typical experiment a cationic water borne resin was prepared by reacting tertiary butyl methacrylate 60 pbw, Zonyl TAN (perfluorinated monomer, 25 pbw), DMAPMA (Ν,Ν-dimethyl aminopropyl methacrylamide, 15 pbw), cellosolve (43 pbw), tert-butyl perbenzoate (1.2 pbw), water (100 pbw), glacial acetic acid (6 pbw). The reaction was carried out at 130 °C for 6 hours with drop wise monomer addition for 3 hours followed by digestion for 3 hours. Subsequently water and acetic acid
was added to disperse the resin into water at 50°C resulting into a 40% solids resin with an amine value of 99 mg KOH/g.
Even though a high quantity of fluoro monomer was used along with the tertiary butyl Methacrylate contact angle hysteresis < 25 ° could not be achieved by the resin film alone.
Moreover, since this resin has an amine backbone, the same is responsive to stimuli of change of temperature which is demonstrated in the example 27. In addition it is responsive to the stimulus of humidity.
Example 9: Preparation of acid functional water borne/ water reducible resin in the absence of fluoro monomer but with a high content of acid monomer
In a typical experiment the anionic water reducible resin was prepared by the reaction of MMA (methyl methacrylate, 65 pbw), BA (butyl acrylate, 29.5 pbw), MAA (glacial methacrylic acid, 5.5 pbw), cellosolve (43 pbw), tert-butyl perbenzoate (1.2 pbw), water (100 pbw), triethylamine (4.5 pbw). The reaction was carried out at 130 °C for 6 hours with drop wise monomer addition for 3 hours followed by digestion for 3 hours. Subsequently water and triethylamine was added to disperse the resin into water at 50°C resulting into a 40% solids resin with an acid value of 36 mg KOH/g.
Since this resin is acid functional, the same is responsive to stimuli of change of pH and humidity. Example 10: Preparation of acid functional water borne/ water reducible resin by incorporation of a fluoro monomer with a high content of acid monomer.
In a typical experiment an anionic water borne resin was prepared by reacting tertiary butyl methacrylate 64.5 pbw, Zonyl TAN (perfluorinated monomer, 30 pbw), MAA (glacial methacrylic acid, 5.5 pbw), cellosolve (43 pbw), tert-butyl perbenzoate (1.2 pbw), water (100 pbw), triethylamine (4.5 pbw). The reaction was carried out at 130 °C for 6 hours with drop wise monomer addition for 3 hours followed by digestion for 3 hours. Subsequently water and triethylamine was added to disperse the resin into water at 50°C resulting into a 40% solids resin with an acid value of 36 mg KOH/g.
Even though a high quantity of fluoro monomer was used along with the tertiary butyl methacrylate, contact angle hysteresis < 25° could not be achieved by the resin film alone.
Since this resin is devoid of amine backbone and is acid functional instead, the same is responsive to stimuli of change of pH and humidity.
Example 11: Selection of type of resin in the aqueous coating composition of the present invention The treated hydrophobic and oleophobic Ti02 obtained by the sol-gel process as discussed under Example 1 when mixed with a solvent borne resin of Example 5 and 6 in 50:50 or even in 80:20 ratios, the same resulted in a coating that did not exhibit the desired superhydrophobicity (Contact angle hysteresis < 25°). This may be due to the excellent pigment wetting and binding characteristics of solvent borne resins of Examples 5 and 6 which may be responsible for non-migration of the treated Ti02 particles to the surface. There is a continuous layer of polymer film at the surface with insufficient protrusions which is responsible for Contact angle hysteresis >25°. Thus the surface structure is not conducive towards obtaining superhydrophobicity. Moreover, the solvent borne polymers are less preferred over water borne coatings especially in decorative applications as environmental regulations are driving the use of low VOC (volatile organic content) coatings which contain minimum level of solvents. Further, water can be used to clean the brushes used by the painters for application thereby being relatively easier to apply in addition to being environmentally friendly.
Example 12: Evaluation of incorporation of only one kind of functionalized particle in making paint/ coating composition with water borne resin
In an experiment, either the treated Ti02 of Example 1 or Example 2 or Ti02 treated with SY231 under Table 1 were used in high doses (50:50 or 80:20, particles : binder) in combination with either the resin of Example 8 or the resin of example 10 to prepare the paint/ coating composition.
In yet another experiment, treated silica of Example 3 was used in high doses (50:50 particles: binder) in combination with either the resin of Example 8 or the resin of example 10 to prepare the paint/ coating composition.
It was found by way of the present invention that paint/ coating composition obtained from the above experiments though showed thermoresponsive and pH responsive character none of the paint/ coating compositions obtained of the above experiments imparted complete water rolling property, even when the above particles are specifically present in combination with the resins of examples 8 and 10.
Example 13: Evaluation of one. and two kinds of functionalized particles in making paint/ coating composition with water borne resin
Blends of treated Ti02 (micron sized treated with silres sy231) and treated Cabosil M5 fumed silica (nano sized treated with VTMQ) have been evaluated with different resins of experiments 7, 8, 9 and 10. Slurry of treated ΤΊ02 in isopropanol and the slurry of sol-gel treated fumed silica in isopropanol is added individually and a mixture is made with resins 7, 8, 9 and 10.
Treated Ti02 slurry = 70% R902 in isopropanol
Fumed silica slurry = 15% in isopropanol
Resin =10 grams (at 40% solids)
ΤΊ02 slurry = 5.4 grams .
Silica slurry = 5.6 grams
Ratio of Micron to nano sized particles = 4.5
Ratio of particles to polymer = 1.155
Table 2
The coatings were made by simple mixing, diluted with water and applied by brush on fiber cement board panels coated with Asian Paints water based exterior primer (Asian paints exterior primer is based on a modified acrylic emulsion polymer with titanium dioxide along with fillers like calcium carbonate, clays, silicates with a PVC in the range 50 to 80%. It contains additives such as rheology modifiers, pigment dispersing and wetting agents, film and in can stabilizers.), and allowed to dry under ambient conditions for 7 days. It was observed that a combination of treated Ti02 and treated silica when mixed in water reducible resins 8 and 10 gave contact angle hysteresis <10°. Charcoal powder when deposited on the coating gets cleaned with a stream of water giving the benefit of superhydrophobic cleaning. The coating subsequently gets completely wet offering the benefits of superhydrophilic cleaning. The balance of hydrophilicity and hydrophobicity of the polymer, pigments and extenders with a high content of particles/ binder may be responsible for this behavior. The coating was also oleophobic due to the presence of fluorine based monomer. The pigmented coating of resin 8 when placed under a continuous stream of water shows complete wettability. However, as the coating was dried overnight at ambient conditions it regained its superhydrophobicity (CAH < 10°). These low values of Contact angle hysteresis (<10°) were not observed for resins 7 and 9 indicating the importance of the presence of hydrophobic monomers such as tertiary butyl Methacrylate and fluorine based monomer.
Use of only a high quantity of treated Ti02 in a very high quantity leads to chalking of the paint film with resin 8 and 10, while the use of a very high quantity of only treated fumed silica leads to cracking of the paint film due to its high oil absorption value. Example 14: Further evaluation of two kinds of commercially available functionalized particles in making paint/ coating composition with water borne resin
The following slurries were prepared, 54.54% of Hoffmann PF 777 clay in isopropanol and 14.28% of Aerosil R972 in isopropanol.
In an experiment, 6.7 grams of Hoffmann PF777 clay slurry, 7 grams of Aerosil R972 slurry and the resin of example 8 (12.8 g) were used to prepare an aqueous paint composition. Ratio of Micron to nano sized particles = 3.654
Ratio of particles to polymer = 0.9089
The coating showed a CAH<10° and also a stimuli responsive behaviour.
Example 15: Further evaluation of two kinds of functionalized particles in making paint/ coating composition with water borne resin .
The following slurries were prepared, 60% of treated R902 Ti02 in isopropanol and 15% of Aerosil R972 in isopropanol.
In an experiment, 7 grams of Ti02 slurry, 7 grams of Aerosil R972 slurry and the resin of example 8 (12 g) were used to prepare an aqueous paint composition.
Ratio of Micron to nano sized particles = 4
Ratio of particles to polymer = 1.094
The coating showed a CAH<10° and also a stimuli responsive behaviour.
Example 16: Preparation of amine functional water borne/ water reducible resin using styrene and 2-ethyl hexyl acrylate in place of MMA and BA in absence of a fluoro monomer with a high content of amine monomer
In a typical experiment a water borne resin was prepared by reacting styrene 60 pbw, 2- ethyl hexyl acrylate (25 pbw), N,N-dimethylaminopropylmethacrylamide, (15 pbw), cellpsolve (43 pbw), tert- butyl perbenzoate (1.2 pbw), water (100 pbw), acetic acid (6 pbw). The reaction was carried out at 130 °C for 6 hours with drop wise monomer addition for 3 hours followed by digestion for 3 hours. Subsequently water and acetic acid was added
to disperse the resin into water at 50°C resulting into a 40% solids resin with an amine value of 99 mg KOH/g and a pH of 6.
The following slurries were prepared, 60% of treated R902 Ti02 in isopropanol and 15% of Aerosil R972 in isopropanol.
In an experiment, 7 grams of Ti02 slurry, 7 grams of Aerosil R972 slurry and the resin of example 8 (12 g) were used to prepare an aqueous paint composition.
Ratio of Micron to nano sized particles = 4
Ratio of particles to polymer = 1.094
The coating did not show rolling of water and showed a CAH>20°
Example 17: Preparation of amine functional water borne/ water reducible resin using isobutyl Methacrylate, t-butyl Methacrylate and fluoro monomer with a high content of amine monomer
In a typical experiment a water borne resin was prepared by reacting t-butyl Methacrylate (30 pbw), isobutyl Methacrylate ( 30 pbw), N,N-dimethylaminopropylmethacrylamide, (15 pbw), zonyl TAN (25 pbw), cellosolve (43 pbw), tert-butyl perbenzoate (1.2 pbw), water (100 pbw), acetic acid (6 pbw). The reaction was carried out at 130 °C for 6 hours with drop wise monomer addition for 3 hours followed by digestion for 3 hours. Subsequently water and acetic acid was added to disperse the resin into water at 50°C resulting into a 40% solids resin with an amine value of 99 mg KOH/g and a pH of 6.
The following slurries were prepared, 60% of treated R902 Ti02 in isopropanol and 15% of Aerosil R972 in isopropanol.
In an experiment, 7 grams of Ti02 slurry, 7 grams of Aerosil R972 slurry and the resin of example 8 (12 g) were used to prepare an aqueous paint composition.
Ratio of Micron to nano sized particles = 4
Ratio of particles to polymer = 1.094
The coating showed a CAH<10° and also a stimuli responsive behaviour indicating the importance of selective monomers (in this example isobutyl Methacrylate, t-butyl methacrylate and perfluorinated monomer) in the composition.
Example 18: Preparation of acid functional water borne/ water reducible resin in the presence of fluoro monomer but with a high content of acid monomer
In a typical experiment the anionic water reducible resin was prepared by the reaction of tert-butyl acrylate (67.4 pbw), zonyl TAN (25 pbw), MAA (glacial methacrylic acid, 7.6 pbw), cellosolve (43 pbw), tert-butyl perbenzoate (1.2 pbw), water (98.4 pbw), triethylamine (7.6 pbw). The reaction was carried out at 130 °C for 6 hours with drop wise monomer addition for 3 hours followed by digestion for 3 hours. Subsequently water and triethylamine was added to disperse the resin into water at 50°C resulting into a 40% solids resin with an acid value of 50 mg KOH/g.
Example 19: Preparation of acid functional water borne/ water reducible resin in the absence of fluoro monomer but with a high content of acid monomer
In a typical experiment the anionic water reducible resin was prepared by the reaction of tert-butyl acrylate (58.5 pbw), 2-ethyl hexyl acrylate (35.5 pbw), MAA (glacial methacrylic acid, 6 pbw), cellosolve (43 pbw), tert-butyl perbenzoate (1.2 pbw), water (98.95 pbw), triethylamine (7.05 pbw). The reaction was carried out at 130 °C for 6 hours with drop wise monomer addition for 3 hours followed by digestion for 3 hours. Subsequently water and triethylamine was added to disperse the resin into water at 50°C resulting into a 40% solids resin with an acid value of 40 mg KOH/g. Example 20: Preparation of acid functional solvent borne (unneutralized) resin in the presence of fluoro monomer but with a high content of acid monomer
In a typical experiment solvent borne (unneutralized) resin was prepared by the reaction of tert-butyl acrylate (53.3 pbw), 2-ethyl hexyl acrylate (17 pbw),- MAA (glacial methacrylic acid, 4.7 pbw), Zonyl TM (25 pbw), butyl cellosolve (43 pbw), tert-butyl perbenzoate (0.9 pbw). The reaction was carried out at 130 °C for 6 hours with drop wise monomer addition for 3 hours followed by digestion for 3 hours. The resin was discharged without neutralization at 70% solids in cellosolve for subsequent use for the preparation of a stimuli responsive coating with reversible wettability switching and CAH < 25°.
Example 21: Further evaluation of two kinds of functionalized particles in making paint/ coating composition with water borne resin
A. In an experiment, treated Ti02 of Example 1 (4 g), treated amorphous silica of Example 3 (1 g) and the resin of example 8 (lOg) were used to prepare an aqueous paint composition.
B. In yet another experiment, treated Ti02 of Example 2 (4 g), treated Silica of Example 3 (1 g) and the resin of example 8 (lOg) were used to prepare an aqueous paint composition. C. In yet another experiment, treated Ti02 of Example 2 (4 g), treated Silica of Example 3 (1 g) and the resin of example 10 (lOg) were used to prepare an aqueous paint composition.
It was found by way of the present invention that in case of all the above experiments under A and B above a thermoresponsive aqueous coating/ paint composition was achieved due to the amine backbone in the resin exhibiting superhydrophobicity and superhydrophilicity with a low contact angle hysteresis of < 10 degrees having self cleaning behaviour as it helps the dirt get cleaned from its surface with the flow of rain water on the paint surface without leaving behind water spots, dirt lines or streaks only when the resin having a specific backbone of acrylic, fluorinated and amine/ acid is present in combination with both kinds of surface treated particles with a balance of hydrophobicity and hydrophilicity. However, in case of experiment C above the only difference was that . a pH and humidity responsive aqueous coating/ paint composition was achieved with other characteristics of the coating composition remaining the same. The coating can be brush applied on a cement/ concrete panel. The coating is recoatable and 2 coats can be applied on a waterborne primer. Alternately a self priming system can be used. Charcoal powder when deposited on the coating is cleaned with a stream of water giving the benefit of superhydrophobic cleaning. The coating subsequently gets completely wet offering the benefits of superhydrophilic cleaning. The balance of hydrophilicity and hydrophobicity of the polymer, pigments and extenders with a high ratio of particles/binder may be responsible for this behaviour.
Example 22: Evaluation of stimuli responsive resin having the specific acrylic and amine/ acid backbone in combination with both functionalized Ti02 and nanosilica based pigments to achieve the desired contact angle hysteresis vis-a-vis the resin beyond the scope of the present invention
D. In an experiment the resin of Example 9 (lOg) devoid of the fluoro and tertiary butyl Methacrylate backbone in combination with the treated Ti02 of Example 1 (4 g), and silica of Example 3 (1 g) were used to prepare an aqueous paint composition.
E. In yet another experiment the resin of Example 9 (lOg) devoid of the fluoro and tertiary butyl methacrylate backbone in combination with the treated Ti02 of Example 2 (4 g), treated Silica of Example 3 (1 g) were used to prepare an aqueous paint composition.
It was thus found by way of the present invention that the aqueous coating composition under 22 D and E above failed to exhibit the desired superhydrophobicity (low contact angle hysteresis < 10 °) as compared to the aqueous coating compositions under Examples 21 A, B and C above, and hence it was thus found by way of the present invention that the backbone of the resin plays an equally important role to impart the desired superhydrophobicity and superhydrophilicity having self cleaning behaviour and low contact angle hysteresis of < 10 degrees when present in combination with two types of functionalized pigments as discussed above. Thus it can be seen that superhydrophobic coatings can be prepared using externally treated combination of micron and nano sized particles in combination with a polymer; in this example an acrylic polymer with suitably selected monomers.
Example 23: Evaluation of the ratio of functionalized particles of two different sizes present in combination with resin involving the selective acrylic fluorinated amine/acid backbone to attain very low contact angle hysteresis of < 10 degrees
This study reveals the importance of functionalized particles with a balance of hydrophobicity and hydrophilicity present in the resm of the aqueous coating/ paint composition of the present invention under a certain binder to particle (pigment/ filler) ratio. Single coat of the composition was brush applied over Asian Paints exterior primer applied on a fiber cement board panel (6 inch x 3 inch). The coating was dried under ambient conditions for 7 days.
Table 3
Single waterborne resin 40% solids in water and
Resin 8 10 cellosolve
treated Ti02 4
treated silica 1
ttl particles
ratio of micron to nano particles
ratio of total particles to resin
Contact angle hysteresis attained
Blend of resins
A
actual solids
5.12 (40% in water and
Resin blend of 18 and 19 cellosolve)
ΤΊ02 slurry in cellosolve 4.69
silica slurry in cellsolve 0.994
ratio of micron to nano particles 4.718309859
ttl particles 5.684
ratio of total particles to resin 1.11015625
<10°
Contact angle hysteresis attained
B
PBW actual solids
Resin blend of 18 and 19 12.8 5.12
tio2 slurry 7 4.9 (70% solids in cellosolve) silica slurry 7.2 1.0224
ratio of micron to nano particles 4.792644757
ttl particles 5.9224
ratio of total particles to resin 1.15671875
16
<10c
Contact angle hysteresis attained
actual solids
Resin blend of 18 and 19 5.12
tio2 slurry 5.18
silica slurry 1.0792
ratio of micron to nano particles 4.799851742 ttl particles 6.2592 ratio of total particles to resin 1.2225
<10°
Contact angle hysteresis attained
The ratios are as follows: micronized to nano sized particles = 3.5 to 8 (higher ratios being preferred for 2 coat application).
Ratio of particulate mixture (micron sized and nano sized particles) to polymer = 0.9 to 2.5 (higher ratios being preferred for 2 coat application)"
The CAH of <25°, preferably<20°, more preferably<10° and more preferably <5° is desired in one state and higher than 25° in the other switched state. The selective combination of hydrophobic and hydrophilic silicones with suitable acrylic, fiuorinated and hydrophilic backbone gives the desired durability and water resistance to the coating composition along with reversible hydrophilic-hydrophobic switching under variable environmental conditions including variations in at least one or more of temperature, pH, humidity or other external stimuli including light . Example 24: Preparation of acid functional water borne/ water reducible resin using tertiary butyl Methacrylate and fluoro monomer with a high content of acid monomer by dispersion polymerization
In a typical experiment the anionic water reducible resin was prepared by the reaction of t- BMA (t-butyl methacrylate, 42.4 pbw), Zonyl TM (50 pbw), MAA (glacial methacrylic acid, 7.6 pbw), butyl cellosolve (43 pbw), t-butyl perbenzoate (2.2 pbw), water (98.4 pbw), triethylamine (7.6 pbw). The reaction was carried out at 140 °C for 6 hours with drop wise monomer addition for' 3 hours followed by digestion for 3 hours. Subsequently water and
triethylamine was added to disperse the resin into water at 50°C resulting into a 40% solids resin with an acid value of 50 mg KOH/g and pH=8.5.
Since this resin is acid functional, the same is responsive to stimuli of change of pH. Example 25: Details of acrylic latex prepared using emulsion polymerization
Standard acrylic emulsion was used as co-binder in the present invention. It is a pure anionic acrylic emulsion with a solid content of 45% NVM (Non volatile matter), pH = 9 with a particle size of 0.05 to 0.25 pm. The emulsion film has a water contact angle of 74°, hexadecane contact angle of 2.9° and a surface energy of 36.9 ml\l/m.
The blend of example 24 with the emulsion of example 25 shows the following surface energies. Thus a low surface energy was obtained even for a blend of the 2 polymers (emulsion and dispersion).
Table 4
Example 26: Evaluation of the ratio of dispersion to emulsion polymer involving the selective acrylic fluorinated amine/acid backbone in combination with the specific ratios of ^micronized to nano sized particles to attain desired low contact angle hysteresis of <10 degrees
Single and two coats of the blend of dispersion and emulsion polymer with the particle mixture composition was brush applied over Asian Paints exterior primer that is based on a modified acrylic emulsion polymer with titanium dioxide along with fillers like calcium carbonate, clays, silicates with a PVC in the range 50 to 80%. It contains additives such as
rheology modifiers, pigment dispersing and wetting agents, film and in can stabilizers, applied on a fiber cement board panel (6 inch x 3 inch). The coating was dried under ambient conditions for 7 days.
Table 5: Study on the ratio of emulsion to dispersion
Here a blend of dispersion polymer and
emulsion polymer has been used in 40/60 ratio
PBW actual solids
dispersion 24 5.12 2.048 40% in water and cellosolve
emulsion 25 7.68 3.456 45% in water
tio2 slurry 10 7
silica slurry 10 1.42
ratio of micron to 4.929577465
nano particles
ttl particles 8.42 ttl resin 5.504
ratio of total 1.529796512
particles to resin
Contact angle <10° in only
hysteresis first coat
attained
The above 2 ratios was unable to exhibit low
contact angle hysteresis in 2 coats
A; Different ratio was studied
PBW actual solids
dispersion 24
5.12 2.048 40% in water and cellosolve emulsion 25
7.68 3.456 45% in water
tio2 slurry
10 7
silica slurry 8 1.136
ratio of micron to
nano particles 6.161971831
ttl particles 8.136
ttl resin 5.504
ratio of total
partilces to resin 1.478197674
Contact angle <10°in only first
hysteresis coat
attained
Above 2 ratios remained
unable again to exhibit low
contact angle hysteresis in 2
coats.
B; Different ratio was studied
PBW actual solids
dispersion 24 5.12 2.048 40% in water and cellosolve emulsion 25 7.68 3.456 45% in water
tio2 slurry 10 7
silica slurry 12 1.704
ratio of micron to
nano particles 4.107981221
ttl particles 8;704
ttl resin 5.504
ratio of total
particles to resin 1.581395349
Contact angle <10°in only first
hysteresis coat
attained
C, Different ratio
was studied
PBW actual solids
dispersion 24 5.12 2.048 40% in water and cellosolve
emulsion 25 7.68 3.456 45% in water
tio2 slurry 10 7 silica slurry 15 2.13
ratio of micron to 3.286384977
nano particles
ttl particles 9.13 ttl resin 5.504
ratio of total 1.658793605
partilces to resin
Contact angle <10°in only first
hysteresis coat
attained
Above ratios again remained
unable to exhibit low contact
angle hysteresis in 2 coats.
D; different ratio tried
PBW actual solids
dispersion 24 5.12 2.048 40% in water and cellosolve emulsion 25 7.68 3.456 45% in water
tio2 slurry 8 5.6
silica slurry 10 1.42
ratio of micron to
nano particles 3.943661972
ttl particles 7.02
ttl resin 5.504
ratio of total
particles to resin 1.275436047
Contact angle <10°in only first
hysteresis coat
attained
Above ratio also remained
unable to exhibit low contact
angle hysteresis in 2 coats.
PBW actual solids
dispersion 24 5.12 2.048 40% in water and cellosolve emulsion 25 7.68 3.456 45% in water
tio2 slurry 12 8.4
silica slurry 10 1.42
ratio of micron to
nano particles 5.915492958
ttl particles 9.82
ttl resin 5.504
ratio of total
partilces to resin 1.784156977
Contact angle < 10°in first and
hysteresis second coat
attained
Above ratio was preferred to
impart low contact angle
hysteresis for 2 coat
application
F PBW actual solids
dispersion 24 5 12 2.048 40% in water and cellosolve emulsion 25 η gg 3.456 45% in water
tio2 slurry ^ 10.5
silica slurry I.42
ratio of micron to
nano particles 7.394366197 ttl particles I I.92 ttl resin 13.956
ratio of total
partilces to resin 2.165698
Contact angle <10°in first and
hysteresis second coat
attained
The emulsion is described in Example 25 above and the dispersion polymer is prepared in accordance to the present invention as per example 24 above. Dispersion polymerization is known to a person skilled in the art wherein the polymer with acid or amine functionality is prepared in water compatible cosolvent such as cellosolve, carbitol or any other water compatible co solvent or other suitable solvent. After preparation of the polymer, amine such as liquor ammonia etc. or acid such as acetic acid etc. is added to neutralize the polymer (anionic or cationic) and water is added to disperse the polymer into water at a high speed (500 rpm using a stirrer). The dispersion polymers are typically free from surfactants. Although polymerizable or free surfactants maybe added for improving stability, ease of processing, adjusting viscosity, etc. The remaining cosolvent can be stripped out under vacuum resulting in a low VOC resin or alternately VOC exempt solvents can be used for the preparation leading to a reduction in the cycle time involved with stripping of the solvent. Such examples of low VOC solvents are Ecosoft PB, dimethyl carbonate etc.
The low contact angle hysteresis <10° was obtained above for 1 coat application in accordance with the invention, however for 2 coat applications higher ratios of total particles to binder and micron to. nanometer particles were required to achieve contact angle hysteresis < 10 degrees since the surface profile of the topcoat may change depending on the surface profile of the substrate/ undercoat.
Experiment E under Table 5 above provided for the preferred low contact angle hysteresis of <10 degrees wherein the ratio of emulsion (25) to dispersion polymer (24) was in 60/40 ratio. However, several ratios of emulsion to dispersion polymers of 70/30, 80/20 and 90/10 revealed that 70/30 and 80/20 showed the desired low contact angle hysteresis of <10 degrees, whereas 90/10 showed high contact angle hysteresis of >10 degrees, and hence a sufficient high quantity of dispersion polymer is preferred to be present in the paint/coating composition of the present invention.
However, presence of emulsion polymer having surfactants may still give low hysteresis provided a dispersion polymer is present in the system. Ratio of dispersion polymer to emulsion polymer (5/95 to 100% dispersion polymer is preferred). Emulsion polymers are not preferred in the composition of the present invention as the film formation mechanism coupled with the presence of surfactants increases the contact angle hysteresis values possibly due to a non-continuous film covering the pigment and extender particles.
Only emulsion polymers may be used, however, the CAH values will be higher, more than 20 degrees for emulsions of example 25 and 44 (shown in example 55 ) and further the time of switching from hydrophobicity to hydrophilicity will be decreased to under few minutes. Also if the emulsion has a high contents of external surfactants, water marks may appear on the paint. Polymerizable surfactants may be used to prepare emulsions (shown in example 44), however the CAH value will be higher for a given composition when compared with dispersion. Thus dispersion polymers are preferred.
Other components to prepare a coating formulation can be present in the paint formulation and low hysteresis can be obtained. Other components include but not limited to thickeners, biocides, dispersing agents, defoamers, buffers etc. These components are known to those skilled in the art. This is shown in example number 34.
It is thus apparent form the tables above that apart from the surface chemistry, the surface morphology of the particles comprising of micro- to nano structures, or even micro structures alone, the ratios between said micro to nano structures as well as the ratios between the particulate mixtures to polymer all contribute to lowering the contact angle hysteresis of the composition to < 10 degrees. These ratios are valid where a combination of micron sized and nano sized particles are used. It is also possible to achieve low hysteresis values using only micron sized particles of a variety of sizes (preferably less than 325 mesh) or irregularly shaped particles as shown in latter examples. It is also possible to achieve low hysteresis using only nano particles; however the formulation will be comparatively more expensive. Moreover, the ratio of dispersion to emulsion polymers in the aqueous coating composition of the present invention also plays a role in determining the contact angle hysteresis. The adhesion of dirt particles to these surfaces is minimal because they touch only the tips of the structure leading to the dirt particles being only lightly deposited on the coating surface. The particles are easily washed away by water droplets, which roll off.
Example 27: Characterization and study of the water-borne acrylic random copolymer dispersions in combination with the functionaiized pigments in accordance with the present invention
A series of water-borne acrylic random copolymer dispersions comprising specialty monomers; tertiary butyl Methacrylate (TBMA) for hydrophobicity and perfluorinated monomer (Zonyl TAN/ Zonyl TM) for oleophobicity and improved dirt pickup resistance of coating were synthesized by conventional free radical polymerization as per examples 8 and 10.
They can be acid or amine functional. These copolymers were processed with several functionaiized pigments to give stimuli responsive (pH/ temperature/ humidity) water-borne coatings as shown above.
One of these coatings (cationic) was taken up for the study that was prepared by dispersing the treated pigments in the amino functional acrylic dispersions to get weight solids of about 40 % in water. The resulting coating composition was free flowing liquid that could be applied as such or with little dilution using the conventional brush, roller or airless spray application techniques.
Here for the purpose of characterization and to minimize the errors in dry film thickness (DFT), coatings were applied on glass panels of dimensions 6 x 4 square inches using a 100-μιη SHEEN draw-down applicator to form uniform films of about 50-jjm DFT. The coated panels were matured for 7 days and tested for contact angle measurements using a Digidrop Contact Angle Meter of GBX Surface Science Technologies make. The contact angle values were measured using the static sessile drop method after a time period of 60 seconds. An average of 5 readings taken at different parts of the coating is reported.
The contact angle measurements were first carried out at ambient temperature of about 30°C. It was an interesting observation to note that the coating surface behaved superhydrophobic with contact angle hysteresis ΘΑ - 9R <10 degrees. When a 5 μΙ_ water droplet is pumped through a syringe needle, it touches the surface and becomes non- coaxial with the needle as soon as it begins to advance. The droplet then moves to the right and around to the back of the needle. As the water droplet is removed with the syringe, it remains stationary until it spontaneously dewets the surface and rises into the needle.
However, to quantify the contact angle of our pigmented coating in this study, the syringe was further pumped to elute water droplet at increasing intervals of 5 pL as per Figure 5. It was thus observed that water droplet is released from the syringes only after it reaches a volume of 45 μΙ_. The contact angle of this droplet was found to be 143°.
The study of contact angle was also carried out using hexadecane as a probe. The minimum volume eluting from the syringe needle to form a droplet was 15 pL in the case of hexadecane due to its low surface tension. The contact angle value for hexadecane was found to be 98° at ambient temperature. The value clearly indicates that the coating is not just hydrophobic but also oleophobic in nature.
Table 6: Surface energy of pigmented coating
Table 6 above shows the surface energies of the coating. The surface energy was calculated by the Owens-Wendt equation utilizing the contact angles of distilled water and hexadecane using Windrop++ software. The surface energy obtained for the coating is 5.3 mN/m.
The switching behaviour from superhydrophobicity to superhydrophilicity is demonstrated from the following experiments. The coated glass panel was placed in a deep freezer at 0°C for 5 hours and then immediately subjected to contact angle measurement. It was possible to measure the contact angle with just a 5 pL water droplet with the angle reducing down to <90°. When the panel removed from the freezer is placed under a continuous flow of water, the pigmented coating exhibits superhydrophilicity as a thin sheet of water covers the surface. This complete wetting of the surface with water enables cleaning of the surface thereby preventing dirt streak marks, which is associated with non-responsive superhydrophobic coatings. When the same panel was allowed to attain ambient temperature, it regained superhydrophobicity indicating the thermoresponsive wettability
switching of the coating. The experimental values were found to be reproducible for more than five times explaining that the phenomenon of switching from superhydrophobicity to superhydrophilicity is reversible. The rheological behavior of the liquid pigmented system (50% non volatile content) was measured in a cup and bob assembly (CC27/ T200/ AL) using the Anton Paar Physica MCR 301 rheometer. The temperature sweep at shear rates 0.001, 0.01, 0.1 and 1 s"1 was carried out over a range of 0 to 75 °C at 5 °C/min. The switching behavior from superhydrophilicity to superhydrophobicity with the temperature variation was further substantiated by carrying out rheological studies of the liquid pigmented system. As the polymer conformational change is a delicate molecular phenomenon the rheological measurements were performed at low shear rates 0.001, 0.01, 0.1 and 1 s"1. The viscosity values are plotted as a function of temperature in Figure 6. The rheograms show an initial decrease in viscosity with temperature followed by an anomalous increase between 20-50°C. The viscosity passes through a maxima at a low shear rate (< 1 s"1) while at a higher shear rate (1 s"1) there is only a slight increase in viscosity before it again decreases. This indicates that at low shear rate the system shows some kind of interchain associations (probably driven by hydrophobic associations) in the temperature range of 20-50°C. This interchain association results into the observed temperature independent viscosity behaviour because there are two opposing tendencies, which apparently seem to balance each other. The usual Arrhenius-like decrease in viscosity with increase in temperature is balanced by association driven increase in viscosity with increase in temperature.
Thermal changes in the dried pigmented coating were studied using Differential Scanning Calorimetry (DSC). TA instruments Q10 apparatus was used to record DSC thermograms at a heating rate of 2°C/min over a temperature range of -40 °C to 90 °C, under nitrogen atmosphere.
The changes in polymer conformation with temperature for the dried pigmented coating were carried out by DSC studies. Figure 7 depicts an endotherm corresponding to the temperature region (30-55°C) where the coating is switching from hydrophilicity to hydrophobicity.
Both the rheology and thermal analysis data support our observation that the coating becomes hydrophobic with increase in temperature for the pigmented coating system.
A SEM (Scanning electron microscopy) micrograph of stimuli responsive paint as shown in Figure 8 also supports the fact that use of functionalized pigments helps in creating two levels of topography on the paint surfaces. This topography can increase receding contact angles and decrease contact angle hysteresis.
The topography of the paint surface is further substantiated by AFM (Atomic Force Microscopy) studies. The AFM picture as shown in Figure 9 depicts high surface heterogeneity having a root mean square surface roughness of 210 nm. The AFM image of Figure 10 shows the thermoresponsive behaviour of the paint, wherein the images on the left are under hot condition and the images on the right are under cold condition.
It is thus possible by the technically advanced stimuli responsive water based paint/ coating composition of the present invention exhibiting reversible hydrophilic-hydrophobic switching with variations in external stimuli to achieve a low contact hysteresis of < 10 degrees due to the involvement of the specific polymeric dispersion containing acrylic, fluorinated and amine/ acid backbone in combination with a particulate mixture comprising surface treated combination of micron and nano sized particles.
The stimuli responsive coating of the present invention can be thus used to make self- cleaning/ easy cleaning, anti-graffiti and stain resistant coatings wherein the said coatings are made by a simple, facile and economically viable process and can be applied by conventional techniques such as brush, roller and airless spray application.
Example 28: Preparation Of acid functional water borne/ water reducible resin using tertiary butyl Methacrylate and fluoro monomer with a high content of acid monomer and a triacrylate crosslinker.
In a typical experiment the anionic water reducible resin was prepared by the reaction of t- BMA (t-butyl methacrylate, 42.4 pbw), Zonyl TM (perfluorinated monomer, 50 pbw), MAA (glacial methacrylic acid, 7.6 pbw), TMPTA (trimethylolpropane triacrylate, 0.5 pbw), butyl cellosolve (43 pbw), di-tert butyl peroxide (2.2 pbw), water (98.4 pbw), triethylamine (7.6 pbw). The reaction was carried out at 140 °C for 6 hours with drop wise monomer addition for 3 hours followed by digestion for 3 hours. Subsequently water and triethylamine was added to disperse the resin into water at 50°C resulting into a 40% solids resin with an acid value of 50 mg KOH/g and pH=8.
Since this resin is acid functional, the same is responsive to stimuli of change of pH and humidity.
Example 29: Preparation of acid functional water borne/ water reducible resin using tertiary butyl Methacrylate and fluoro monomer with a high content of acid monomer and a triacrylate crosslinker using an alternate initiator
In a typical experiment the anionic water reducible resin was prepared by the reaction of t- BMA (t-butyl methacrylate, 42.4 pbw), Zonyl TM (perfluorinated monomer, 50 pbw), MAA (glacial methacrylic acid, 7.6 pbw), TMPTA (trimethylolpropane triacrylate, 0.5 pbw), butyl cellosolve (43 pbw), t-butyl perbenzoate (2.2 pbw), water (98.4 pbw), triethylamine (7.6 pbw). T e reaction was carried out at 140 °C for 6 hours with drop wise monomer addition for 3 hours followed by digestion for 3 hours. Subsequently water and triethylamine was added to disperse the resin into water at 50°C resulting into a 40% solids resin with an acid value of 50 mg KOH/g and pH=8.5.
Since this resin is acid functional, the same is responsive to stimuli of change of pH and humidity.
Example 30: Preparation of acid functional water borne/ water reducible resin using tertiary butyl acrylate and fluoro monomer with a high content of acid monomer
In a typical experiment the anionic water reducible resin was prepared by the reaction of t- BA (t-butyl acrylate, 42.4 pbw), zonyl TM (50 pbw), MAA (glacial methacrylic acid, 7.6 pbw), butyl cellosolve (43 pbw), t-butyl perbenzoate (1 pbw), water (98.4 pbw), triethylamine (7.6 pbw). The reaction was carried out at 130 °C for 6 hours with drop wise monomer addition for 3 hours followed by digestion for 4 hours. Subsequently water and triethylamine was added to disperse the resin into water at 50°C resulting into a 40% solids resin with an acid value of 50 mg KOH/g and pH=8.5.
Since this resin is acid functional, the same is responsive to stimuli of change of pH and humidity.
Example 31: Preparation of acid functional water borne/ water reducible resin using tertiary butyl methacrylate and fluoro monomer with a high content of acid monomer and high initiator content.
In a typical experiment the anionic water reducible resin was prepared by the reaction of t- B A (t-butyl methacrylate, 42.4 pbw), MAA (glacial methacrylic acid, 7.6 pbw), Zonyl TM (50 pbw), butyl cellosolve (43 pbw), ditertiarybutyl peroxide (2.2 pbw), water (98.4 pbw), triethylamine (7.6 pbw). The reaction was carried out at 140 °C for 6 hours with drop wise monomer addition for 3 hours followed by digestion for 3 hours. Subsequently water and triethylamine was added to disperse the resin into water at 50°C resulting into a 40% solids resin with an acid value of 50 mg KOH/g and pH=8.5.
Since this resin is acid functional, the same is responsive to stimuli of change of pH and humidity.
Example 32: Preparation of acid functional water borne/ water reducible resin using tertiary butyl methacrylate and fluoro monomer with a high content of acid monomer, multifunctional acrylate and a silane monomer In a typical experiment the anionic water reducible resin was prepared by the reaction of t- BMA (t-butyl methacrylate, 40.4 pbw), MAA (glacial methacrylic acid, 7.6 pbw), Zonyl TM (50 pbw), 3-methacryloxypropyltrimethoxysilane (0.5 pbw), Trimethylolpropanetriacrylate (0.5 pbw), butyl cellosolve (43 pbw), ditertiarybutyl peroxide (2.2 pbw), water (98.4 pbw), triethylamine (7.6 pbw). The reaction was carried out at 140 °C for 6 hours with drop wise monomer addition for 3 hours followed by digestion for 3 hours. Subsequently water and triethylamine was added to disperse the resin into water at 50°C resulting into a 40% solids resin with an acid value of 50 mg KOH/g and pH=8.5.
Since this resin is acid functional, the same is responsive to stimuli of change of pH and humidity.
Example 33: Preparation of acid functional water borne/ water reducible resin using tertiary butyl methacrylate and fluoro monomer with a high content of acid monomer
In a typical experiment the anionic water reducible resin was prepared by the reaction of t- BA (t-butyl acrylate, 41.9 pbw), MAA (glacial methacrylic acid, 7.6 pbw), Zonyl TAN (50 pbw), Trimethylolpropanetriacrylate (0.5 pbw), butyl cellosolve (43 pbw), ditertiarybutyl peroxide (1 pbw), water (98,4 pbw), triethylamine (7.6 pbw). The reaction was carried out at 140 "C for 6 hours with drop wise monomer addition for 3 hours followed by digestion for 4 hours. Subsequently water and triethylamine was added to disperse the resin into water at 50°C resulting into a 40% solids resin with an acid value of 50 mg KOH/g and pH=8.5.
¾9
Since this resin is acid functional, the same is responsive to stimuli of change of pH and humidity.
Example 34: Sample Paints prepared using the water-borne acrylic random copolymer dispersions in combination with the functionalized particles (pigments and extenders) in accordance with the present invention
Table 7
Using the above mill base, several different paints were prepared. Blends of dispersion and emulsion have been used in this example with a balance of hydrophobicity and
hydrophilicity. Conventional paint ingredients have been used in this formula. These coatings exhibit good water repellency after application as shown in Table 8, 9 and 10.
The water, dispersion polymer, dispersing agent, wetting agent, defoamer, glycol, biocides, dipentene, rheological additive are mixed on a high speed disperser for 15 minutes at 500 rpm, after which the pigments and extenders are added. The grinding stage is carried out at 2000 rpm for 20 minutes after which speed is reduced to 800 rpm and coalescent is added followed by the binder. The examples show the contact angle hysteresis values after 1 coat application on fiber cement boards after 7 days air drying.
Table 8 a) MB1 = 3.2 grams
Resin 24= 3.2 grams
CAH> 25° b) MB1 = 3.2 grams
Resin 24= 1.28 grams
Emulsion 25 = 1.92 grams
CAH> 25° c) MB1 = 3.2 grams
Resin 24= 1.28 grams
Emulsion 25 = 1.92 grams
Aerosil R972 slurry (14.3%) = 1 gram
CAH< 25° d) Resin 24= 3.2 grams
Hoffman Clay slurry (63.63% in butyl cellosolve) = 3.3 grams
Aerosil R972 (14.3%) = 2.5
CAH<20° e) MB1 = 3.2 grams
Resin 24= 3.2 grams
9i
Hoffman Clay slurry (63.63% in butyl cellosolve) Aerosil R972 (14.3% slurry) = 2.5 grams
CAH< 25° f) MBl = 3.2 grams
Resin 30 = 3.2 grams
Hoffman Clay slurry (63.63% in butyl cellosolve)
Aerosil R972 (14.3% slurry) = 2.5 grams
CAH< 25° g) MBl = 3.2 grams
Resin 24 = 1.28 grams
Emulsion 25 = 1.92 grams
Hoffman Clay slurry (63.63% in butyl cellosolve) = 3.3 grams Aerosil R972 slurry (14.3%) = 2.5 grams
CAH< 25° h) MB2 = 3.2 grams
Resin 24= 3.2 grams
Hoffman Clay slurry (63.63% in butyl cellosolve) = 3.3 grams Aerosil R972 (14.3% slurry) = 2.5 grams
CAH< 25° i) MB2 = 3.2 grams
Resin 30 = 3.2 grams
Hoffman Clay slurry (63.63% in butyl cellosolve) = 3.3 grams Aerosil R972 (14.3% slurry) = 2.5 grams
CAH< 25° j) MB2 = 3.2 grams
Resin 24 = 1.28 grams
Emulsion 25 = 1.92 grams
Hoffman Clay slurry (63.63% in butyl cellosolve) = 3.3 grams Aerosil R972 slurry (14.3%) = 2.5 grams
CAH< 25°
91
k) MB2 = 3.2 grams
Resin 30 = 1.28 grams
Emulsion 25 = 1.92 grams
Hoffman Clay slurry (63.63% in butyl cellosolve)
Aerosil R972 slurry (14.3%) = 2.5 grams
CAH< 25°
I) MB2 = 3.2 grams
Resin 32 = 1.28 grams
Emulsion 25 = 1.92 grams
Hoffman Clay slurry (63.63% in butyl cellosolve)
Aerosil R972 slurry (14.3%) = 2.5 grams
CAH< 25°
m) MB2 = 3.2 grams .
Resin 29 = 3.2 grams
Hoffman Clay slurry (63.63% in butyl cellosolve)
Aerosil R972 slurry (14.3%) = 2.5 grams
CAH< 25° n) B2 = 3.2 grams
Resin 29 = 1.28 grams
Emulsion 25 = 1.92 grams
Hoffman Clay slurry (63.63% in butyl cellosolve)
Aerosil R972 slurry (14.3%) = 2.5 grams
CAH< 25° o) MB3 = 3.2 grams
Resin 24 = 1.28 grams
Emulsion 25 = 1.92 grams
Hoffman Clay slurry (63.63% in butyl cellosolve) = 3.3 grams Aerosil R972 slurry (14.3%) = 2.5 grams
CAH< 25°
p) MB3 = 3.2 grams
Resin 30 = 1.28 grams
Emulsion 25 = 1.92 grams
Hoffman Clay slurry (63.63% in butyl cellosolve) = 3.3 grams
Aerosil R972 slurry (14.3%) = 2.5 grams
CAH< 25° q) MB3 = 3.2 grams
Resin 33 = 1.28 grams
Emulsion 25 = 1.92 grams
Hoffman Clay slurry (63.63% in butyl cellosolve) = 3.3 grams
Aerosil R972 slurry (14.3%) = 2.5 grams
CAH< 25°
The examples show the contact angle hysteresis values after 1 coat application in the presence of conventional paint ingredients.
A Sample PVC calculated for example d indicates the coating is 23.13% above the theoretical CPVC (critical Pigment Volume concentration).
Table 9
Resin 34 (60:40)
Hoffman Clay 3.3 3 3.3 3
slurry (63.63% in
butyl cellosolve)
Aerosil R972 2.5 2 2.5 2
slurry (14.3%)
Contact angle < 25° > 25° < 25° > 25°
hysteresis
Tables 9 and 10 above show that it is possible to attain low CAH values provided certain ratios of micron and nano sized particles (commercially available treated clay and treated silica) are present along with conventional paint ingredients and specific polymers.
Example 35: Sample Paints prepared using the water-borne acrylic random copolymer dispersions in combination with the functionalized particles in accordance with the present invention
Table 11
Table 11 shows that it is possible to attain low CAH values provided certain ratios of micron and nano sized particles (commercially available treated Titanium dioxide and treated silica) are present along with conventional paint ingredients and specific polymers. Example 36: Sample Paints prepared using the water-borne acrylic random copolymer dispersions in combination with the functionalized pigments in accordance with the present invention and their exterior exposure data
Blends of dispersion and emulsion have been used in this example with a balance of hydrophobicity and hydrophilicity. Conventional paint ingredients have been used in this formula. These coatings exhibit good water repellency after application.
Table 12
The paints pass the water immersion test over a period of 7 days without blistering. The wet scrub resistance of these coatings was 60 cycles. Accelerated anti-algal chamber testing for 30 days shows the paints 1 and 2 to have a rating of 7.5/10. 2 coats of paint were brush applied over 1 coat of Asian Paints exterior primer.
Table 13: Millbase formula
The water, dispersion polymer, surfactant, defoamer, glycol, biocides, dipentene, rheologicai additive are mixed on a high speed disperser for 15 minutes at 500 rpm, after which the pigments and extenders are added. The grinding stage is carried out at 2000 rpm for 20 minutes after which speed is reduced to 800 rpm and coalescent is added followed by the binder.
Exterior exposure was studied for these paints on a vertical concrete wall over a period of 4 months .during the rainy season. SC 4 was found to exhibit, over all best performance including anti algal performance.
Table 14
Algal growth No No Initiated No
Performance rating 10 10 9 10
Performance rating [0=Poor & 10=Best]
These paints demonstrate the durability of the coating and paint SC4 demonstrates the good hydrophobicity achieved even after 4 months of exterior exposure. The paints had a good DPUR (dirt pickup resistance) and did not show dirt streaks, lines and water spots. In addition, all paints showed good antimicrobial properties. Algal growth was initiated for SC3 where no biocide was present. Example 37: Durability study of paints according to this invention.
Table 15
MMA: methyl methacrylate, n-BMA: n-Butyl methacrylate, MAA: Glacial methacrylic acid, Zonyl TAN and Zonyl TM: Perfluorinated monomer, 2-EHA: 2-ethyl hexyl acrylate
Paints with 65, 70 and 75 PVC were prepared according to this invention and exposed to QUV B, 313 nm for 1100 hours.
Table 16
All paints showed excellent durability after 1100 hours of QUV exposure, with the fluorinated binder showing lower delta E values and hence better durability.
Example 38: Water-borne acrylic random copolymer dispersions prepared with a combination of different monomers
A series of different polymers (A to D) were prepared with different starting monomers. All the polymers have an acid value of 50 mg KOH/g and contain the perfluorinated monomer at a fixed content of 5% on monomer composition (except E which does not contain the fluorine based monomer). The resin E does not contain the perfluorinated monomer and is shown to compare the surface energies of the coatings with and without the perfluorinated monomer.
MMA: methyl methacrylate, n-BMA: n-Butyl methacrylate, MAA: Glacial methacrylic acid, t- BMA: Tertiary butyl methacrylate, MA 2000: Polyethylene glycol methacrylate with molecular weight 2000, Zonyl TM: Perfluorinated monomer, BA: Butyl acrylate, 2-EHA: 2- ethyl hexyl acrylate
The film cast from resin E which does not contain fluorinated monomer shows a surface energy of 31.9 mN/nrv while films of resins A to D which contain 5% fluorinated monomer on resin solids give a surface energy of <25 mN/m and is preferred for exhibiting dirt pickup resistance.
The reaction was carried out at 80 °C for 6 hours with drop wise monomer addition for 3 hours followed by digestion for 3 hours. Subsequently water and liquor ammonia was added to disperse the resin into water at 50°C resulting into a 25-30% solids resin with an acid value of 50 mg KOH/g and pH=8.5. Since this resin is acid functional, the same is responsive to stimuli of change of pH and humidity. The water absorption of the resin films varied between 5 to 20% after 96 hours in water for the different resins.
Example 39: Paints prepared using the water-borne acrylic random copolymer dispersions of example 38 in combination with the functionalized pigments in accordance with the present invention and their exterior exposure data with only hydrophobic organosilicone resin which is hydrophobic.
Nine paints were prepared using the resins 39A, B, C and D. The paint formulation is given in Table below. In these paints micron sized pigments and extenders have been used to prepare the paints. The Ti02 used has an average particle size of 0.32 microns. The water, surfactants, defoamer, glycol, biocides, rheological additive are mixed on a high speed disperser for 15 minutes at 600 rpm, after which the pigments and extenders are added. Insitu treatment of the pigments and extenders was carried out during paint making using a hydrophobic organosilicone in the formulation. The grinding stage is carried out at 2000 rpm for 20 minutes after which speed is reduced to 800 rpm and the binder is added followed by thickener and coalescent.
Table 18 Paint Formula
agent 0.186 0.201 0.199 0.184 0.186 0.175 0.176 0.176 0.189
Cellulosic
thickener 0 0 0 0 0 0 0 0 0.472
Clay thickener 0 0 0 0 0 0 0.439 0.439 0
PU thickener 0.186 0.175 0.199 0.184 0.186 0.175 0 0 0 pH adjustment 0.093 0.087 0.1 0.092 0.0934 0.087 0.088 0.088 0.094
Ti02 Rutile,
0.32 microns 18.679 17.515 19.99 18.41 18.679 17.519 17.55 17.55 18.91
Quartz (particle
size 4 microns) 5.977 5.604 6.396 5.891 0 5.606 5.616 5.616 0
Quartz (particle
size 14 microns) 23.91 22.419 25.587 23.56 29.8874 22.425 22.47 22.47 30.266
Antialgai agent 1.955 1.833 2.092 1.927 1.955 1.833 1.837 1.837 1.979
Acrylic binder 8.321 7.881 8.665 8.726 8.321 8.759 8.776 8.776 7.348
(C) (A) (C) (D) (C) (B) (C) (C) (D)
Hydrophobic
organosilicone 4.669 4.378 4.997 4.602 4.669 4.379 4.388 4.388 4.729
Emulsion of
example 25 0 0 13.213 0 0 0 0 0 0
Acrylic binder 18.679 21.893 0 19.33 18.679 21.023 21.06 19.06 18.916
Rheological
additive 0.467 0.437 0.499 0.46 0.467 0.438 0 0 0
Polyurethane
dispersion 0 0 0 0 0 0 0 2 0
Coalescent 1.401 1.313 1.499 1.381 1.401 1.313 1.316 1.316 1.418
TOTAL 100 100 100 100 100 100 100 100 100
Asian Paints exterior primer followed by 2 coats of these paints at 25% volume dilution was applied on a vertical wall with a horizontal ledge. The wet scrub resistance of these coatings was 500 cycles. As seen after the exposure all the samples except sc6 show good properties in terms of DPUR, whiteness, hydrophobicity and good antimicrobial properties. This demonstrates the importance of other acrylic monomers such as t-butyl methacrylate, n- butyl methacrylate as part of the resin composition, apart from the perfluorinated monomer
and a high acid value (50 mg KOH/g) in this example. However, over a period of time (12 months) exterior exposure, the coatings show dirt streak marks indicating that only the hydrophobic organosilicones are not suitable to resist dirt streak marks. The styrene containing coating (sc6-involving Acrylic binder B illustrated in Table 17) shows inferior results in terms of DPUR, microbial growth and even exhibits chalking. Hence it is preferred to use a styrene free polymer. A study is carried out as shown in example 40 that highlights the importance of a combination of hydrophobic and hydrophilic organosilicones with monomers having a balance of hydrophobicity and hydrophilicity. Example 40: Water-borne acrylic random copolymer dispersions prepared with a combination of different monomers and varying acid values
A series of different polymers were prepared with varying acid values from 25 to 50 mg KOH/g. 6 resins made with different hydrophobic monomers were made. All the resins had a pH of 8 to 9.
Table 19
MMA: methyl methacrylate, n-BMA: n-Butyl methacrylate, MAA: Glacial methacrylic acid, t- BMA: Tertiary butyl methacrylate, Zonyl TM : Perfluorinated monomer, BA: Butyl acrylate, 2- EUA: 2-ethylhexyl acrylate
The above resins with a range of acid values can be used in the present invention in combination with hydrophobic and hydrophilic organosilicones and the particulate mixture to achieve the desired wettability switching. Monomer water solubility of common monomers and octanol water coefficient values indicates that a combination of monomers is preferred with a balance of hydrophobicity and hydrophilicity to prepare polymers suited for the current invention.
Table 20: The values of octanol water coefficients for the monomers are taken from "Journal of Biomedical Materials Research" Volume 15, 787-793 (1981), S. Fujisawa and E. Masuhara (log Kow values for acrylic acid, methacrylic acid and t-butyl acrylate are from literature)
Example 41: Paints prepared using the water-borne acrylic random copolymer dispersions of example 40 in combination with the functionalized pigments in accordance with the present invention and their exterior exposure data
Ten paints were prepared using the resins 40A, B, C, D, E and F to study permanency via showering, Atlas and QUV testing. The paint formulation is given in Table 21 below. The water, surfactants, defoamer, glycol, biocides, rheological additive are mixed on a high speed disperser for 15 minutes at 600 rpm, after which the pigments and extenders are added. Insitu treatment of the pigments and extenders was carried out during paint making using several different combinations of the hydrophobic and hydrophilic silicones. In these examples, only the hydrophobic organosilicone and a combination of a hydrophobic organosilicone at 2 levels and a hydrophilic organosilicone at 3 levels have been studied. The grinding stage is carried out at 2000 rpm for 20 minutes after which speed is reduced to 800 rpm and the binder is added followed by thickener and texanol. All paints showed a scrub resistance of > 500 cycles.
Paints were diluted at 20 to 40% by volume and 2 coats were brush applied over 1 coat of Asian Paints exterior primer after a gap of 4 hours. Table 21
0.55 microns 18 18 18 18 18 18 18 18 18 18
Quartz (14
microns) 28.8 28.8 28.8 28.8 28.8 28.8 28.8 28.8 28.8 28.8
Hydrophilic
organosilicone 0 4 9 4 4 4 0 0 4 9
Hydrophobic
organosilicone 2.7 2.7 2.7 2.7 2.7 2.7 3 2.7 2.7 2.7
Polymer 26.87 26.87 26.87 26.285 24.51 25.26 23.734 18.82 18.82 18.82
Glycol 0.4 0.4 0.4 0.4 0.4 0.4 0.4 0.4 0.4 0.4
Coalescent 1.5 1 1.5 1 1 1 1 1.5 1.5 1.5
Rozone 2000 1 1 1 1 1 1 1 1 1 1
Fluorosurfactant 0.1 0.1 0.1 0.1 0.1 0.1 0.1 0.1 0.1 0.1
Acrysol set 275 0.1 0.2 0.1 0.2 0.2 0.3 0.2 0.2 0.2 0.2
Total 100 100 106.72 100 100 100 100 100 100 100
After 6 months of exterior exposure, the Styrene acrylic based paints showed the maximum algal growth. Hence it is preferred to use a styrene free polymer. All the paints had a water contact angle of paint > 130° and the contact angle hysteresis for all the paints <10 degrees. (C2 is styrene based as is revealed from Table 19 above). Both the contact angle and CAH values were maintained even after QUV exposure of 2000 hours and the Atlas exposure. After continuous showering of 15 days and 24 hours drying period the contact angle of the paint when checked is maintained. The ΔΕ values of the coatings are < 2 even after 2000 hours indicating the coatings in accordance with this invention have excellent durability as well as retain the contact angle hysteresis values of < 10 degrees.
Table 22
ΔΕ: Colour difference, YI: Yellowness index, WI: whiteness index, CR: Contrast ratio The styrene based resin system C2 shows heavy chalking after Atlas, QUV exposure and the yellowness index is above 3 while for the styrene free systems, the yellowness index is below 3 and the extent of chalking was negligible.
Natural exterior exposure over a period of 6 months shows the following results:
Table 24
For the red oxide cleaning test, aqueous slurry of fine red oxide particles was applied onto the coatings and allowed to dry. a) After a period of 2 hours, the dried stain was washed off under running water. It is observed that the best cleanability and least residual stain are obtained for the systems containing a balance of both the silicones (hydrophobic and hydrophilic). For the styrene based system C2 above, poor cleanability was observed. b) After application of the stain, the panels were kept in an air circulating oven at 50°C for a period of 4 hours. The stain was then washed off under running water. Similar observations were attained. With increasing hydrophilic silicone contents, lesser residual stains were attained. However when only the single hydrophobic silicone (Al, El and Fl) was used, residual stains were more indicating an importance of the balance between the two. For the styrene based system, poor cleanability was observed. Contact angles conducive to self cleaning and the wettability switching property have been shown to be maintained even after weathering with the present invention.
Example 42: Study of different contents of hydrophobic and hydrophilic organosilicones in paint formulation of example 41
Table 25
(pbw in
coating - formulation)
Natural 9/10 5/10 5/10 4/10 2/10 2/10 exposure
rating (DPU ,
whiteness)
(higher the
better)
According to the above data a balance of hydrophobic and hydrophilic organosilicones results in cleaner systems after a period of 90 days of exterior exposure emphasizing a balance of hydrophobicity and hydrophilicity.
Example 43: Paints prepared with different micron sized particles using the water-borne acrylic random copolymer dispersions with a low content of short chain fluorinated monomer in combination with the functionalized pigments and extenders in accordance with the present invention
Table 26
Low content of fluorinated monomers result into an economic benefit and short chain fluorinated monomers (up to C6 fluorinated carbons) are environmentally friendly. All the resins had a pH of 8 to 9.
MMA: methyl methacrylate, n-BMA: n-Butyl methacrylate, MAA: Glacial methacrylic acid, t- BMA: Tertiary butyl methacrylate, BA: Butyl acrylate, 2-EHA: 2-ethylhexyl acrylate, Bisomer amine D700 form Cognis
Table 27
Quartz, 40 microns 27 0 0 0 28.8 0
Hydrophilic
organosilicone 4 4 4 4 4 4
Hydrophobic
organosilicone 2.5 2.5 2.5 2.5 2.5 2.5
Dispersion polymer 16.91 19 16.875 16.875 19 19
Glycol 1 1 1 1 1 1
Coalescent 1 1 1 1 1 1
Algaecide 2 2 2 2 2 2
Fluorosurfactant 0.05 0.05 0.05 0.05 0.05 0.05
Results 5/10 9/10 9/10 7/10 5/10 8/10
Paint 43-A shows dirt streak marks after a 12 month exterior exposure while the others do not show any dirt streaks, lines or water spots. This shows the importance of the particle size of the fillers and extenders added. Quartz used in 43A has a d50 particles size of 40 microns (size range of 1 to 160 microns). Thus it is preferred to have a coating with plurality of particles with particles having a size less than d50 of 40 microns to avoid dirt streak lines.
Example 44: Emulsions/ latex polymers prepared using the preferred monomer backbone in accordance with the present invention and their paint data
Sample latex with the preferred monomers was prepared as shown in the following table. A combination of polymerizable surfactants (ADEKA SR10, Ether sulfate with 100% solids and ER40, nonionic, 60% solids) was used to prepare the latex. It was found that using the above latex the paints showed a CAH of >20 degrees (refer example 55) even though the paint formulas were same as that used above and the monomer combination of the latex was the same. Therefore the latex polymer of example 44 for a given coating formulation alone does not reduce the value of CAH below 20 degrees and it is preferred to have a dispersion polymer in the formulation either alone or in combination with the latex polymer. Table 28
The properties of the latex prepared at 75 to 80. are as follows: 40% solids, Acid value = 15 mg KOH/g, pH=9.35, median particle size = 106.8 nm, surface energy = 29.9 ml\l/m
Example 45: Paints prepared with respirable crystalline silica free extenders using the water-borne acrylic random copolymer dispersions of example 43 (2) in combination with
the functionalized pigments in accordance with the present invention and their exterior exposure data
Table 29
Two coats of the above paints with 25% dilution were brush applied over a concrete wall over which one coat of Asian Paints exterior primer was applied. The paints 1, 2 and 3 had excellent appearance over a period of 180 days of exterior exposure. According to the above formulas, the coatings where calcium carbonate 325 mesh (44 microns) is used the coating shows some streak marks. This is reduced where nano Ti02 is used in the coating of formula 4 as compared to the coating of formula 5. Further the coatings do not show any significant chalking. Conventionally, only inorganic polymers/ binders are used to prepare paints with photocatalytic activity since organic binders undergo degradation. However, we have observed Over a period of 180 days in the field exposure that the patch does not show significant chalking (removal of surface layer due to degradation of binder). This may be Occurring in our system due to the combined presence of organic and inorganic binders and also a high particles (non degradable) to binder ratio. Further the particles may be covered with/ treated with hydrophobic and hydrophilic organosilicones during paint making or film formation which may modify their photocatalytic activity. The paints 4 and 5 were subject to QUV B (313 nm) accelerated tests for a period of 45 days. Both the paints showed negligible chalking, CAH < 10° and the desired wettability switching property after 45 days exposure in QUV B. Further, the paint containing nano Ti02 (paint 4), showed improved color retention and better durability than (paint 5).
As shown in Figure 11, the paint 1 given in Table 29 (1) when exposed outdoors for a period of 6 months on a vertical surface shows excellent resistance to dirt streaks and water marks (left side of figure: paint according to current invention, right side of figure: commercial self cleaning paint).
Example 46: Microbiology data of the paints prepared using the water-borne acrylic random copolymer dispersions in combination with the functionalized pigments in accordance with the present invention i i£r
Various encapsulated and free biocides were evaluated with paints according to the invention such as Rocima 344, Rocima 350, Rozone 2000 alone and in combination with different chemistries. Paints in accordance with the present invention, show excellent anti algal properties in accelerated algal chamber testing, showing a rating of at least 8 to 9 out of 10 after 30 days testing. Further the coatings also show excellent anti fungal and anti bacterial properties after accelerated testing.
Example 47: Photocata lytic activity of the paints 4 and 5 of example 45 prepared using the water-borne acrylic random copolymer dispersions in combination with the functionalized pigments in accordance with the present invention
The paints 4 and 5 were subject to the methylene blue discoloration test. They were brush applied on a fiber cement board panel (6 inch x 3 inch). The coating was dried under ambient conditions for 7 days.
The photoactivity of the paints was determined . by determining the photocatalytic decolorization of a methylene blue solution by UV-vis measurements. The peak of methylene blue was monitored at 664 nm.
30% of the panels were dipped in a 100 μΙ_ aqueous solution of methylene blue in a 500 mL glass beaker. The entire setup was irradiated with UV light from 2 tubes of wavelength 254 nm and 365 nm. The decolonization of the methylene blue dye was quantified on a UV- 2450, Shimadzu UV-vis spectrophotometer. The table " below shows the concentration of methylene blue in the solution after 4 hours and 8 hours of exposure showing paint 4 with 0.2% nano titania having a higher photocatalytic activity than paint 5.
Table 30: showing the concentration of methylene blue in solution after 4 h and 8 h of UV exposure
Bird droppings: Fresh liquid bird droppings were spread with a spatula on 6 inch x 3 inch panels of both the paints. These were kept under natural exterior exposure and monitored over a period of 30 days. It was observed that due to a combined effect of sunlight and rain, the paint 4 showed a lower residual stain of bird droppings as compared to paint 5. In addition the panel appeared cleaner and whiter. For comparison a non stimuli responsive paint (commercially available) which exhibits a contact angles <90 degrees and shows hydrophilicity when water is poured on its surface was also subject to the liquid bird droppings. This showed the highest residual stain of bird droppings, with the white paint becoming yellowish in the spot where the dropping was deposited. Thus paints according to this invention show improved resistance to organic deposits such as bird droppings.
Examples 48: Photocatalytic activity of the paints prepared using the water-borne acrylic random copolymer dispersions in combination with the functionalized pigments in accordance with the present invention
The coating was brush applied over Asian Paints exterior primer applied on a fibre cement board panel (6 inch x 3 inch). The coating was dried under ambient conditions for 7 days at room temperature (25 to 30 °C) and a relative humidity of 50 to 60%. The photoactivity of the paints was determined by determining the photocatalytic decolorization of a methylene blue solution by UV-vis measurements. The peak of methylene blue was monitored at 664 nm.
30% of the panels were . dipped in a 100 microliter aqueous solution of methylene blue in a 500 mL glass beaker. The entire setup was irradiated with UV light with a wavelength of 365 nm. The decolorization of the methylene blue dye was quantified on a UV-2450, Shimadzu UV-vis spectrophotometer.
Six paints containing 3 levels of nano Ti02 (0, 0.5 and 1% Evonik P25) with (sc 1, 2, and 3) and without (sc 4, 5, 6) the hydrophilic and hydrophobic organosilicones were also exposed to accelerated QUV A (333 nm) chamber testing for a period of 450 hours.
Defoamer 0.1 0.1 0.1 0.1 0.1 0.1 0.1 0.1
Thickener 0.3 0.3 0.3 0.3 0.3 0.3 0,3 0.3 pH Adjustment 0.05 0.05 0.05 0.05 0.05 0.05 0.05 0.05
Dispersing
aqent 0.5 0.5 0.5 0.5 0.5 0.5 0.5 0.5
Wetting agent 1 1 1 1 1 1 1 1
Glycol 0.5 0.5 0.5 0.5 0.5 0.5 0.5 0.5
In can
preservative 0.1 0.1 0.1 0.1 0.1 0.1 0.1 0.1
Algaecide 1.5 1.5 1.5 1.5 1.5 1.5 0 0
ΤΙ(¼ Rutile,
0.405 microns 23 23 23 23 23 23 23 23
Calcium
carbonate, 20
microns 26 26 26 26 26 26 26 26
Hydrophilic
organosilicone 4 4 4 0 0 0 8 8
Hydrophobic
organosilicone 2.5 2.5 2.5 0 0 0 O O
Nano Titan ia 0 0.5 1 0 0.5 1 0 0
Antifungal agent 0.2 0.2 0.2 0.2 0.2 0.2 0.2 0.2
Fluorosurfactant 0.05 0.05 0.05 0.05 0.05 0.05 0.05 0.05
Coalescent 1 1 1 1 1 1 1 1
Dispersion
Polymer,
example 43 (2) 19.375 19.375 19.375 19.375 19.375 19.375 19.375 16.25
Total 99 99.5 100 92.5 93 93.5 98.8 95.675
Table 32
For all the paints there was negligible chalking indicating the binder was not getting affected. The DE value of 0.5% nTi'02 is lower for both the cases indicating enhanced durability with a low dosage of nano Titania. The DE value is higher with silicones which may be due to the enhancement of photocatalytic activity caused by the presence of organosilicones.
Example 49: Enhancement of photocatalytic activity with oragnosilicones (both hydrophilic and hydrophobic) : 2 sets of paints were prepared each with 0%, 0.5% and 1% n Ti02 (p 25 from Evonik). One set contains 2 organosilicones (scl, 2 and 3) whereas the other is without organosilicones (sc4, sc5, sc6) as shown in Table 31.
30% of the panels were dipped in a 100 microliter aqueous solution of methylene blue in a 500 mL glass beaker. The entire setup was irradiated with UV light with a wavelength of 365 nm. The decolorization of the methylene blue dye was quantified on a UV-2450, Shimadzu UV-vis spectrophotometer.
Simultaneously a control sample of methylene blue (without paint panel) was also exposed under UV for 8 hours. The table shows the reduction in the concentration of methylene blue after the test as measured by the UV visible spectrophotometer. All the 6 paints contained Rutile Ti02 (0.405 microns particle size) R902 plus.
Table 33
The control sample shows that methylene blue is itself undergoing some degradation under UV (365 nm) i.e. (about 23% in 8 hours). Both paints Scl and sc4 with 0% nano Ti02 show comparable degradation under UV (~35% in 8 hours), with sc4 showing a marginally higher value due to the presence of organosilicones. This may be due to the photocata lytic activity conferred by rutile Ti02, DuPont R902 plus. In the paints sc2 to sc 6, increasing nano Titania is enhancing the activity and paints containing a combination of organosilicones is enhancing the degradation of methylene blue and hence the photocatalytic activity. With 0.5% nano Ti02 increase in activity between without and with silicones is 87% whereas with 1% nano Ti02 the enhancement is 77%. The total degradation of methylene blue is 70.67% in 8 hours for sc2 which has a combination of organosilicones while it is 37.82% for sc5 which does not have the organosilicones. Single siljcones (only hydrophobic or hydrophilic) may also be used to enhance the photocatalytic activity. However, according to the present invention a combination of hydrophobic and hydrophilic organosilicones/silanes is preferred.
Example 50
Paint without nano Titania using an alternate grade of rutile Ti02
Table 34
Paint formulations are the same as shown in example 48. Organosilicone treatment according to the present invention is creating an enhancement of the photocatalytic activity of Rutile Ti02 R931 from DuPont (particle size = 0.55 microns) by 10.64%.
Example 51: Photocatalytic activity of the coating under diffused sunlight
30% of the paint panels (6 inch x 3 inch) (scl to sc6 shown in Table 31) were immersed overnight in a 1000 microliter solution of methylene blue. Subsequently they were kept indoors away from the windows, receiving only diffused sunlight for a period of 7 days at room temperature (25 to 30 °C) and a relative humidity of 50 to 60%. The DE values of the stains were measured as shown in Table 37.
It was visually observed that the paints containing organosilicones (scl, 2 and 3) stained less to start with compared with paints sc4, 5 and 6 which did not contain any organosilicones. Even after 48 hours in dispersed sunlight the stains of the paints scl, 2 and 3 began to discolour at a faster rate than sc4, 5 and 6.
Table 35
Example 52: Rust resistance of the paints prepared according to the current invention
Different paints were prepared with 0, 0.5% and 3% nano Titania (p25 from Evonik). All the paints had the composition according to the present invention. These paints were applied on a vertical surface over an exterior primer immediately below a rusted iron bar. Observations were made over a period of 15 and 30 days during the monsoon season. For comparison 2 non stimuli responsive paints (one commercially available and one experimental, not in
lit
accordance with present invention) were applied which exhibit contact angles <90 degrees and show hydrophilicity when water is poured on their surface.
Table 36
After 15 days, the rust marks/ lines were visible only on coating 4 and 5. Coating 1, 2 and 3 show good water repellency which indicates that the water containing rust would roll off from the surface. The coatings according to the present invention have a reduced tendency of spreading of rust marks thereby leading to coatings which can maintain their aesthetic appeal for a longer time. Increased contents of nano Titania are leading to increased hydrophilicity and hence the coatings are showing increased spreading of the rust stains. The 2 non stimuli responsive hydrophilic paints taken for comparison (paint 4 and 5 in Table 36) showed inferior performance. Thus, a disadvantage of only hydrophilic surfaces is that corrosion/ rust stains tend to spread over large areas leading to loss of the aesthetic appeal of the surface. Hence it is desired to have a reversible switchable surface in accordance with the present invention.
Example 53: Paint film Water absorption
■ Table 37
Example 54: Surface energy measurements of coating according to the invention after 7 days drying under ambient conditions (30 °C and a relative humidity of 50-60%).
Table 38
**Binder used is example 43 (1).
1- prepared 15%solution of R972 in IPA.
2- 60% solution of R105 in IPA.
3- added 1 part of R972 solution to 5 parts of R105 solution
4- mixed the above solution with binder in 1 :2 ratios, i.e., 1 part of above solution to 2 parts of polymer dispersion
The paint formulas of scl to sc6 are given in Table 31.
Example 55: Contact angle hysteresis measurements of coating according to the present invention for the paints of example 54
The contact angle hysteresis is measured as the difference between advancing angle al and rl. Advancing angle for 10, 20 and 30 pL is measured (al, a2 and a3 respectively) and receding angle is measured for 20 and 10 pL (r2 and rl respectively). All measurements were carried out at 30 deg C. For the hydrophobic and hydrophilic organosilicones, their clear coats are applied on aerated cement blocks and the average value of CAH is taken as shown in Table 39.
Table 39
with only
hydrophil
ic
silicone
(sc8,
Table 31) 126.8 115.3 11.5 127.3 124.4 2.9 7.2
Paints
with
treated
particles
** 100.3 78.8 21.5 113.1 108.9 4.2 12.85
Hydrophobic
organo- silicone
applied
on
aerated
cement
block 129.3 117.3 12 130.5 126.4 4.1 8.05
Hydrophil
ic
organo- silicone
applied
on
aerated
cement
block 122 102.9 19.1 124.5 118.7 5.8 12.45
Paint
with
Emulsion
25 122.6 95 27.6 109.7 104.7 5 13.5
Paint
with
Emulsion
44 118.1 94.2 22 112.3 107,3 5 16.3
Example 56: Paints prepared with different contents of wetting and dispersing agents showing the effect on accelerated storage stability (30 days at 55 C) in accordance with the present invention with varying contents of glycol
Table 40
Dispersing
aqent 0.5 1 1 0.5
Hydrophilic
organosilicones 4 4 4 4
Hydrophobic
organosilicone . 2.5 2.5 2.5 2.5
Polymeric
Dispersion of
example 43 (2) 16.875 16.875 16.875 16.875
100 101 100.5 100.5
Initial Stormer
Viscosity 353 235 365 225 g
Viscosity in 15 Gelled in Gelled in
days 7days 900 7days 650 g
Viscosity in Above
30days Results Discontinued 1090 g Discontinued 925 g
The above data shows that a balance of the wetting agent (in this study Polyoxyethylene (9) C12-C15 alcohol (HLB =13) and the anionic dispersing agent are important to achieve accelerated storage stability (30 days at 55 deg C). This test indicates the shelf life of the paint; hence it is important to add suitable quantities of wetting and dispersing agents along with glycols to achieve storage stability.
Conventional paint systems based on latex polymers do not show such an observation of viscosity pickup on increase of dispersing agent. This was also the case when the emulsion of example 25 or example 44 was used in the paint formula; no viscosity pickup was observed on increase of the dispersing agent.
Hence according to the present invention it is also important to have a balance between the wetting agent and the dispersing agent in order to obtain a suitable shelf life and paint stability at 55D for 30 days. It is thus possible by way of the present advancement to provide for said aqueous coating composition that is oleophobic and has a water contact angle hysteresis of < 25° under certain environmental conditions with variations in at least one or more of temperature, humidity, pH, light which is reversibly switchable to a water contact angle hysteresis of >25° under said environmental conditions offering benefits of both superhydrophobic and superhydrophilic cleaning, good organic and inorganic dirt pickup resistance, resistance to dirt streaks, dirt lines, water spotting, water marks, good cleanability with water and forced air or wind, reduced chalking, good resistance to organic dirt and deposits such as bird droppings and oily stains, and reduced tendency for spreading of water containing rust with the additional benefits of better binding, durability .
Claims
1. Aqueous coating composition exhibiting reversible hydrophilic-hydrophobic switching under variable environmental conditions including variations in at least one or more of temperature, pH, humidity or other external stimuli including light comprising :
a) One or more oligomeric or polymeric dispersion/binder comprising acrylic, fluorinated and hydrophilic (acid/amine/ionic/non-ionic) backbone;
b) One or more hydrophobic organosilicone or silane (monomeric, oligomeric or polymeric); c) One or more hydrophilic organosilicone or silane (monomeric, oligomeric or polymeric); and
d) At least partially surface treated particles with a mean particle size less than or equal to 100 microns, preferably less than or equal to 325 mesh (44 microns)
to selectively provide a balance of hydrophobicity and hydrophilicity such as to provide desired reversible hydrophilic-hydrophobic switching.
2. Aqueous coating composition as claimed in claim 1 that is oleophobic and has a water contact angle hysteresis of <25° under certain environmental conditions with variations in at least one or more of temperature, humidity, pH, light which is reversibly switchable to a water contact angle hysteresis of >25° under said environmental conditions offering benefits of both superhydrophobic and superhydrophilic cleaning, good organic and inorganic dirt pickup resistance, resistance to dirt streaks, dirt lines, water spotting, water marks, good cleanability with water and forced air or wind, reduced chalking, good resistance to organic dirt and deposits such as bird droppings and oily stains, and reduced tendency for spreading of water containing rust. '
3. Aqueous coating composition as claimed in anyone of claims 1 or 2 that reversibly switches from superhydrophobic (contact angle hysteresis <25° and water droplet rolling effect) to superhydrophilic (contact angle hysteresis >25° and complete wetting) for at least 2000 hours of accelerated exterior exposure (Atlas and QUV testing), preferably the contact angle hysteresis values are <20°, more preferably < 10° and most preferably <5° in one state and higher in the switched state for a deposited 0.02 ml. water droplet, wherein preferably the contact angle exhibited in one state is >100° with water, more preferably >120°, even more preferably superhydrophobic having contact angle >150° and contact angle <90° in the switched state for a deposited 0.02 ml_ droplet; and wherein said
switching takes place within a varying period of time from a few seconds to several minutes dependent on a particular stimuli.
4. Aqueous coating composition as claimed in anyone of the preceding claims having low surface energy of <40 ml\l/m and preferably <20 mN/m and most preferably <10 mN/m even with the second coat and hence recoatable to enhance film build-up to attain desired opacity/ translucency/ hiding/ whiteness.
5. Aqueous coating composition as claimed in anyone of the preceding claims comprising at least one or more wetting and dispersing agents, encapsulated or free antimicrobial agents selected from fungicide, algaecide, in-can preservative and optionally involving, one or more nanoparticles exhibiting photocata lytic activity
6. Aqueous coating composition as claimed in anyone of the preceding claims wherein said polymeric dispersion comprises dispersion polymers involving primary or secondary dispersion in water or in a water/ co-solvent mixture with or without emulsion/latex polymers.
7. Aqueous coating composition as claimed in anyone of the preceding claims wherein said polymeric dispersion comprises dispersion polymers with or without emulsion/ latex polymers that is preferably styrene free and is a free radical polymerization or controlled radical polymerization product of hydrophobic monomers C1-C8 straight, branched or cyclic esters of methacrylic acid/ acrylic acid selected from methyl methacrylate, ethyl methacrylate, butyl Methacrylate, isobutyl Methacrylate, tertiary butyl methacrylate, isobornyl methacrylate, 2-ethylhexyl methacrylate and various isomers and their corresponding acrylates also involving >C8 esters of methacrylic acid/ acrylic acid selected from lauryl acrylate, stearyl acrylate.
8. Aqueous coating composition as claimed in anyone of the preceding claims wherein said polymeric dispersion comprises dispersion polymers with or without emulsion/ latex polymers and has monomers selected such that the resulting polymer has an average octanol-water partition coefficient (log Kow) greater than 0.6, preferably greater than 1.5 and more preferably greater than 2.
9. Aqueous coating composition as claimed in anyone of the preceding claims wherein said polymeric dispersion including dispersion polymers with or without emulsion/ latex polymers in being styrene free involves styrene or alkylstyrene group as part of the polymer or as a part of additives including opacifiers in less than 15% of the film forming composition favouring reduced chalking of said composition on UV exposure.
10. Aqueous coating composition as claimed in anyone of the preceding claims wherein said polymeric dispersion comprising dispersion polymers and emulsion/ latex polymers is in the ratio range of dispersion to emulsion polymers of 95: 5 to 5:95.
11. Aqueous coating composition as claimed in anyone of the preceding claims wherein said polymeric dispersion comprising dispersion polymers and emulsion/ latex polymers is preferably in the ratio range of 30:70. '
12. Aqueous coating composition as claimed in anyone of the preceding claims wherein said polymeric dispersion comprising dispersion polymers includes primary or secondary dispersion in water or in a water/ co-solvent mixture involves co-solvent that is VOC exempt under Green Seal standard GS-11 with resulting coating VOC's in the range of 1 to 100 grams/liter.
13. Aqueous coating composition as claimed in anyone of the preceding claims wherein said polymeric dispersion comprising dispersion polymers with or without emulsion/ latex polymers includes polymers selected from natural polymer, a neoprene polymer, a nitrile polymer, a vinyl acrylic polymer, acrylic polymer; styrene acrylic polymer, styrene butadiene polymer, copolymer of vinyl acetate and butyl acrylate- or veova monomers or ethylene/ propylene/ butylene, butadiene, isoprene involving crosslinkers selected from diacrylates, triacrylates, multifunctional acrylates, silane functional monomers, acetoacetate functional monomers, monomers with aziridine, hydrazine, carbodiimide or allylic functionality.
14. Aqueous coating composition as claimed in anyone of the preceding claims wherein said polymeric dispersion comprising dispersion polymers with or without emulsion/ latex polymers includes polymers of fluorinated monomers comprising 2 to 20 carbon atoms and polymeric fluorine content in the range of 0.1 to 50% on monomer solids and preferably in
the range of less than 5% on monomer solids resulting in economic benefits and a surface energy of <50 ml\l/m , preferably <30 ml\l/m and more preferably <20 mN/m
15. Aqueous coating composition as claimed in anyone of the preceding claims wherein said polymeric dispersion comprising fluorinated backbone comprises fluorinated monomers including esters of acrylic or methacrylic acid with a linear or branched perfluoroalkyl functional group preferably involving 1-6 fluorinated carbons selected from trifluoroethyl meth(acrylate), pentafluoropropyl meth(acrylate), heptafluorobutyl meth(acrylate).
16. Aqueous coating composition as claimed in anyone of the preceding claims wherein said ionic or ionizable or non-ionic hydrophilic monomers are present within the range of 2 to 20% of polymer solids thus favoring reduction of water absorption of said composition to less than 15%, preferably less than 10% and even more preferably less than 5% after remaining immersed in water for 96 hours.
17. Aqueous coating composition as claimed in anyone of the preceding claims wherein said ionic, or ionizable or non-ionic hydrophilic monomers are selected from acrylic acid, methacrylic acid and their aklali metal or quaternary ammonium ion salts; monoolefinic sulphonic acid compounds and their alkali metal salts including 2-acrylamido-2- methylpropanesulphonic acid; acrylates or methacrylates of aminoalcohols, acrylamides selected from Ν,Ν-dimethylaminopropylacrylamide or N, N-
Dimethylaminoethylmethacrylate, polyalkylene oxide group containing monomers or a polymeric chain prepared by polymerizing a polyalkylene glycol (meth)acrylate, hydroxy! alkyl (ethyl, propyl or butyl) (meth)acrylates, wherein the hydrophilic component comprises at least one oxygen, sulfur, phosphorus and/or nitrogen atom.
18. An aqueous coating composition as claimed in anyone of the preceding claims wherein said acrylic, fluorinated and amine backbone or acrylic, fluorinated and acid backbone of the primary or secondary polymeric dispersion comprises sufficient content of acid or amine monomer including ionic or non-ionic external surfactants adapted for acid or amine value in the range 5 to 100 mg KOH/g.
19. Aqueous coating composition as claimed in anyone of the preceding claims wherein said particles comprising either micron sized particles or including particulate mixture of both
micron sized and nano sized particles and polymeric dispersion are present in selective ratios of 0.9 to 2.5 wherein higher ratios closer to 2 is preferred for a two coat application.
20. Aqueous coating composition as claimed in anyone of the preceding claims wherein said particles comprise either micron sized particles or includes particulate mixture of both micron sized and nano sized particles, and including particulate pigments wherein the pigment volume concentration (PVC) is in the range of about 40 to 90 % and preferably in the range of 60 to 80% for the coating and preferably the coating PVC is lower than the practical critical pigment volume concentration (CPVC).
21. Ah aqueous coating composition as claimed in anyone of the preceding claims wherein said particles comprising either micron sized particles or including particulate mixture of both micron sized and nano sized particles comprises micronized and nanosized particles present in selective ratios of 3.5 to 8 with ratios closer to 8 being preferred for a two coat application.
22. Aqueous coating composition as claimed in anyone of the preceding claims wherein said at least partially surface treated particles involve organic and/or inorganic particles having size ranging from 1 nm -100 microns includes surface treated nanometer sized organic and/or inorganic particles have size ranging from about 1 nm to 300 nm, and wherein said particles preferably has mean particle size less than 325 mesh (44 microns) and even more preferably less than 40 microns.
23; Aqueous coating composition as claimed in anyone of the preceding claims wherein the mass ratio of said hydrophobic organosilicone to hydrophilic organosilicone is in the range 10/90 to 60/40.
24. Aqueous coating composition as claimed in anyone of the preceding claims wherein said hydrophilic organosilicone/silane having a contact angle hysteresis of > 10 degrees based on the average of readings for 10 and 20 pL of water when applied on an aerated cement block is present in the range of 10% to 90% by weight of dispersion solids in said composition.
25. Aqueous coating composition as claimed in anyone of the preceding claims wherein said hydrophobic organosilicone/silane having a contact angle hysteresis of <10 degrees based on the average of readings for 10 and 20 pL of water when applied on an aerated cement
block is present in the range of 5% to 80% by weight of dispersion solids in said composition.
26. Aqueous coating composition as claimed in anyone of the preceding claims wherein said hydrophobic and hydrophilic organosilicone content present as a part of the continuous phase in said composition is 10-90 % of the weight of the dispersion/binder.
27. Aqueous coating composition as claimed in anyone of the preceding claims wherein said wetting agent and dispersing agent is balanced by having said dispersing agent upto 1% and preferably in the range of 0.1 to 0.5% and the wetting agent upto 2% and preferably in the range of 0.2 to 1% by weight in said composition favouring accelerated storage stability for atieast 30 days at 55°C and a composition that passes at least 5 cycles of freeze thaw stability test (-15°C to 30°C).
28. Aqueous coating composition as claimed in anyone of the preceding claims including photocatalytically active agents comprising one or more nano particles exhibiting photocatalytic properties in amounts less than l% .and preferably less than 0.5% of total formulation especially for coatings containing particles more than or equal to 325 mesh, preferably comprise nano titania of the anatase or rutile grade in the range of 0.1 to 1% of the formulation with a particle size of 1 to 300 nm and more preferably comprise titania in anatase form adapted for enhancement of photocatalytic activity of said composition while maintaining its durability by even micron sized particles (>300 nm) of Titania (anatase or rutile) against organic pollutants of atieast >5%, more preferably >20% and most preferably >80% of the activity of the control.
29. Aqueous coating composition as claimed in anyone of the preceding claims wherein said at least partially surface treated particles are selected from substantially spherical particles, nodular particles, platy particles, cubical paraticles, various irregularly shaped particles, and mixtures thereof.
30. Aqueous coating composition as claimed in anyone of the preceding claims wherein said at least partially surface treated particles comprise at least one material selected from the group of silicates, doped silicates, minerals, metal oxides, silicas and metal powders and are selected from the group of aluminum oxides (alumina), titanium oxide, zirconium oxide, silver, nickel, nickel oxide, iron oxide, and alloys, polystyrene particles, (meth)acrylate
particles, PTFE particles, silica particles, polyolefin particles, polycarbonate particles, polysiloxane particles, silicone particles, polyester particles, polyamide particles, polyurethane particles, ethylenically unsaturated polymer particles, polyanhydride particles, biodegradable particles, polycaprolactone, nanofibers, nanotubes, nanowires, or combinations thereof that are preferably insoluble in organic solvents and water; and preferably comprise particles other than respirable crystalline silica wherein said silica is amorphous.
31. An aqueous coating composition as claimed in anyone of the preceding claims wherein said at least partially surface treated particles comprise particles that are surface treated with compounds selected from silanes (alkylsilanes, perfluoroalkylsilanes, and alkyldisilazanes), silicones and fluorinated compounds or fluorinated acrylics or other organic/ inorganic treatments to achieve a balance between hydrophobicity and hydrophilicity.
32. An aqueous coating composition as claimed in anyone of the preceding claims wherein said at least partially surface treated particles comprise preferably externally surface treated particles with selective hydrophobic and hydrophilic balance and are obtained of sol-gel treatment to prevent any colour change or graying of white particles present in said coating composition, especially where fluorosilanes or fluorosilicones are used for treatment or is alternatively in-situ treated while preparation of the coating with said mixture of hydrophobic and hydrophilic silicones/silanes.
33. Aqueous coating composition as claimed in anyone of the preceding claims wherein said coating composition is flowable comprising at least one organic diluent or solvent, wherein the solvent is selected from the group comprising of water, an alcohol, an ether, a ketone, an ester, a glycol, a glycol ether, an alkylene carbonate, a C5 - C18 aliphatic hydrocarbon, a C6 - Cig aromatic hydrocarbon, various polar/non-polar solvents and mixtures thereof.
34. Aqueous coating composition as claimed in anyone of the preceding claims wherein said polymeric dispersion comprises thermoplastic or thermosetting polymers including a single polymer or a blend of more than one polymer or self crosslinking polymers selected from acrylics, polyesters, polyurethanes, polycarbonates, polyolefins, alkyds, epoxies, polyamides, fluoropolymers, silicone polymers and their hybrids, wherein said fluoropolymers include polyperfluoropolyethers, and a polymer having one or more
13$
monomer repeat units selected from the group consisting of ethylene, propylene, styrene, tetrafluoroethylene, vinylidene fluoride, hexafluoropropylene, perfluoro(methyl vinyl ether), perfluoro(ethyl vinyl ether), and perfluoro(propyl vinyl ether) and/ or includes a colloid, a latex, or a suspension of a fluoropolymer selected from a group comprising of polytetrafluoroethylene; perfluoroalkoxy polymer resin; copolymer of tetrafluoroethylene and hexafluoropropylene; copolymers of hexafluoropropylene and vinylidene fluoride; terpolymers of tetrafluoroethylene, vinylidene fluoride, and hexafluoropropylene.
35. Aqueous coating composition as claimed in anyone of the preceding claims wherein said polymeric dispersion comprising polymers have a Tg of -30 to 100 deg C or preferably Tg in the range of 0 to 50°C.
36. Aqueous coating composition as claimed in anyone of the preceding claims wherein said polymeric dispersion comprising polymers have surface energy of <50 mN/m and preferably less than 30 mIM/m and more preferably <20 mN/m.
37. An aqueous coating composition as claimed in anyone of the preceding claims comprising components that are multifunctional or having individual functions selected from thickeners, biocides, wetting and dispersing agents, fluorosurfactants, defoamers, opacifying polymers, cosolvents, coalescents, plasticizers, pigments, special effect pigments, extenders, colorants, freeze thaw stabilizers, buffers, fire retardants, uv absorbers, organic fiber material, and inorganic fiber materials, flow and levelling agents, adhesion promoters, oil and water repellents and other components known to be provided in paint formulations.
38. An aqueous coating composition as claimed in anyone of the preceding claims that is ambient curing including air curing, infra-red curing and thermal curing.
39. An aqueous coating composition as claimed in anyone of the preceding claims wherein a dry film of said coating has a dry film thickness of 1 to 200 microns and preferably 40 to
120 microns and withstands at least 30 to 1000 cycles and preferably 100 to 3000 cycles in a wet scrub resistance test with a scrub brush and a 0.45 kg load.
40. An aqueous coating composition as claimed in anyone of the preceding claims adapted for applications comprising self cleaning, easy cleaning, anti-graffiti, stain resistant, soil-
degrading, deodorizing, air-cleaning, antifouling, antimicrobial, water-cleaning, superhydrophobicizing, superhydrophilicizing and/or antifogging effects.
41. A process for the preparation of aqueous coating composition as claimed in anyone of the preceding claims 1-40 comprising the steps of:
a) providing at least partially surface treated organic or inorganic particles to selectively provide a balance of hydrophobicity and hydrophilicity that is either micron sized or includes a particulate mixture of micron sized and nanometer sized particles;
b) providing for one or more oiigomeric or polymeric dispersion/binder comprising acrylic, fluorinated and hydrophilic (acid/amine/ionic/non-ionic) backbone;
c) providing for one or more hydrophobic organosilicone or silane (monomeric, oiigomeric or polymeric) and one or more hydrophilic organosilicone or silane (monomeric, oiigomeric or polymeric);
d) mixing said particles with said polymeric dispersion and silane; to obtain said aqueous coating composition therefrom having desired reversible hydrophilic-hydrophobic switching adapted for coating various types of substrates by brush, spray or roller type applications and also dipping, padding, doctor blade, wipe, wet-on-wet procedure or spin coating type applications.
42. A process for the preparation of aqueous coating composition as claimed in claim 41 wherein said at least partially surface treated organic or inorganic particles is provided by subjecting the particles to sol-gel treatment with compounds selected from silicones, fluorinated compounds to prevent color change of the particles.
43. A process for the preparation of aqueous coating composition as claimed in anyone of claims 41 or 42 wherein said step (a) of providing treated organic or inorganic particles that is either micron sized has a mean particle size of less than or equal to 100 microns, preferably less than or equal to 325 mesh (44 microns) or providing a particulate mixture of micron and nanometer sized particles involves providing micro and nanosized particles in selective ratios of 3.5 to 8, and are externally or in-situ treated particles with a balance of hydrophobicity and hydrophilicity;
wherein said step (d) comprising mixing said particles preferably in a slurry form in a water miscible co-solvent to the polymer dispersion including organosilicone or silane is mixed in selective ratios of 0.9 to 2.5.
44. A process for the preparation of aqueous coating composition as claimed in anyone of claims 41-43 adapted for substrates selected from masonary, concrete, cementitious, plaster, baked clay tiles, cellulosic, wood, one or more polymer, dry or damp surfaces, brick, tile, stone, grout, mortar, composite materials, gypsum board, , porous and non porous surfaces, interior surfaces or surfaces exposed to weathering on at least one surface of the substrate, which surface is preferably pretreated for better adhesion of the aqueous coating composition with various types of surface treatments.
45. A commercial or industrial material, a structure or building or a substrate comprising aqueous coating reversible switchable composition of claims 1-40 exhibiting reversible hydrophilic-hydrophobic switching under variable environmental conditions including variations in at least one or more of temperature, pH, humidity or other external stimuli including light.
46. A method of producing reversible switchable coated commercial or industrial material, structure or building or substrate comprising
selectively applying the aqueous coating of said reversible switchable composition of claims 1-40 to atleast a portion of commercial or industrial material, a structure or building or a substrate requiring said reversible coating characteristic such as to exhibit reversible hydrophilic-hydrophobic switching under variable environmental conditions including variations in at least one or more of temperature, pH, humidity or other external stimuli including light.
Applications Claiming Priority (2)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| IN3538MU2012 | 2012-12-17 | ||
| IN3538/MUM/2012 | 2012-12-17 |
Publications (1)
| Publication Number | Publication Date |
|---|---|
| WO2014097309A1 true WO2014097309A1 (en) | 2014-06-26 |
Family
ID=48464052
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| PCT/IN2013/000101 WO2014097309A1 (en) | 2012-12-17 | 2013-02-18 | Stimuli responsive self cleaning coating |
Country Status (1)
| Country | Link |
|---|---|
| WO (1) | WO2014097309A1 (en) |
Cited By (80)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| CN104888497A (en) * | 2015-05-28 | 2015-09-09 | 南京理工大学 | Super-hydrophobic and super-oleophylic fluorocarbon resin block and preparation method thereof |
| CN104910792A (en) * | 2015-05-27 | 2015-09-16 | 天津市美冠科技有限公司 | Aqueous UV coating material and preparation method thereof |
| US20160032227A1 (en) * | 2014-07-31 | 2016-02-04 | Jsr Corporation | Cleaning composition for semiconductor substrate and cleaning method |
| WO2016088131A1 (en) * | 2014-12-01 | 2016-06-09 | Asian Paints Ltd. | Surfactant stabilized vam-veova10 terpolymer based emulsion with good freeze thaw stability and a process thereof |
| WO2016191890A1 (en) * | 2015-06-04 | 2016-12-08 | Greencentre Canada | Switchable water-based paint or coating compositions |
| CN106221469A (en) * | 2016-08-26 | 2016-12-14 | 上海绿特丹保温工程有限公司 | A kind of water-water reflective insulation anti-fog haze multicolor finish |
| CN106280834A (en) * | 2015-01-12 | 2017-01-04 | 王滨 | The application in concrete for marine works protects of the hydrophobic and oil repellent type protective agent |
| WO2017012033A1 (en) | 2015-07-21 | 2017-01-26 | Rohm And Haas Company | An aqueous coating composition |
| CN106554711A (en) * | 2016-11-29 | 2017-04-05 | 国网山东省电力公司电力科学研究院 | Super-amphiphobic coating and preparation method thereof is damaged in polyurethane integration heat resistanceheat resistant |
| US9718737B2 (en) | 2015-04-21 | 2017-08-01 | Behr Process Corporation | Decorative coating compositions |
| WO2017132350A1 (en) | 2016-01-29 | 2017-08-03 | Tolmar Inc. | Purification and decolorization of polymers |
| CN107189603A (en) * | 2017-07-10 | 2017-09-22 | 漳州鑫展旺化工有限公司 | A kind of fire-proof and explosion-proof vehicle glass coating of aqueous high-temperature-resistant |
| WO2017201257A1 (en) * | 2016-05-18 | 2017-11-23 | Ppg Industries Ohio, Inc. | Waterborne compositions and compact processes of forming multi-component composite coating compositions on substrates |
| CN107429119A (en) * | 2015-06-25 | 2017-12-01 | 旭化成株式会社 | Film |
| WO2017220591A1 (en) * | 2016-06-20 | 2017-12-28 | Université de Mons | Omniphobic surface coatings |
| EP3279273A1 (en) * | 2016-08-05 | 2018-02-07 | Malgorzata Brzezinska-Kisiel P.P.H. "ALCHEM" | A composition, use of the composition, an impregnant and a method for the preparing the composition |
| WO2018027271A1 (en) * | 2016-08-11 | 2018-02-15 | Guard It Solutions Pty Ltd | Compositions for sealing and/or protecting porous substrates |
| CN107779030A (en) * | 2017-11-10 | 2018-03-09 | 重庆大学 | A kind of preparation method of high strength alumin ium alloy durability super-double-hydrophobic surface |
| CN108248271A (en) * | 2018-02-05 | 2018-07-06 | 河南工学院 | A kind of multicolor woodcut and its making material |
| WO2018138253A1 (en) * | 2017-01-26 | 2018-08-02 | Sika Technology Ag | Multilayer decorative coating composition with low discolouration |
| WO2018185710A1 (en) * | 2017-04-07 | 2018-10-11 | Sabic Global Technologies B.V. | Durable surface hardened coating or overcoating for protecting plants from pests |
| CN109096857A (en) * | 2018-07-26 | 2018-12-28 | 深圳市莱莉雅环保科技有限公司 | The aqueous anti-graffiti coating and its preparation process of scintilla water dispersion |
| CN109161320A (en) * | 2018-08-03 | 2019-01-08 | 苏州东沧涂料科技有限公司 | A kind of aqueous matt coating and preparation method thereof |
| CN109374793A (en) * | 2018-12-20 | 2019-02-22 | 北京市环境保护科学研究院 | A method of organic carbon-water partition coefficient of aromatic hydrocarbon in the practical soil of measurement |
| CN109666465A (en) * | 2017-10-17 | 2019-04-23 | 中石化石油工程技术服务有限公司 | A kind of preparation method and drilling fluid of drilling fluid latex sealing agent |
| CN109722062A (en) * | 2018-12-21 | 2019-05-07 | 英德科迪颜料技术有限公司 | A kind of preparation method of core-shell structure transparent ferric oxide coated pigment |
| CN110079169A (en) * | 2014-09-09 | 2019-08-02 | 霍尼韦尔国际公司 | Low VOC and high solid fluoropolymer for coating application |
| US10377647B2 (en) | 2010-12-15 | 2019-08-13 | Queen's University at Kingson | Systems and methods for use of water with switchable ionic strength |
| WO2019166899A1 (en) * | 2018-02-28 | 2019-09-06 | Battrion Ag | Method for producing a coating |
| CN110325484A (en) * | 2017-02-28 | 2019-10-11 | 法国圣戈班玻璃厂 | The article for moist chamber containing hydrophobicity glass pane |
| EP3556817A1 (en) * | 2018-04-18 | 2019-10-23 | The University of Akron | Functionalized polymer compositions for low voc coalescence of water based emulsions |
| CN110373076A (en) * | 2019-07-12 | 2019-10-25 | 成都新柯力化工科技有限公司 | A kind of environment protecting building paint and preparation method of photocatalytic degradation haze precursor |
| CN110433671A (en) * | 2019-07-23 | 2019-11-12 | 江苏大学 | A kind of preparation method and its usage of visible light-inducing automatically cleaning carbon-fiber film |
| CN110564237A (en) * | 2019-08-13 | 2019-12-13 | 安徽朗凯奇建材有限公司 | waterproof material for metal roof and preparation method thereof |
| US10533096B2 (en) | 2015-02-27 | 2020-01-14 | Kimberly-Clark Worldwide, Inc. | Non-fluorinated water-based superhydrophobic compositions |
| US10584249B2 (en) | 2015-10-28 | 2020-03-10 | 3M Innovative Properties Company | Articles subject to ice formation comprising a repellent surface |
| AU2017255540B2 (en) * | 2016-04-26 | 2020-04-09 | 3M Innovative Properties Company | Spray application systems components comprising a repellent surface comprising a siloxane material and methods |
| CN111019456A (en) * | 2019-11-27 | 2020-04-17 | 昆明理工大电力工程技术有限公司 | Durable super-hydrophobic three-proofing coating as well as preparation method and use method thereof |
| RU2719471C2 (en) * | 2015-06-24 | 2020-04-17 | ЭйЭм ТЕКНОЛОДЖИ ЛИМИТЕД | Photocatalytic composition based on air binder and use thereof to produce water-based paints, particularly for internal works |
| WO2020113005A1 (en) * | 2018-11-29 | 2020-06-04 | Henry Company, Llc | Self-cleaning, dirt pick-up resistant compositions for roof coatings |
| CN111266679A (en) * | 2019-11-27 | 2020-06-12 | 南京航空航天大学 | Preparation method of self-propelled functional surface and surface tension storage tank based on surface |
| WO2020120006A1 (en) * | 2018-12-14 | 2020-06-18 | Merck Patent Gmbh | Surface coating compositions |
| WO2020139601A1 (en) * | 2018-12-26 | 2020-07-02 | Nitto Denko Corporation | Superhydrophilic coating composition |
| CN111454394A (en) * | 2020-04-22 | 2020-07-28 | 昆明理工大学 | Water-based acrylic composite resin and preparation method thereof |
| EP3536719A4 (en) * | 2016-11-01 | 2020-07-29 | Unimatec Co., Ltd. | Fluoropolymer and rust preventive containing same as active ingredient |
| WO2020180760A1 (en) * | 2019-03-05 | 2020-09-10 | Board Of Trustees Of Michigan State University | Omniphobic polyurethane compositions, related articles, and related methods |
| CN111644209A (en) * | 2020-08-05 | 2020-09-11 | 上海山恒生态科技股份有限公司 | Preparation method of photocatalyst for treating organic sewage |
| US10793737B2 (en) | 2017-12-29 | 2020-10-06 | Behr Process Corporation | Fast dry stain formula |
| WO2020210988A1 (en) | 2019-04-16 | 2020-10-22 | Dow Global Technologies Llc | Freeze-thaw additive composition |
| CN111954699A (en) * | 2018-04-13 | 2020-11-17 | 宣伟投资管理有限公司 | Coating composition for polymeric roofing materials |
| CN112154020A (en) * | 2018-02-15 | 2020-12-29 | 唐纳森公司 | Filter media construction |
| CN112159117A (en) * | 2020-10-10 | 2021-01-01 | 东北石油大学 | Method for rapidly preparing underwater super-oleophobic anticorrosive coating |
| WO2021003182A1 (en) * | 2019-07-01 | 2021-01-07 | Basf Corporation | Surface modified kaolin pigment and method thereof |
| US10907070B2 (en) | 2016-04-26 | 2021-02-02 | 3M Innovative Properties Company | Articles subject to ice formation comprising a repellent surface comprising a siloxane material |
| US10987686B2 (en) | 2014-10-28 | 2021-04-27 | 3M Innovative Properties Company | Spray application system components comprising a repellent surface and methods |
| CN112745752A (en) * | 2020-12-29 | 2021-05-04 | 广东邦固化学科技有限公司 | Water-based nano super-amphiphobic coating and preparation method thereof |
| WO2021095043A1 (en) * | 2019-11-17 | 2021-05-20 | The State Of Israel, Ministry Of Agriculture & Rural Development, Agricultural Research Organization (Aro) (Volcani Center) | Superhydrophobic coatings based on pickering emulsions |
| CN112852191A (en) * | 2020-12-22 | 2021-05-28 | 湖北大学 | Preparation method of tungsten trioxide super-smooth coating with long-term stability and applicable to mist collection |
| CN112981973A (en) * | 2021-03-19 | 2021-06-18 | 南昌航空大学 | Preparation method of pH response super-hydrophobic material |
| WO2021207805A1 (en) * | 2020-04-13 | 2021-10-21 | Bonilha Mariana | Water-based composition for producing an antiviral, antimicrobial coating film |
| CN113631674A (en) * | 2019-03-29 | 2021-11-09 | Ppg工业俄亥俄公司 | Single component hydrophobic coating |
| US11189824B2 (en) | 2016-09-06 | 2021-11-30 | Battrion Ag | Method and apparatus for applying magnetic fields to an article |
| CN113717557A (en) * | 2020-05-25 | 2021-11-30 | 富思特新材料科技发展股份有限公司 | Self-layering inorganic hydrophobic coating material and preparation method thereof |
| CN113856243A (en) * | 2021-10-15 | 2021-12-31 | 山东大学 | Oil-water separation material containing fluorine silsesquioxane modified epoxy resin and preparation method and application thereof |
| CN113897114A (en) * | 2021-10-14 | 2022-01-07 | 江苏利信新型建筑模板有限公司 | Composite green aluminum alloy building template and processing technology thereof |
| CN113912936A (en) * | 2021-09-30 | 2022-01-11 | 成都金发科技新材料有限公司 | Toughened and antistatic polypropylene composition and preparation method and application thereof |
| CN114292593A (en) * | 2022-01-17 | 2022-04-08 | 山东中凯华瑞工程材料有限公司 | Nano micron and submicron micron spacing structure bionic all-hydrophobic material and preparation method thereof |
| CN114377478A (en) * | 2016-08-16 | 2022-04-22 | 唐纳森公司 | Hydrocarbon fluid-water separation |
| US20220354122A1 (en) * | 2021-05-07 | 2022-11-10 | The University Of Hong Kong | pH/CO2-RESPONSIVE SMART ANTI-PATHOGEN COATINGS THAT CONTROL, REPEL, AND/OR INACTIVATE VIRUSES AND BACTERIA |
| US11498853B2 (en) | 2010-02-10 | 2022-11-15 | Queen's University At Kingston | Water with switchable ionic strength |
| CN115350887A (en) * | 2022-07-25 | 2022-11-18 | 华北理工大学 | Method for preparing super-hydrophobic surface with mechanical durability |
| US11525068B2 (en) | 2017-10-11 | 2022-12-13 | Build Care Oy | Method of producing an elastic coating and an elastic coating |
| WO2023016657A1 (en) * | 2021-08-13 | 2023-02-16 | Daw Se | Magnetizable aqueous putty, dry preparation for magnetizable coating compositions, magnetizable emulsion paints and uses thereof |
| CN115821640A (en) * | 2022-11-22 | 2023-03-21 | 启东欣联壁纸有限公司 | Waterproof stain-resistant wallpaper and preparation method thereof |
| CN116179025A (en) * | 2022-12-08 | 2023-05-30 | 江苏金陵特种涂料有限公司 | Water-based carbon nano cold zinc spraying coating and preparation method and application thereof |
| CN116355471A (en) * | 2023-03-31 | 2023-06-30 | 广东晟毅新材料科技有限公司 | Super-hydrophobic coating, super-hydrophobic glass and super-hydrophobic toughened glass and preparation method thereof |
| EP4216258A1 (en) * | 2014-12-19 | 2023-07-26 | Applied Materials, Inc. | Components for a chemical mechanical polishing tool |
| CN116891682A (en) * | 2023-09-04 | 2023-10-17 | 北京特思迪半导体设备有限公司 | Modified polydimethylsiloxane amphiphobic coating and preparation method and application thereof |
| CN117363136A (en) * | 2023-10-24 | 2024-01-09 | 江门职业技术学院 | Cross-linked self-repairing coating and preparation method thereof |
| CN117363136B (en) * | 2023-10-24 | 2024-04-09 | 江门职业技术学院 | Cross-linked self-repairing coating and preparation method thereof |
Citations (95)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| CH268258A (en) | 1946-07-30 | 1950-08-16 | Rhone Poulenc Chemicals | Water repellent coating. |
| US3227675A (en) | 1963-05-01 | 1966-01-04 | Huber Corp J M | Silane-treated clay reinforced resin compositions |
| US3354022A (en) | 1964-03-31 | 1967-11-21 | Du Pont | Water-repellant surface |
| DE2107082A1 (en) | 1970-02-16 | 1971-08-26 | Ici Ltd | Surface treated powdered silica and process for the manufacture thereof |
| DE2150736A1 (en) | 1971-10-12 | 1973-04-19 | Hoeveling Fa Emil G V | Outdoor paints - for facades based on a siloxane resin methylmethacrlate copolymer combination |
| DE2161591A1 (en) | 1971-12-11 | 1973-06-14 | Stockhausen & Cie Chem Fab | Glass wetting - eg wind-screen clearing, with aq solns contg cation active high mol wt polyelectrolytes |
| DE2307422A1 (en) | 1972-02-17 | 1973-08-23 | Cabot Corp | COATING COMPOUNDS AND METHOD FOR ITS USE |
| DE2352242A1 (en) | 1973-10-16 | 1975-04-24 | Hoeveling Emil G Von | PIGMENTED PAINT, IN PARTICULAR FAÇADE PROTECTIVE PAINT |
| US4061503A (en) | 1976-09-29 | 1977-12-06 | Union Carbide Corporation | Silane treatment of titanium dioxide pigment |
| US4147851A (en) | 1978-06-13 | 1979-04-03 | E. I. Du Pont De Nemours And Company | Fluorine-containing oil- and water-repellant copolymers |
| US4297145A (en) | 1979-08-17 | 1981-10-27 | Degussa Aktiengesellschaft | Silane/filler preparations, process for their production and their use |
| EP0156270A2 (en) | 1984-03-30 | 1985-10-02 | Th. Goldschmidt AG | Particles superficially modified by hydrophilic and hydrophobic groups |
| US4616058A (en) | 1985-02-01 | 1986-10-07 | Kansai Paint Co., Ltd. | Process for preparing a stable dispersion in an aqueous medium of particles of polymer |
| JPH01172389A (en) | 1987-12-14 | 1989-07-07 | Technicon Instr Corp | Large scale synthesis of 12-membered diaza monocyclic compound |
| US4859754A (en) | 1987-10-20 | 1989-08-22 | Asahi Glass Company, Ltd. | Water and oil repellant having desoiling properties |
| JPH023775A (en) | 1988-06-08 | 1990-01-09 | Nec Corp | Automatic transmission control device for automobile |
| US5173523A (en) | 1985-12-07 | 1992-12-22 | Basf Aktiengesellschaft | Aqueous polymer emulsions and their preparation |
| EP0542598A1 (en) | 1991-11-12 | 1993-05-19 | Elf Atochem S.A. | Fluorinated copolymers and their use for coating and impregnation of various substrates |
| US5319019A (en) | 1991-07-17 | 1994-06-07 | Cargill Incorporated | Acrylic polymer aqueous dispersion with co-solvent |
| WO1996004123A1 (en) | 1994-07-29 | 1996-02-15 | Wilhelm Barthlott | Self-cleaning surfaces of objects and process for producing same |
| US5584921A (en) | 1994-06-03 | 1996-12-17 | Bayer Aktiengesellschaft | Method for the preparation and use of new mixtures for coatings |
| WO1997000230A1 (en) | 1995-06-16 | 1997-01-03 | Minnesota Mining And Manufacturing Company | Water and oil repellent masonry treatments |
| US5599489A (en) | 1993-01-18 | 1997-02-04 | Onoda Cement Co., Ltd. | Preparing molded articles of fluorine-containing polymer with increased water-repellency |
| JPH09225321A (en) | 1995-04-14 | 1997-09-02 | Sekisui Chem Co Ltd | Photocatalyst |
| US5759980A (en) | 1997-03-04 | 1998-06-02 | Blue Coral, Inc. | Car wash |
| EP0862858A1 (en) | 1997-03-06 | 1998-09-09 | Hüls Aktiengesellschaft | Process for preparing antimicrobial plastics |
| US5817370A (en) | 1996-10-09 | 1998-10-06 | Basf Lacke + Farben, Ag | Water-dilutable binders, aqueous coating materials containing these binders, and processes for the priming or one-layer coating of plastics |
| JPH11181339A (en) | 1997-12-22 | 1999-07-06 | Toto Ltd | Hydrophilic coating composition |
| JPH11228873A (en) | 1998-02-16 | 1999-08-24 | Isamu Paint Co Ltd | Titanium dioxide photocatalyst-containing coating material composition |
| EP0942052A1 (en) | 1997-03-14 | 1999-09-15 | Matsushita Electric Works, Ltd. | Antifouling silicone emulsion coating composition, process for producing the same and antifouling article coated therewith |
| US5965659A (en) | 1993-12-29 | 1999-10-12 | Daikin Industries Ltd. | Fluorine-containing-oil-in-water emulsion and surface treatment composition |
| US6013724A (en) | 1997-03-05 | 2000-01-11 | Nippon Paint Co., Ltd. | Raindrop fouling-resistant paint film, coating composition, film-forming method, and coated article |
| JP2000017096A (en) | 1998-07-03 | 2000-01-18 | Achilles Corp | Thermoplastic resin foamed sheet having photocatalytic function |
| WO2000006633A1 (en) | 1998-07-27 | 2000-02-10 | Pilkington Plc | Coating for plastic substrate |
| US6048910A (en) | 1997-02-06 | 2000-04-11 | Shin-Etsu Chemical Co., Ltd. | Coating compositions, hydrophilic films, and hydrophilic film-coated articles |
| JP2000204194A (en) | 1999-01-14 | 2000-07-25 | Otsuka Chem Co Ltd | Resin molded article containing titanium oxide and production of the resin molded article and resin composition containing titanium oxide |
| EP1035184A1 (en) | 1997-11-18 | 2000-09-13 | Daikin Industries, Ltd. | Water-base synthetic resin dispersion composition |
| WO2000058410A1 (en) | 1999-03-25 | 2000-10-05 | Wilhelm Barthlott | Method of producing self-cleaning detachable surfaces |
| EP1106630A1 (en) | 1999-12-06 | 2001-06-13 | Atofina | Fluorocopolymers for oil repelling and waterproofing treatment of various substrates |
| US6277953B1 (en) | 1998-09-25 | 2001-08-21 | Mcwhorter Technologies, Inc. | Stable aqueous polymer dispersions and a process for their preparation |
| DE10008177A1 (en) | 2000-02-23 | 2001-08-30 | Creavis Tech & Innovation Gmbh | Copolymers of allyl triphenylphosphonium salts |
| DE10014726A1 (en) | 2000-03-24 | 2001-09-27 | Creavis Tech & Innovation Gmbh | Antimicrobial coating obtained from aliphatic hydrocarbon-containing monomers useful for medical technology and hygiene items and for protective coatings is permanently antimicrobial and solvent resistant |
| DE10022406A1 (en) | 2000-05-09 | 2001-11-15 | Creavis Tech & Innovation Gmbh | New antimicrobial copolymers used for disinfection of water or production of microbicidal coatings, obtained by copolymerisation of amino-functional acrylate or acrylamide with non-functionalised monomers |
| DE10024270A1 (en) | 2000-05-17 | 2001-11-22 | Creavis Tech & Innovation Gmbh | Antimicrobial polymer production, comprises polymerizing optionally substituted acrylamide monomers, used e.g. for coating medical and hygiene articles |
| US6337129B1 (en) | 1997-06-02 | 2002-01-08 | Toto Ltd. | Antifouling member and antifouling coating composition |
| US20020048679A1 (en) | 1999-01-08 | 2002-04-25 | Gunther Lohmer | Hydrophobicization process for polymeric substrates |
| JP2002294154A (en) | 2001-03-28 | 2002-10-09 | Nisshin Steel Co Ltd | Coating material composition for precoated metal plate having excellent resistance to adhesion of dirt, and precoated metal plate |
| US20020150725A1 (en) | 2001-04-12 | 2002-10-17 | Creavis Gesellschaft Fuer Techn. Und Innov. Mbh | Surfaces rendered self-cleaning by hydrophobic structures, and process for their production |
| US20020150726A1 (en) | 2001-04-12 | 2002-10-17 | Creavis Gesellschaft Fuer Techn. Und Innov. Mbh | Properties of structure-formers for self-cleaning surfaces, and the production of the same |
| US20020150723A1 (en) | 2001-04-12 | 2002-10-17 | Creavis Gesellschaft F. Techn. U. Innovation Mbh | Surfaces which are self-cleaning by hydrophobic structures, and a process for their production |
| US20020150724A1 (en) | 2001-04-12 | 2002-10-17 | Creavis Gesellschaft F. Techn. U. Innovation Mbh | Surfaces rendered self-cleaning by hydrophobic structures, and process for their production |
| US20030013795A1 (en) | 2001-07-16 | 2003-01-16 | Creavis Gesellschaft F. Techn. U. Innovation Mbh | Surfaces rendered self-cleaning by hydrophobic structures and a process for their production |
| JP2003020450A (en) | 2001-07-09 | 2003-01-24 | Nippon Paint Co Ltd | Antifouling coating composition and method for forming coated film |
| US6521730B1 (en) | 1998-06-03 | 2003-02-18 | E.I. Du Pont De Nemours And Company | Fluorinated hydrophilic polymers |
| US20030147932A1 (en) | 2001-08-10 | 2003-08-07 | Creavis Gesellschaft Fuer Tech. Und Innovation Mbh | Self-cleaning lotus effect surfaces having antimicrobial properties |
| US6605351B1 (en) | 1997-09-27 | 2003-08-12 | Gerd Rossmy | Amphiphilic particles or molecules with predominantly hydrophilic and predominantly hydrophobic domains distributed anisotropically on their surface |
| US6635341B1 (en) | 2000-07-31 | 2003-10-21 | Ppg Industries Ohio, Inc. | Coating compositions comprising silyl blocked components, coating, coated substrates and methods related thereto |
| US6683126B2 (en) | 2000-05-08 | 2004-01-27 | Basf Aktiengesellschaft | Compositions for producing difficult-to-wet surface |
| US20040026053A1 (en) | 2000-08-07 | 2004-02-12 | Tembou N'zudie Denis | Novel fluorinated copolymers, their use for coating and impregnating substrates, and resulting treated substrates |
| US20040067247A1 (en) * | 2002-09-27 | 2004-04-08 | Xavier De Sloovere | Composition for combating/repelling insects, birds, dirts and parasites |
| US20040082494A1 (en) | 2001-02-26 | 2004-04-29 | Lionel Queval | Use of amphilic block copolymers in order to increase the water affinity of low-energy surfaces |
| JP2005097608A (en) | 2003-09-04 | 2005-04-14 | Showa Denko Kk | Polyolefin film and its manufacturing process |
| US20050118433A1 (en) | 2002-02-07 | 2005-06-02 | Creavis Gesellschaft Fuer | Method for the production of protective layers with dirt and water repelling properties |
| US6919398B1 (en) | 1998-12-24 | 2005-07-19 | Sto Ag | Forming or coating material and utilization thereof |
| US20060029808A1 (en) * | 2004-08-06 | 2006-02-09 | Lei Zhai | Superhydrophobic coatings |
| US20060058490A1 (en) | 2004-09-15 | 2006-03-16 | Kang Yang G | Films or structural exterior materials using coating composition having self-cleaning property and preparation method thereof |
| US20060110542A1 (en) | 2003-12-18 | 2006-05-25 | Thomas Dietz | Processing compositions and method of forming the same |
| WO2006067638A2 (en) * | 2004-12-20 | 2006-06-29 | Amo Groningen B.V. | Amphiphilic block copolymers and their use |
| JP2006233343A (en) | 2005-02-22 | 2006-09-07 | Nippon Soda Co Ltd | Photocatalyst liquid composition |
| US7196043B2 (en) | 2002-10-23 | 2007-03-27 | S. C. Johnson & Son, Inc. | Process and composition for producing self-cleaning surfaces from aqueous systems |
| WO2007044784A2 (en) | 2005-10-11 | 2007-04-19 | Luna Innovations Incorporated | Self-decontaminating surface coatings and articles prepared therefrom |
| US20070197717A1 (en) | 2004-04-09 | 2007-08-23 | Akihiko Ueda | Polymer for masonry treatment and treating agent |
| WO2008071957A1 (en) | 2006-12-12 | 2008-06-19 | University Of Leeds | Reversible micelles and applications for their use |
| EP1955767A1 (en) | 2005-09-30 | 2008-08-13 | Mitsui Chemicals, Inc. | Photocatalyst-containing organic material |
| US20090018249A1 (en) | 2006-01-30 | 2009-01-15 | Subbareddy Kanagasabapathy | Hydrophobic self-cleaning coating compositions |
| US7521039B2 (en) | 2002-11-08 | 2009-04-21 | Millennium Inorganic Chemicals, Inc. | Photocatalytic rutile titanium dioxide |
| US7544411B2 (en) | 2001-02-10 | 2009-06-09 | Ferro Gmbh | Self-cleaning paint coating and a method and agent for producing the same |
| EP2159573A1 (en) * | 2008-08-28 | 2010-03-03 | Koninklijke Philips Electronics N.V. | 2D electrophoresis device and method of manufacturing |
| US7695814B2 (en) | 2006-02-14 | 2010-04-13 | The Procter & Gamble Company | Responsive coated particles comparing hydrophobic and hydrophilic polymers |
| DE102009045651A1 (en) * | 2008-10-17 | 2010-04-29 | Basf Se | Textile finishing process comprises treating textile materials with an aqueous formulation comprising polyalkylene oxide unit-containing copolymer, which comprises blocks formed from polyethylene oxide and alkylene oxide and anchor block |
| EP2210921A1 (en) | 2009-01-21 | 2010-07-28 | Xerox Corporation | Superhydrophobic nano-fabrics and coatings |
| US7781027B2 (en) | 1997-02-03 | 2010-08-24 | Cytonix Llc | Hydrophobic coating compositions, articles coated with said compositions, and processes for manufacturing same |
| US20100311572A1 (en) | 2007-08-28 | 2010-12-09 | Basf Se | Photoactive tio2 in coating materials |
| US7923106B2 (en) | 2006-02-14 | 2011-04-12 | The Procter & Gamble Company | Responsive coated substrates |
| US20110111659A1 (en) | 2009-11-09 | 2011-05-12 | E. I. Du Pont De Nemours And Company | Method using fluoropolymer emulsions |
| US20110129204A1 (en) | 2008-06-24 | 2011-06-02 | Energy Korea Inc. | Coating composition comprising photocatalyst coated with apatite and radiant heating system having the same |
| US7955430B2 (en) | 2004-04-15 | 2011-06-07 | Sto Ag | Coating material |
| US7964244B2 (en) | 2002-07-13 | 2011-06-21 | Evonik Degussa Gmbh | Method for producing a surfactant-free suspension based on nanostructured, hydrophobic particles, and use of the same |
| US20110312065A1 (en) | 2010-06-21 | 2011-12-22 | Toyota Motor Corporation | Clearcoat containing thermoase c160 for easy-cleaning of insect body stains |
| US20110313095A1 (en) | 2009-02-26 | 2011-12-22 | Basf Se | Self-cleaning polymers |
| US8147607B2 (en) | 2009-10-26 | 2012-04-03 | Ashland Licensing And Intellectual Property Llc | Hydrophobic self-cleaning coating compositions |
| US8187707B2 (en) | 2005-05-25 | 2012-05-29 | Dsm Ip Assets B.V. | Hydrophobic coating |
| WO2012075033A2 (en) | 2010-11-29 | 2012-06-07 | President And Fellows Of Harvard College | Environmentally responsive optical microstructured hybrid actuator assemblies and applications thereof |
| US8202614B2 (en) | 2006-08-09 | 2012-06-19 | Luna Innovations Incorporated | Additive particles having superhydrophobic characteristics and coatings and methods of making and using the same |
| WO2012107612A1 (en) * | 2011-02-08 | 2012-08-16 | Consejo Superior De Investigaciones Científicas (Csic) | Hydrophobic/hydrophilic molecular switch, device containing said switch and method for controlling surface hydrophobicity |
-
2013
- 2013-02-18 WO PCT/IN2013/000101 patent/WO2014097309A1/en active Application Filing
Patent Citations (96)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| CH268258A (en) | 1946-07-30 | 1950-08-16 | Rhone Poulenc Chemicals | Water repellent coating. |
| US3227675A (en) | 1963-05-01 | 1966-01-04 | Huber Corp J M | Silane-treated clay reinforced resin compositions |
| US3354022A (en) | 1964-03-31 | 1967-11-21 | Du Pont | Water-repellant surface |
| DE2107082A1 (en) | 1970-02-16 | 1971-08-26 | Ici Ltd | Surface treated powdered silica and process for the manufacture thereof |
| DE2150736A1 (en) | 1971-10-12 | 1973-04-19 | Hoeveling Fa Emil G V | Outdoor paints - for facades based on a siloxane resin methylmethacrlate copolymer combination |
| DE2161591A1 (en) | 1971-12-11 | 1973-06-14 | Stockhausen & Cie Chem Fab | Glass wetting - eg wind-screen clearing, with aq solns contg cation active high mol wt polyelectrolytes |
| DE2307422A1 (en) | 1972-02-17 | 1973-08-23 | Cabot Corp | COATING COMPOUNDS AND METHOD FOR ITS USE |
| DE2352242A1 (en) | 1973-10-16 | 1975-04-24 | Hoeveling Emil G Von | PIGMENTED PAINT, IN PARTICULAR FAÇADE PROTECTIVE PAINT |
| US4061503A (en) | 1976-09-29 | 1977-12-06 | Union Carbide Corporation | Silane treatment of titanium dioxide pigment |
| US4147851A (en) | 1978-06-13 | 1979-04-03 | E. I. Du Pont De Nemours And Company | Fluorine-containing oil- and water-repellant copolymers |
| US4297145A (en) | 1979-08-17 | 1981-10-27 | Degussa Aktiengesellschaft | Silane/filler preparations, process for their production and their use |
| EP0156270A2 (en) | 1984-03-30 | 1985-10-02 | Th. Goldschmidt AG | Particles superficially modified by hydrophilic and hydrophobic groups |
| US4715986A (en) | 1984-03-30 | 1987-12-29 | Th. Goldschmidt Ag | Particles, modified at their surface by hydrophilic and hydrophobic groups |
| US4616058A (en) | 1985-02-01 | 1986-10-07 | Kansai Paint Co., Ltd. | Process for preparing a stable dispersion in an aqueous medium of particles of polymer |
| US5173523A (en) | 1985-12-07 | 1992-12-22 | Basf Aktiengesellschaft | Aqueous polymer emulsions and their preparation |
| US4859754A (en) | 1987-10-20 | 1989-08-22 | Asahi Glass Company, Ltd. | Water and oil repellant having desoiling properties |
| JPH01172389A (en) | 1987-12-14 | 1989-07-07 | Technicon Instr Corp | Large scale synthesis of 12-membered diaza monocyclic compound |
| JPH023775A (en) | 1988-06-08 | 1990-01-09 | Nec Corp | Automatic transmission control device for automobile |
| US5319019A (en) | 1991-07-17 | 1994-06-07 | Cargill Incorporated | Acrylic polymer aqueous dispersion with co-solvent |
| EP0542598A1 (en) | 1991-11-12 | 1993-05-19 | Elf Atochem S.A. | Fluorinated copolymers and their use for coating and impregnation of various substrates |
| US5599489A (en) | 1993-01-18 | 1997-02-04 | Onoda Cement Co., Ltd. | Preparing molded articles of fluorine-containing polymer with increased water-repellency |
| US5965659A (en) | 1993-12-29 | 1999-10-12 | Daikin Industries Ltd. | Fluorine-containing-oil-in-water emulsion and surface treatment composition |
| US5584921A (en) | 1994-06-03 | 1996-12-17 | Bayer Aktiengesellschaft | Method for the preparation and use of new mixtures for coatings |
| WO1996004123A1 (en) | 1994-07-29 | 1996-02-15 | Wilhelm Barthlott | Self-cleaning surfaces of objects and process for producing same |
| JPH09225321A (en) | 1995-04-14 | 1997-09-02 | Sekisui Chem Co Ltd | Photocatalyst |
| WO1997000230A1 (en) | 1995-06-16 | 1997-01-03 | Minnesota Mining And Manufacturing Company | Water and oil repellent masonry treatments |
| US5817370A (en) | 1996-10-09 | 1998-10-06 | Basf Lacke + Farben, Ag | Water-dilutable binders, aqueous coating materials containing these binders, and processes for the priming or one-layer coating of plastics |
| US7781027B2 (en) | 1997-02-03 | 2010-08-24 | Cytonix Llc | Hydrophobic coating compositions, articles coated with said compositions, and processes for manufacturing same |
| US6048910A (en) | 1997-02-06 | 2000-04-11 | Shin-Etsu Chemical Co., Ltd. | Coating compositions, hydrophilic films, and hydrophilic film-coated articles |
| US5759980A (en) | 1997-03-04 | 1998-06-02 | Blue Coral, Inc. | Car wash |
| US6013724A (en) | 1997-03-05 | 2000-01-11 | Nippon Paint Co., Ltd. | Raindrop fouling-resistant paint film, coating composition, film-forming method, and coated article |
| EP0862858A1 (en) | 1997-03-06 | 1998-09-09 | Hüls Aktiengesellschaft | Process for preparing antimicrobial plastics |
| EP0942052A1 (en) | 1997-03-14 | 1999-09-15 | Matsushita Electric Works, Ltd. | Antifouling silicone emulsion coating composition, process for producing the same and antifouling article coated therewith |
| US6337129B1 (en) | 1997-06-02 | 2002-01-08 | Toto Ltd. | Antifouling member and antifouling coating composition |
| US6605351B1 (en) | 1997-09-27 | 2003-08-12 | Gerd Rossmy | Amphiphilic particles or molecules with predominantly hydrophilic and predominantly hydrophobic domains distributed anisotropically on their surface |
| EP1035184A1 (en) | 1997-11-18 | 2000-09-13 | Daikin Industries, Ltd. | Water-base synthetic resin dispersion composition |
| JPH11181339A (en) | 1997-12-22 | 1999-07-06 | Toto Ltd | Hydrophilic coating composition |
| JPH11228873A (en) | 1998-02-16 | 1999-08-24 | Isamu Paint Co Ltd | Titanium dioxide photocatalyst-containing coating material composition |
| US6521730B1 (en) | 1998-06-03 | 2003-02-18 | E.I. Du Pont De Nemours And Company | Fluorinated hydrophilic polymers |
| JP2000017096A (en) | 1998-07-03 | 2000-01-18 | Achilles Corp | Thermoplastic resin foamed sheet having photocatalytic function |
| WO2000006633A1 (en) | 1998-07-27 | 2000-02-10 | Pilkington Plc | Coating for plastic substrate |
| US6277953B1 (en) | 1998-09-25 | 2001-08-21 | Mcwhorter Technologies, Inc. | Stable aqueous polymer dispersions and a process for their preparation |
| US6919398B1 (en) | 1998-12-24 | 2005-07-19 | Sto Ag | Forming or coating material and utilization thereof |
| US20020048679A1 (en) | 1999-01-08 | 2002-04-25 | Gunther Lohmer | Hydrophobicization process for polymeric substrates |
| JP2000204194A (en) | 1999-01-14 | 2000-07-25 | Otsuka Chem Co Ltd | Resin molded article containing titanium oxide and production of the resin molded article and resin composition containing titanium oxide |
| WO2000058410A1 (en) | 1999-03-25 | 2000-10-05 | Wilhelm Barthlott | Method of producing self-cleaning detachable surfaces |
| EP1106630A1 (en) | 1999-12-06 | 2001-06-13 | Atofina | Fluorocopolymers for oil repelling and waterproofing treatment of various substrates |
| DE10008177A1 (en) | 2000-02-23 | 2001-08-30 | Creavis Tech & Innovation Gmbh | Copolymers of allyl triphenylphosphonium salts |
| DE10014726A1 (en) | 2000-03-24 | 2001-09-27 | Creavis Tech & Innovation Gmbh | Antimicrobial coating obtained from aliphatic hydrocarbon-containing monomers useful for medical technology and hygiene items and for protective coatings is permanently antimicrobial and solvent resistant |
| US6683126B2 (en) | 2000-05-08 | 2004-01-27 | Basf Aktiengesellschaft | Compositions for producing difficult-to-wet surface |
| DE10022406A1 (en) | 2000-05-09 | 2001-11-15 | Creavis Tech & Innovation Gmbh | New antimicrobial copolymers used for disinfection of water or production of microbicidal coatings, obtained by copolymerisation of amino-functional acrylate or acrylamide with non-functionalised monomers |
| DE10024270A1 (en) | 2000-05-17 | 2001-11-22 | Creavis Tech & Innovation Gmbh | Antimicrobial polymer production, comprises polymerizing optionally substituted acrylamide monomers, used e.g. for coating medical and hygiene articles |
| US6635341B1 (en) | 2000-07-31 | 2003-10-21 | Ppg Industries Ohio, Inc. | Coating compositions comprising silyl blocked components, coating, coated substrates and methods related thereto |
| US20040026053A1 (en) | 2000-08-07 | 2004-02-12 | Tembou N'zudie Denis | Novel fluorinated copolymers, their use for coating and impregnating substrates, and resulting treated substrates |
| US7544411B2 (en) | 2001-02-10 | 2009-06-09 | Ferro Gmbh | Self-cleaning paint coating and a method and agent for producing the same |
| US20040082494A1 (en) | 2001-02-26 | 2004-04-29 | Lionel Queval | Use of amphilic block copolymers in order to increase the water affinity of low-energy surfaces |
| JP2002294154A (en) | 2001-03-28 | 2002-10-09 | Nisshin Steel Co Ltd | Coating material composition for precoated metal plate having excellent resistance to adhesion of dirt, and precoated metal plate |
| US20020150725A1 (en) | 2001-04-12 | 2002-10-17 | Creavis Gesellschaft Fuer Techn. Und Innov. Mbh | Surfaces rendered self-cleaning by hydrophobic structures, and process for their production |
| US20020150724A1 (en) | 2001-04-12 | 2002-10-17 | Creavis Gesellschaft F. Techn. U. Innovation Mbh | Surfaces rendered self-cleaning by hydrophobic structures, and process for their production |
| US20020150726A1 (en) | 2001-04-12 | 2002-10-17 | Creavis Gesellschaft Fuer Techn. Und Innov. Mbh | Properties of structure-formers for self-cleaning surfaces, and the production of the same |
| US20020150723A1 (en) | 2001-04-12 | 2002-10-17 | Creavis Gesellschaft F. Techn. U. Innovation Mbh | Surfaces which are self-cleaning by hydrophobic structures, and a process for their production |
| JP2003020450A (en) | 2001-07-09 | 2003-01-24 | Nippon Paint Co Ltd | Antifouling coating composition and method for forming coated film |
| US20030013795A1 (en) | 2001-07-16 | 2003-01-16 | Creavis Gesellschaft F. Techn. U. Innovation Mbh | Surfaces rendered self-cleaning by hydrophobic structures and a process for their production |
| US20030147932A1 (en) | 2001-08-10 | 2003-08-07 | Creavis Gesellschaft Fuer Tech. Und Innovation Mbh | Self-cleaning lotus effect surfaces having antimicrobial properties |
| US20050118433A1 (en) | 2002-02-07 | 2005-06-02 | Creavis Gesellschaft Fuer | Method for the production of protective layers with dirt and water repelling properties |
| US7964244B2 (en) | 2002-07-13 | 2011-06-21 | Evonik Degussa Gmbh | Method for producing a surfactant-free suspension based on nanostructured, hydrophobic particles, and use of the same |
| US20040067247A1 (en) * | 2002-09-27 | 2004-04-08 | Xavier De Sloovere | Composition for combating/repelling insects, birds, dirts and parasites |
| US7196043B2 (en) | 2002-10-23 | 2007-03-27 | S. C. Johnson & Son, Inc. | Process and composition for producing self-cleaning surfaces from aqueous systems |
| US7521039B2 (en) | 2002-11-08 | 2009-04-21 | Millennium Inorganic Chemicals, Inc. | Photocatalytic rutile titanium dioxide |
| JP2005097608A (en) | 2003-09-04 | 2005-04-14 | Showa Denko Kk | Polyolefin film and its manufacturing process |
| US20060110542A1 (en) | 2003-12-18 | 2006-05-25 | Thomas Dietz | Processing compositions and method of forming the same |
| US20070197717A1 (en) | 2004-04-09 | 2007-08-23 | Akihiko Ueda | Polymer for masonry treatment and treating agent |
| US7955430B2 (en) | 2004-04-15 | 2011-06-07 | Sto Ag | Coating material |
| US20060029808A1 (en) * | 2004-08-06 | 2006-02-09 | Lei Zhai | Superhydrophobic coatings |
| US20060058490A1 (en) | 2004-09-15 | 2006-03-16 | Kang Yang G | Films or structural exterior materials using coating composition having self-cleaning property and preparation method thereof |
| WO2006067638A2 (en) * | 2004-12-20 | 2006-06-29 | Amo Groningen B.V. | Amphiphilic block copolymers and their use |
| JP2006233343A (en) | 2005-02-22 | 2006-09-07 | Nippon Soda Co Ltd | Photocatalyst liquid composition |
| US8187707B2 (en) | 2005-05-25 | 2012-05-29 | Dsm Ip Assets B.V. | Hydrophobic coating |
| EP1955767A1 (en) | 2005-09-30 | 2008-08-13 | Mitsui Chemicals, Inc. | Photocatalyst-containing organic material |
| WO2007044784A2 (en) | 2005-10-11 | 2007-04-19 | Luna Innovations Incorporated | Self-decontaminating surface coatings and articles prepared therefrom |
| US20090018249A1 (en) | 2006-01-30 | 2009-01-15 | Subbareddy Kanagasabapathy | Hydrophobic self-cleaning coating compositions |
| US7695814B2 (en) | 2006-02-14 | 2010-04-13 | The Procter & Gamble Company | Responsive coated particles comparing hydrophobic and hydrophilic polymers |
| US7923106B2 (en) | 2006-02-14 | 2011-04-12 | The Procter & Gamble Company | Responsive coated substrates |
| US8202614B2 (en) | 2006-08-09 | 2012-06-19 | Luna Innovations Incorporated | Additive particles having superhydrophobic characteristics and coatings and methods of making and using the same |
| WO2008071957A1 (en) | 2006-12-12 | 2008-06-19 | University Of Leeds | Reversible micelles and applications for their use |
| US20100311572A1 (en) | 2007-08-28 | 2010-12-09 | Basf Se | Photoactive tio2 in coating materials |
| US20110129204A1 (en) | 2008-06-24 | 2011-06-02 | Energy Korea Inc. | Coating composition comprising photocatalyst coated with apatite and radiant heating system having the same |
| EP2159573A1 (en) * | 2008-08-28 | 2010-03-03 | Koninklijke Philips Electronics N.V. | 2D electrophoresis device and method of manufacturing |
| DE102009045651A1 (en) * | 2008-10-17 | 2010-04-29 | Basf Se | Textile finishing process comprises treating textile materials with an aqueous formulation comprising polyalkylene oxide unit-containing copolymer, which comprises blocks formed from polyethylene oxide and alkylene oxide and anchor block |
| EP2210921A1 (en) | 2009-01-21 | 2010-07-28 | Xerox Corporation | Superhydrophobic nano-fabrics and coatings |
| US20110313095A1 (en) | 2009-02-26 | 2011-12-22 | Basf Se | Self-cleaning polymers |
| US8147607B2 (en) | 2009-10-26 | 2012-04-03 | Ashland Licensing And Intellectual Property Llc | Hydrophobic self-cleaning coating compositions |
| US20110111659A1 (en) | 2009-11-09 | 2011-05-12 | E. I. Du Pont De Nemours And Company | Method using fluoropolymer emulsions |
| US20110312065A1 (en) | 2010-06-21 | 2011-12-22 | Toyota Motor Corporation | Clearcoat containing thermoase c160 for easy-cleaning of insect body stains |
| WO2012075033A2 (en) | 2010-11-29 | 2012-06-07 | President And Fellows Of Harvard College | Environmentally responsive optical microstructured hybrid actuator assemblies and applications thereof |
| WO2012107612A1 (en) * | 2011-02-08 | 2012-08-16 | Consejo Superior De Investigaciones Científicas (Csic) | Hydrophobic/hydrophilic molecular switch, device containing said switch and method for controlling surface hydrophobicity |
Non-Patent Citations (17)
| Title |
|---|
| ANDREW R. PARKER, NATURE, vol. 414, 1 November 2001 (2001-11-01), pages 33 - 34 |
| ASAKAWA ET AL., JOURNAL OF FLUORINE CHEMISTRY, vol. 104, no. 1, June 2000 (2000-06-01), pages 47 - 51 |
| BENEE, L. S.; SNOWDEN, M. J.; CHOWDHRY B. Z., LANGMUIR, vol. 18, 2002, pages 6025 |
| CHENG, X.; CANAVAN, H. E.; STEIN, M. J.; HULL, J. R.; KWESKIN, S. J.; WAGNER, M. S.; SOMORJAI, G. A.; CASTNER, D. G.; RATNER, B. D, LANGMUIR, vol. 21, 2005, pages 7833 |
| H. S. LIM; J. T. HAN; D. KWAK; M. JIN; K. CHO, J. AM. CHEM. SOC., vol. 128, 2006, pages 14458 |
| J. LAHANN; S. MITRAGOTRI; T. TRAN; H. KAIDO; J. SUNDARAM; I. S. CHOI; S. HOFFER; G. A. SOMORJAI; R. LANGER, SCIENCE, vol. 17, 2003, pages 371 |
| KJA RAJ ET AL., INDIAN JOURNAL OF CHEMISTRY, vol. 49A, no. 07, July 2010 (2010-07-01), pages 867 |
| L. JIANG; R. WANG; B. YANG; T. LI; D. A. TRYK; A. FUJISHIMA; K. HASHIMOTO; D. ZHU, PURE APPL. CHEM., vol. 72, 2000, pages 73 |
| MIKHAIL MOTORNOV, JOURNAL OF COLLOID AND INTERFACE SCIENCE, vol. 310, 2007, pages 481 - 488 |
| N VERPLANCK, NANOSCALE RES LETT, vol. 2, 2007, pages 577 - 596 |
| RUBNER; COHEN'S, NANO LETT., vol. 6, no. 6, 2006, pages 1213 - 1217 |
| SCIENTIFIC AMERICAN, vol. 299, 2008, pages 88 - 95 |
| SUN, T.; WANG, G.; FENG, L.; LIU, B.; MA, Y.; JIANG, L.; ZHU, D., ANGEW. CHEM., INT. ED., vol. 43, 2004, pages 357 |
| WANG, J.; HU, J.; WEN, Y.; SONG, Y.; JIANG, L., CHEM. MATER., vol. 18, 2006, pages 4984 - 4986 |
| X. FENG; L. FENG; M. JIN; J. ZHAI; L. JIANG; D. ZHU, J. AM. CHEM. SOC., vol. 126, 2004, pages 62 |
| X. YU; Z. WANG; Y. JIANG; F. SHI; X. ZHANG, ADV. MATER., vol. 17, 2005, pages 1289 |
| XIA, F.; FENG, L.; WANG, S.; SUN, T.; SONG, W.; JIANG, W.; JIANG, L., ADV. MATER., vol. 18, 2006, pages 432 |
Cited By (125)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US11498853B2 (en) | 2010-02-10 | 2022-11-15 | Queen's University At Kingston | Water with switchable ionic strength |
| US10377647B2 (en) | 2010-12-15 | 2019-08-13 | Queen's University at Kingson | Systems and methods for use of water with switchable ionic strength |
| US10023827B2 (en) | 2014-07-31 | 2018-07-17 | Jsr Corporation | Cleaning composition for semiconductor substrate and cleaning method |
| US20160032227A1 (en) * | 2014-07-31 | 2016-02-04 | Jsr Corporation | Cleaning composition for semiconductor substrate and cleaning method |
| CN110079169B (en) * | 2014-09-09 | 2021-07-09 | 霍尼韦尔国际公司 | Low VOC and high solids fluoropolymers for coating applications |
| CN110079169A (en) * | 2014-09-09 | 2019-08-02 | 霍尼韦尔国际公司 | Low VOC and high solid fluoropolymer for coating application |
| US10987686B2 (en) | 2014-10-28 | 2021-04-27 | 3M Innovative Properties Company | Spray application system components comprising a repellent surface and methods |
| US10987685B2 (en) | 2014-10-28 | 2021-04-27 | 3M Innovative Properties Company | Spray application system components comprising a repellent surface and methods |
| WO2016088131A1 (en) * | 2014-12-01 | 2016-06-09 | Asian Paints Ltd. | Surfactant stabilized vam-veova10 terpolymer based emulsion with good freeze thaw stability and a process thereof |
| US20170327679A1 (en) * | 2014-12-01 | 2017-11-16 | Asian Paints Ltd. | Surfactant stabilized vam-veova10 terpolymer based emulsion with good freeze thaw stability and a process thereof |
| US10870751B2 (en) | 2014-12-01 | 2020-12-22 | Asian Paints Ltd. | Surfactant stabilized VAM-VeoVa10 terpolymer based emulsion with good freeze thaw stability and a process thereof |
| EP4216258A1 (en) * | 2014-12-19 | 2023-07-26 | Applied Materials, Inc. | Components for a chemical mechanical polishing tool |
| CN106280834A (en) * | 2015-01-12 | 2017-01-04 | 王滨 | The application in concrete for marine works protects of the hydrophobic and oil repellent type protective agent |
| US10533096B2 (en) | 2015-02-27 | 2020-01-14 | Kimberly-Clark Worldwide, Inc. | Non-fluorinated water-based superhydrophobic compositions |
| US10118864B2 (en) | 2015-04-21 | 2018-11-06 | Behr Process Corporation | Decorative coating compositions |
| US9718737B2 (en) | 2015-04-21 | 2017-08-01 | Behr Process Corporation | Decorative coating compositions |
| CN104910792A (en) * | 2015-05-27 | 2015-09-16 | 天津市美冠科技有限公司 | Aqueous UV coating material and preparation method thereof |
| CN104888497A (en) * | 2015-05-28 | 2015-09-09 | 南京理工大学 | Super-hydrophobic and super-oleophylic fluorocarbon resin block and preparation method thereof |
| CN104888497B (en) * | 2015-05-28 | 2016-07-06 | 南京理工大学 | A kind of oleophylic fluorocarbon resin block super-hydrophobic, super and preparation method |
| WO2016191890A1 (en) * | 2015-06-04 | 2016-12-08 | Greencentre Canada | Switchable water-based paint or coating compositions |
| US11236250B2 (en) | 2015-06-04 | 2022-02-01 | Queen's University At Kingston | Switchable water-based paint or coating compositions |
| RU2719471C2 (en) * | 2015-06-24 | 2020-04-17 | ЭйЭм ТЕКНОЛОДЖИ ЛИМИТЕД | Photocatalytic composition based on air binder and use thereof to produce water-based paints, particularly for internal works |
| CN107429119A (en) * | 2015-06-25 | 2017-12-01 | 旭化成株式会社 | Film |
| EP3315571A4 (en) * | 2015-06-25 | 2018-05-09 | Asahi Kasei Kabushiki Kaisha | Coating film |
| AU2015402860B2 (en) * | 2015-07-21 | 2020-06-25 | Rohm And Haas Company | An aqueous coating composition |
| WO2017012033A1 (en) | 2015-07-21 | 2017-01-26 | Rohm And Haas Company | An aqueous coating composition |
| EP3325564A4 (en) * | 2015-07-21 | 2019-03-06 | Rohm and Haas Company | An aqueous coating composition |
| US11136464B2 (en) | 2015-10-28 | 2021-10-05 | 3M Innovative Properties Company | Articles subject to ice formation comprising a repellent surface |
| AU2016346920B2 (en) * | 2015-10-28 | 2020-04-30 | 3M Innovative Properties Company | Spray application system components comprising a repellent surface and methods |
| US10584249B2 (en) | 2015-10-28 | 2020-03-10 | 3M Innovative Properties Company | Articles subject to ice formation comprising a repellent surface |
| WO2017132350A1 (en) | 2016-01-29 | 2017-08-03 | Tolmar Inc. | Purification and decolorization of polymers |
| EP3408305A4 (en) * | 2016-01-29 | 2019-08-28 | Tolmar Inc. | Purification and decolorization of polymers |
| US10556999B2 (en) | 2016-01-29 | 2020-02-11 | Tolmar, Inc. | Purification and decolorization of polymers |
| AU2017255540B9 (en) * | 2016-04-26 | 2020-04-23 | 3M Innovative Properties Company | Spray application systems components comprising a repellent surface comprising a siloxane material and methods |
| AU2017257868B2 (en) * | 2016-04-26 | 2020-05-07 | 3M Innovative Properties Company | Liquid reservoirs and articles comprising a repellent surface comprising a siloxane material |
| US10907070B2 (en) | 2016-04-26 | 2021-02-02 | 3M Innovative Properties Company | Articles subject to ice formation comprising a repellent surface comprising a siloxane material |
| AU2017255540B2 (en) * | 2016-04-26 | 2020-04-09 | 3M Innovative Properties Company | Spray application systems components comprising a repellent surface comprising a siloxane material and methods |
| US10946399B2 (en) | 2016-04-26 | 2021-03-16 | 3M Innovative Properties Company | Liquid reservoirs and articles comprising a repellent surface comprising a siloxane material |
| KR102218338B1 (en) | 2016-05-18 | 2021-02-22 | 피피지 인더스트리즈 오하이오 인코포레이티드 | Water-based composition and compact process of forming multi-component composite coating composition on a substrate |
| WO2017201257A1 (en) * | 2016-05-18 | 2017-11-23 | Ppg Industries Ohio, Inc. | Waterborne compositions and compact processes of forming multi-component composite coating compositions on substrates |
| US10759959B2 (en) | 2016-05-18 | 2020-09-01 | Ppg Industries Ohio, Inc. | Waterborne compositions and compact processes of forming multi-component composite coating compositions on substrates |
| RU2721136C1 (en) * | 2016-05-18 | 2020-05-18 | Ппг Индастриз Огайо, Инк. | Water-soluble compositions and compact methods of producing multicomponent compositions intended for applying composite coatings on substrates |
| KR20190008565A (en) * | 2016-05-18 | 2019-01-24 | 피피지 인더스트리즈 오하이오 인코포레이티드 | A water-based composition, and a compact process for forming a multi-component composite coating composition on a substrate |
| WO2017220591A1 (en) * | 2016-06-20 | 2017-12-28 | Université de Mons | Omniphobic surface coatings |
| EP3279273A1 (en) * | 2016-08-05 | 2018-02-07 | Malgorzata Brzezinska-Kisiel P.P.H. "ALCHEM" | A composition, use of the composition, an impregnant and a method for the preparing the composition |
| WO2018027271A1 (en) * | 2016-08-11 | 2018-02-15 | Guard It Solutions Pty Ltd | Compositions for sealing and/or protecting porous substrates |
| US11806650B2 (en) | 2016-08-16 | 2023-11-07 | Donaldson Company, Inc. | Hydrocarbon fluid-water separation |
| CN114377478B (en) * | 2016-08-16 | 2023-08-22 | 唐纳森公司 | Hydrocarbon Fluid-Water Separation |
| CN114377478A (en) * | 2016-08-16 | 2022-04-22 | 唐纳森公司 | Hydrocarbon fluid-water separation |
| CN106221469A (en) * | 2016-08-26 | 2016-12-14 | 上海绿特丹保温工程有限公司 | A kind of water-water reflective insulation anti-fog haze multicolor finish |
| US11189824B2 (en) | 2016-09-06 | 2021-11-30 | Battrion Ag | Method and apparatus for applying magnetic fields to an article |
| EP3536719A4 (en) * | 2016-11-01 | 2020-07-29 | Unimatec Co., Ltd. | Fluoropolymer and rust preventive containing same as active ingredient |
| CN106554711A (en) * | 2016-11-29 | 2017-04-05 | 国网山东省电力公司电力科学研究院 | Super-amphiphobic coating and preparation method thereof is damaged in polyurethane integration heat resistanceheat resistant |
| US11136472B2 (en) | 2017-01-26 | 2021-10-05 | Sika Technology Ag | Multilayer decorative coating composition with low discolouration |
| WO2018138253A1 (en) * | 2017-01-26 | 2018-08-02 | Sika Technology Ag | Multilayer decorative coating composition with low discolouration |
| CN110325484A (en) * | 2017-02-28 | 2019-10-11 | 法国圣戈班玻璃厂 | The article for moist chamber containing hydrophobicity glass pane |
| WO2018185710A1 (en) * | 2017-04-07 | 2018-10-11 | Sabic Global Technologies B.V. | Durable surface hardened coating or overcoating for protecting plants from pests |
| CN107189603A (en) * | 2017-07-10 | 2017-09-22 | 漳州鑫展旺化工有限公司 | A kind of fire-proof and explosion-proof vehicle glass coating of aqueous high-temperature-resistant |
| US11525068B2 (en) | 2017-10-11 | 2022-12-13 | Build Care Oy | Method of producing an elastic coating and an elastic coating |
| CN109666465A (en) * | 2017-10-17 | 2019-04-23 | 中石化石油工程技术服务有限公司 | A kind of preparation method and drilling fluid of drilling fluid latex sealing agent |
| CN107779030B (en) * | 2017-11-10 | 2019-10-25 | 重庆大学 | A kind of preparation method of high strength alumin ium alloy durability super-double-hydrophobic surface |
| CN107779030A (en) * | 2017-11-10 | 2018-03-09 | 重庆大学 | A kind of preparation method of high strength alumin ium alloy durability super-double-hydrophobic surface |
| US10793737B2 (en) | 2017-12-29 | 2020-10-06 | Behr Process Corporation | Fast dry stain formula |
| CN108248271B (en) * | 2018-02-05 | 2020-03-27 | 河南工学院 | Multicolor woodcut and manufacturing material thereof |
| CN108248271A (en) * | 2018-02-05 | 2018-07-06 | 河南工学院 | A kind of multicolor woodcut and its making material |
| CN112154020A (en) * | 2018-02-15 | 2020-12-29 | 唐纳森公司 | Filter media construction |
| WO2019166899A1 (en) * | 2018-02-28 | 2019-09-06 | Battrion Ag | Method for producing a coating |
| CN111788268A (en) * | 2018-02-28 | 2020-10-16 | 巴璀翁股份有限公司 | Method for the production of coatings |
| CN111954699A (en) * | 2018-04-13 | 2020-11-17 | 宣伟投资管理有限公司 | Coating composition for polymeric roofing materials |
| US11674042B2 (en) | 2018-04-13 | 2023-06-13 | Swimc Llc | Coating compositions for polymeric roofing materials |
| US11084952B2 (en) | 2018-04-18 | 2021-08-10 | Swimc, Llc | Functionalized polymer compositions for low VOC coalescence of water based emulsions |
| EP3556817A1 (en) * | 2018-04-18 | 2019-10-23 | The University of Akron | Functionalized polymer compositions for low voc coalescence of water based emulsions |
| CN109096857A (en) * | 2018-07-26 | 2018-12-28 | 深圳市莱莉雅环保科技有限公司 | The aqueous anti-graffiti coating and its preparation process of scintilla water dispersion |
| CN109161320A (en) * | 2018-08-03 | 2019-01-08 | 苏州东沧涂料科技有限公司 | A kind of aqueous matt coating and preparation method thereof |
| CN109161320B (en) * | 2018-08-03 | 2021-01-19 | 苏州东沧涂料科技有限公司 | Water-based matte coating and preparation method thereof |
| WO2020113005A1 (en) * | 2018-11-29 | 2020-06-04 | Henry Company, Llc | Self-cleaning, dirt pick-up resistant compositions for roof coatings |
| US11680171B2 (en) | 2018-11-29 | 2023-06-20 | Henry Company, Llc | Self-cleaning, dirt pick-up resistant compositions for roof coatings |
| WO2020120006A1 (en) * | 2018-12-14 | 2020-06-18 | Merck Patent Gmbh | Surface coating compositions |
| CN113195645A (en) * | 2018-12-14 | 2021-07-30 | 默克专利股份有限公司 | Surface coating composition |
| CN109374793A (en) * | 2018-12-20 | 2019-02-22 | 北京市环境保护科学研究院 | A method of organic carbon-water partition coefficient of aromatic hydrocarbon in the practical soil of measurement |
| CN109722062A (en) * | 2018-12-21 | 2019-05-07 | 英德科迪颜料技术有限公司 | A kind of preparation method of core-shell structure transparent ferric oxide coated pigment |
| WO2020139601A1 (en) * | 2018-12-26 | 2020-07-02 | Nitto Denko Corporation | Superhydrophilic coating composition |
| WO2020180760A1 (en) * | 2019-03-05 | 2020-09-10 | Board Of Trustees Of Michigan State University | Omniphobic polyurethane compositions, related articles, and related methods |
| US11053337B2 (en) | 2019-03-05 | 2021-07-06 | Board Of Trustees Of Michigan State University | Omniphobic polyurethane compositions, related articles, and related methods |
| CN113631674B (en) * | 2019-03-29 | 2023-03-17 | Ppg工业俄亥俄公司 | Single component hydrophobic coating |
| CN113631674A (en) * | 2019-03-29 | 2021-11-09 | Ppg工业俄亥俄公司 | Single component hydrophobic coating |
| WO2020210988A1 (en) | 2019-04-16 | 2020-10-22 | Dow Global Technologies Llc | Freeze-thaw additive composition |
| EP3956405A4 (en) * | 2019-04-16 | 2022-12-28 | Dow Global Technologies LLC | Freeze-thaw additive composition |
| WO2021003182A1 (en) * | 2019-07-01 | 2021-01-07 | Basf Corporation | Surface modified kaolin pigment and method thereof |
| CN110373076A (en) * | 2019-07-12 | 2019-10-25 | 成都新柯力化工科技有限公司 | A kind of environment protecting building paint and preparation method of photocatalytic degradation haze precursor |
| CN110433671A (en) * | 2019-07-23 | 2019-11-12 | 江苏大学 | A kind of preparation method and its usage of visible light-inducing automatically cleaning carbon-fiber film |
| CN110433671B (en) * | 2019-07-23 | 2021-09-10 | 江苏大学 | Preparation method and application of visible light induced self-cleaning carbon fiber film |
| CN110564237A (en) * | 2019-08-13 | 2019-12-13 | 安徽朗凯奇建材有限公司 | waterproof material for metal roof and preparation method thereof |
| WO2021095043A1 (en) * | 2019-11-17 | 2021-05-20 | The State Of Israel, Ministry Of Agriculture & Rural Development, Agricultural Research Organization (Aro) (Volcani Center) | Superhydrophobic coatings based on pickering emulsions |
| CN111019456A (en) * | 2019-11-27 | 2020-04-17 | 昆明理工大电力工程技术有限公司 | Durable super-hydrophobic three-proofing coating as well as preparation method and use method thereof |
| CN111266679A (en) * | 2019-11-27 | 2020-06-12 | 南京航空航天大学 | Preparation method of self-propelled functional surface and surface tension storage tank based on surface |
| CN111266679B (en) * | 2019-11-27 | 2021-04-02 | 南京航空航天大学 | Preparation method of self-propelled functional surface and surface tension storage tank based on surface |
| WO2021207805A1 (en) * | 2020-04-13 | 2021-10-21 | Bonilha Mariana | Water-based composition for producing an antiviral, antimicrobial coating film |
| CN111454394A (en) * | 2020-04-22 | 2020-07-28 | 昆明理工大学 | Water-based acrylic composite resin and preparation method thereof |
| CN113717557A (en) * | 2020-05-25 | 2021-11-30 | 富思特新材料科技发展股份有限公司 | Self-layering inorganic hydrophobic coating material and preparation method thereof |
| CN111644209A (en) * | 2020-08-05 | 2020-09-11 | 上海山恒生态科技股份有限公司 | Preparation method of photocatalyst for treating organic sewage |
| CN111644209B (en) * | 2020-08-05 | 2020-11-13 | 上海山恒生态科技股份有限公司 | Preparation method of photocatalyst for treating organic sewage |
| CN112159117A (en) * | 2020-10-10 | 2021-01-01 | 东北石油大学 | Method for rapidly preparing underwater super-oleophobic anticorrosive coating |
| CN112852191A (en) * | 2020-12-22 | 2021-05-28 | 湖北大学 | Preparation method of tungsten trioxide super-smooth coating with long-term stability and applicable to mist collection |
| CN112745752A (en) * | 2020-12-29 | 2021-05-04 | 广东邦固化学科技有限公司 | Water-based nano super-amphiphobic coating and preparation method thereof |
| CN112981973A (en) * | 2021-03-19 | 2021-06-18 | 南昌航空大学 | Preparation method of pH response super-hydrophobic material |
| US20220354122A1 (en) * | 2021-05-07 | 2022-11-10 | The University Of Hong Kong | pH/CO2-RESPONSIVE SMART ANTI-PATHOGEN COATINGS THAT CONTROL, REPEL, AND/OR INACTIVATE VIRUSES AND BACTERIA |
| WO2023016657A1 (en) * | 2021-08-13 | 2023-02-16 | Daw Se | Magnetizable aqueous putty, dry preparation for magnetizable coating compositions, magnetizable emulsion paints and uses thereof |
| CN113912936B (en) * | 2021-09-30 | 2023-02-17 | 成都金发科技新材料有限公司 | Toughened and antistatic polypropylene composition and preparation method and application thereof |
| CN113912936A (en) * | 2021-09-30 | 2022-01-11 | 成都金发科技新材料有限公司 | Toughened and antistatic polypropylene composition and preparation method and application thereof |
| CN113897114A (en) * | 2021-10-14 | 2022-01-07 | 江苏利信新型建筑模板有限公司 | Composite green aluminum alloy building template and processing technology thereof |
| CN113897114B (en) * | 2021-10-14 | 2022-08-12 | 江苏利信新型建筑模板有限公司 | Composite green aluminum alloy building template and processing technology thereof |
| CN113856243B (en) * | 2021-10-15 | 2022-06-07 | 山东大学 | Oil-water separation material containing fluorine silsesquioxane modified epoxy resin and preparation method and application thereof |
| CN113856243A (en) * | 2021-10-15 | 2021-12-31 | 山东大学 | Oil-water separation material containing fluorine silsesquioxane modified epoxy resin and preparation method and application thereof |
| CN114292593A (en) * | 2022-01-17 | 2022-04-08 | 山东中凯华瑞工程材料有限公司 | Nano micron and submicron micron spacing structure bionic all-hydrophobic material and preparation method thereof |
| CN115350887A (en) * | 2022-07-25 | 2022-11-18 | 华北理工大学 | Method for preparing super-hydrophobic surface with mechanical durability |
| CN115821640B (en) * | 2022-11-22 | 2023-08-18 | 启东欣联壁纸有限公司 | Waterproof and stain-resistant wallpaper and preparation method thereof |
| CN115821640A (en) * | 2022-11-22 | 2023-03-21 | 启东欣联壁纸有限公司 | Waterproof stain-resistant wallpaper and preparation method thereof |
| CN116179025A (en) * | 2022-12-08 | 2023-05-30 | 江苏金陵特种涂料有限公司 | Water-based carbon nano cold zinc spraying coating and preparation method and application thereof |
| CN116355471A (en) * | 2023-03-31 | 2023-06-30 | 广东晟毅新材料科技有限公司 | Super-hydrophobic coating, super-hydrophobic glass and super-hydrophobic toughened glass and preparation method thereof |
| CN116355471B (en) * | 2023-03-31 | 2024-03-29 | 广东晟毅新材料科技有限公司 | Super-hydrophobic coating, super-hydrophobic glass and super-hydrophobic toughened glass and preparation method thereof |
| CN116891682A (en) * | 2023-09-04 | 2023-10-17 | 北京特思迪半导体设备有限公司 | Modified polydimethylsiloxane amphiphobic coating and preparation method and application thereof |
| CN116891682B (en) * | 2023-09-04 | 2023-11-17 | 北京特思迪半导体设备有限公司 | Modified polydimethylsiloxane amphiphobic coating and preparation method and application thereof |
| CN117363136A (en) * | 2023-10-24 | 2024-01-09 | 江门职业技术学院 | Cross-linked self-repairing coating and preparation method thereof |
| CN117363136B (en) * | 2023-10-24 | 2024-04-09 | 江门职业技术学院 | Cross-linked self-repairing coating and preparation method thereof |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| WO2014097309A1 (en) | Stimuli responsive self cleaning coating | |
| CA2666335C (en) | Improved depolluting coating composition | |
| JP2009541544A (en) | Antifouling coating containing nanoscale hydrophobic particles and method for producing the same | |
| JP2010510338A (en) | Super hydrophobic coating | |
| WO2003102091A1 (en) | Photocatalytic coating material, photocatalytic composite material and process for producing the same, self-cleaning water-based coating compositions, and self-cleaning member | |
| WO2008153687A2 (en) | Hydrophobic self-cleaning coating composition | |
| CA2906367C (en) | Superhydrophilic coating composition | |
| EP2695918A1 (en) | Coating composition for the prevention and/or removal of limescale and/or soap scum | |
| US7138438B2 (en) | Polymeric nanoparticle formulations and their use for improving the dirt pick up resistance of a coating | |
| JPH11116885A (en) | Low-fouling water-based coating material composition | |
| US9023146B2 (en) | Oxidizing agents on pigments | |
| JP2004143452A (en) | Self-cleaning aqueous coating composition and self- cleaning member | |
| Vidales-Herrera et al. | Nanomaterials in coatings: An industrial point of view | |
| KR20030096098A (en) | Polymeric nanoparticle formulations and their use for improving the dirt pick up resistance of a coating | |
| JP2012111689A (en) | Aqueous hybrid binder for joint mortar | |
| Khadem-Hosseini et al. | Durability and mechanical performance of a photo-catalytic water-based nanocomposite coating | |
| KR20050118756A (en) | Water-based ceramic photocatalyst paint and manufacturing method thereof | |
| WO2020205879A1 (en) | Coating compositions for hydrophobic films and articles having hydrophobic surfaces | |
| KR100509562B1 (en) | Waterborne photocatalytic inorganic coating compositions comprising TiCl4 of nano particle | |
| US8961682B2 (en) | Hydrophilic paints using pigments coated with anti-oxidants | |
| US9068099B2 (en) | Hydrophobic coatings that provide renewable hydrophilic surface | |
| JP2004043802A (en) | Polymer nanoparticle composition and its use for microorganism repellent coating | |
| Knight | Recent Advances in Waterborne Acrylic Nanocomposite Paints and Coatings | |
| Sbardella | Synthesis, characterization and application of polyacrylate/silica hybrid films for coatings | |
| CN114026180A (en) | Surface modified kaolin pigments and methods therefor |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| 121 | Ep: the epo has been informed by wipo that ep was designated in this application |
Ref document number: 13723566 Country of ref document: EP Kind code of ref document: A1 |
|
| NENP | Non-entry into the national phase |
Ref country code: DE |
|
| 122 | Ep: pct application non-entry in european phase |
Ref document number: 13723566 Country of ref document: EP Kind code of ref document: A1 |